[English] 日本語
 Yorodumi
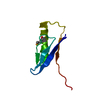
Yorodumi- PDB-1xbl: NMR STRUCTURE OF THE J-DOMAIN (RESIDUES 2-76) IN THE ESCHERICHIA ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1xbl | ||||||
|---|---|---|---|---|---|---|---|
| Title | NMR STRUCTURE OF THE J-DOMAIN (RESIDUES 2-76) IN THE ESCHERICHIA COLI N-TERMINAL FRAGMENT (RESIDUES 2-108) OF THE MOLECULAR CHAPERONE DNAJ, 20 STRUCTURES | ||||||
 Components Components | DNAJ | ||||||
 Keywords Keywords | CHAPERONE / DNA REPLICATION / HEAT SHOCK | ||||||
| Function / homology |  Function and homology information Function and homology informationsigma factor antagonist activity / protein disulfide isomerase activity / protein-disulfide reductase activity / viral process / protein unfolding / heat shock protein binding / : / response to heat / protein refolding / protein folding ...sigma factor antagonist activity / protein disulfide isomerase activity / protein-disulfide reductase activity / viral process / protein unfolding / heat shock protein binding / : / response to heat / protein refolding / protein folding / protein-folding chaperone binding / protein-containing complex assembly / DNA replication / protein homodimerization activity / protein-containing complex / zinc ion binding / ATP binding / membrane / cytoplasm / cytosol Similarity search - Function | ||||||
| Biological species |  | ||||||
| Method | SOLUTION NMR / REDAC | ||||||
 Authors Authors | Pellecchia, M. / Szyperski, T. / Wall, D. / Georgopoulos, C. / Wuthrich, K. | ||||||
 Citation Citation |  Journal: J.Mol.Biol. / Year: 1996 Journal: J.Mol.Biol. / Year: 1996Title: NMR structure of the J-domain and the Gly/Phe-rich region of the Escherichia coli DnaJ chaperone. Authors: Pellecchia, M. / Szyperski, T. / Wall, D. / Georgopoulos, C. / Wuthrich, K. #1:  Journal: Proc.Natl.Acad.Sci.USA / Year: 1994 Journal: Proc.Natl.Acad.Sci.USA / Year: 1994Title: NMR Structure Determination of the Escherichia Coli Dnaj Molecular Chaperone: Secondary Structure and Backbone Fold of the N-Terminal Region (Residues 2-108) Containing the Highly Conserved J Domain Authors: Szyperski, T. / Pellecchia, M. / Wall, D. / Georgopoulos, C. / Wuthrich, K. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1xbl.cif.gz 1xbl.cif.gz | 545.1 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1xbl.ent.gz pdb1xbl.ent.gz | 460.5 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1xbl.json.gz 1xbl.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/xb/1xbl https://data.pdbj.org/pub/pdb/validation_reports/xb/1xbl ftp://data.pdbj.org/pub/pdb/validation_reports/xb/1xbl ftp://data.pdbj.org/pub/pdb/validation_reports/xb/1xbl | HTTPS FTP |
|---|
-Related structure data
| Similar structure data |
|---|
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 |
| |||||||||
| NMR ensembles |
|
- Components
Components
| #1: Protein | Mass: 11846.043 Da / Num. of mol.: 1 Fragment: N-TERMINAL FRAGMENT (RESIDUES 2-108) OF THE MOLECULAR CHAPERONE DNAJ Source method: isolated from a genetically manipulated source Source: (gene. exp.)   |
|---|
-Experimental details
-Experiment
| Experiment | Method: SOLUTION NMR |
|---|---|
| NMR experiment | Type: SEE PAPER *JRNL* |
- Sample preparation
Sample preparation
| Sample conditions | pH: 6 / Temperature: 301 K |
|---|---|
| Crystal grow | *PLUS Method: other / Details: NMR |
-NMR measurement
| NMR spectrometer | Type: Bruker AMX / Manufacturer: Bruker / Model: AMX / Field strength: 600 MHz |
|---|
- Processing
Processing
| NMR software |
| |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method: REDAC / Software ordinal: 1 | |||||||||
| NMR ensemble | Conformer selection criteria: TARGET FUNCTION (SEE PAPER *JRNL*) Conformers calculated total number: 50 / Conformers submitted total number: 20 |
 Movie
Movie Controller
Controller









 PDBj
PDBj


