[English] 日本語
 Yorodumi
Yorodumi- PDB-2co1: Salmonella enterica SafA pilin in complex with a 19-residue SafA ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 2co1 | ||||||
|---|---|---|---|---|---|---|---|
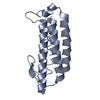
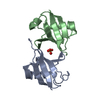
| Title | Salmonella enterica SafA pilin in complex with a 19-residue SafA Nte peptide (F17A mutant) | ||||||
 Components Components | (PUTATIVE OUTER MEMBRANE PROTEIN) x 2 | ||||||
 Keywords Keywords | FIBRIL PROTEIN / PILUS SUBUNIT / FOLD COMPLEMENTATION | ||||||
| Function / homology |  Function and homology information Function and homology informationSaf-pilin pilus formation protein / Saf-pilin pilus formation protein / Dr adhesin / Dr-adhesin superfamily / Adhesion domain superfamily / Prokaryotic membrane lipoprotein lipid attachment site profile. / Immunoglobulin-like / Sandwich / Mainly Beta Similarity search - Domain/homology | ||||||
| Biological species |  SALMONELLA TYPHIMURIUM (bacteria) SALMONELLA TYPHIMURIUM (bacteria) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2.4 Å MOLECULAR REPLACEMENT / Resolution: 2.4 Å | ||||||
 Authors Authors | Remaut, H. / Rose, R.J. / Hannan, T.J. / Hultgren, S.J. / Radford, S.E. / Ashcroft, A.E. / Waksman, G. | ||||||
 Citation Citation |  Journal: Mol.Cell / Year: 2006 Journal: Mol.Cell / Year: 2006Title: Donor-Strand Exchange in Chaperone-Assisted Pilus Assembly Proceeds Through a Concerted Beta-Strand Displacement Mechanism Authors: Remaut, H. / Rose, R.J. / Hannan, T.J. / Hultgren, S.J. / Radford, S.E. / Ashcroft, A.E. / Waksman, G. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  2co1.cif.gz 2co1.cif.gz | 39.2 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb2co1.ent.gz pdb2co1.ent.gz | 27.6 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  2co1.json.gz 2co1.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/co/2co1 https://data.pdbj.org/pub/pdb/validation_reports/co/2co1 ftp://data.pdbj.org/pub/pdb/validation_reports/co/2co1 ftp://data.pdbj.org/pub/pdb/validation_reports/co/2co1 | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  2cnyC  2cnzC  2co2C  2co3C  2co4C  2co6C  2co7C C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
| ||||||||
| Components on special symmetry positions |
| ||||||||
| Details | FOR THE HETERO-ASSEMBLY DESCRIBED BY REMARK 350THE N-TERMINAL EXTENSION PEPTIDE OF ONE SUBUNIT, CHAIN B,INSERTS INTO THE FOLD OF ANOTHER, CHAIN A. THIS INTERACTIONIS REPETED IN THE POLYMER, AN N-TERMINAL EXTENSION OFMOLECULE C WOULD INSERT IN TO THE SUBUNIT OF MOLECULE B , DINTO C ETC |
- Components
Components
| #1: Protein | Mass: 13136.663 Da / Num. of mol.: 1 / Fragment: CORE PILIN DOMAIN, NTE DELETED, RESIDUES 48-170 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  SALMONELLA TYPHIMURIUM (bacteria) / Strain: LT2 / Plasmid: PTRC99A / Production host: SALMONELLA TYPHIMURIUM (bacteria) / Strain: LT2 / Plasmid: PTRC99A / Production host:  |
|---|---|
| #2: Protein/peptide | Mass: 2091.256 Da / Num. of mol.: 1 / Fragment: N-TERMINAL EXTENSION, RESIDUES 27-46 / Mutation: YES / Source method: obtained synthetically / Source: (synth.)  SALMONELLA TYPHIMURIUM (bacteria) / References: UniProt: Q8ZRK4 SALMONELLA TYPHIMURIUM (bacteria) / References: UniProt: Q8ZRK4 |
| #3: Water | ChemComp-HOH / |
| Compound details | ENGINEERED |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.99 Å3/Da / Density % sol: 58.58 % |
|---|---|
| Crystal grow | pH: 4.6 / Details: pH 4.60 |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  ESRF ESRF  / Beamline: ID14-2 / Wavelength: 0.873 / Beamline: ID14-2 / Wavelength: 0.873 |
| Detector | Type: MARRESEARCH / Detector: CCD |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 0.873 Å / Relative weight: 1 |
| Reflection | Resolution: 2.4→20 Å / Num. obs: 13802 / % possible obs: 99.8 % / Observed criterion σ(I): 0 / Redundancy: 5.6 % / Biso Wilson estimate: 45.1 Å2 / Rmerge(I) obs: 0.1 / Net I/σ(I): 13.6 |
| Reflection shell | Resolution: 2.4→2.53 Å / Redundancy: 5.6 % / Rmerge(I) obs: 0.53 / Mean I/σ(I) obs: 3.2 / % possible all: 100 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: SAFA ANTE WT Resolution: 2.4→19.73 Å / Cor.coef. Fo:Fc: 0.935 / Cor.coef. Fo:Fc free: 0.906 / SU B: 15.547 / SU ML: 0.174 / TLS residual ADP flag: LIKELY RESIDUAL / Cross valid method: THROUGHOUT / ESU R: 0.309 / ESU R Free: 0.237 / Stereochemistry target values: MAXIMUM LIKELIHOOD / Details: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS.
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Ion probe radii: 0.8 Å / Shrinkage radii: 0.8 Å / VDW probe radii: 1.4 Å / Solvent model: MASK | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 30.31 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.4→19.73 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller










 PDBj
PDBj




