[English] 日本語
 Yorodumi
Yorodumi- EMDB-7045: BbRAGL-3'TIR synaptic complex with nicked DNA refined with c1 symmetry -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-7045 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | BbRAGL-3'TIR synaptic complex with nicked DNA refined with c1 symmetry | |||||||||
 Map data Map data | BbRAGL-31TIR synaptic complex | |||||||||
 Sample Sample |
| |||||||||
| Biological species |  | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 5.0 Å | |||||||||
 Authors Authors | Zhang Y / Cheng TC / Xiong Y / Schatz DG | |||||||||
 Citation Citation |  Journal: Nature / Year: 2019 Journal: Nature / Year: 2019Title: Transposon molecular domestication and the evolution of the RAG recombinase. Authors: Yuhang Zhang / Tat Cheung Cheng / Guangrui Huang / Qingyi Lu / Marius D Surleac / Jeffrey D Mandell / Pierre Pontarotti / Andrei J Petrescu / Anlong Xu / Yong Xiong / David G Schatz /     Abstract: Domestication of a transposon (a DNA sequence that can change its position in a genome) to give rise to the RAG1-RAG2 recombinase (RAG) and V(D)J recombination, which produces the diverse repertoire ...Domestication of a transposon (a DNA sequence that can change its position in a genome) to give rise to the RAG1-RAG2 recombinase (RAG) and V(D)J recombination, which produces the diverse repertoire of antibodies and T cell receptors, was a pivotal event in the evolution of the adaptive immune system of jawed vertebrates. The evolutionary adaptations that transformed the ancestral RAG transposase into a RAG recombinase with appropriately regulated DNA cleavage and transposition activities are not understood. Here, beginning with cryo-electron microscopy structures of the amphioxus ProtoRAG transposase (an evolutionary relative of RAG), we identify amino acid residues and domains the acquisition or loss of which underpins the propensity of RAG for coupled cleavage, its preference for asymmetric DNA substrates and its inability to perform transposition in cells. In particular, we identify two adaptations specific to jawed-vertebrates-arginine 848 in RAG1 and an acidic region in RAG2-that together suppress RAG-mediated transposition more than 1,000-fold. Our findings reveal a two-tiered mechanism for the suppression of RAG-mediated transposition, illuminate the evolution of V(D)J recombination and provide insight into the principles that govern the molecular domestication of transposons. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_7045.map.gz emd_7045.map.gz | 20.8 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-7045-v30.xml emd-7045-v30.xml emd-7045.xml emd-7045.xml | 13.1 KB 13.1 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_7045.png emd_7045.png | 155.9 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-7045 http://ftp.pdbj.org/pub/emdb/structures/EMD-7045 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-7045 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-7045 | HTTPS FTP |
-Related structure data
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_7045.map.gz / Format: CCP4 / Size: 22.2 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_7045.map.gz / Format: CCP4 / Size: 22.2 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | BbRAGL-31TIR synaptic complex | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.35 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
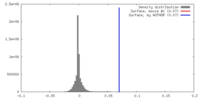
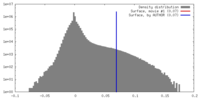
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : BbRAGL-31TIR synaptic complex with nicked DNA
| Entire | Name: BbRAGL-31TIR synaptic complex with nicked DNA |
|---|---|
| Components |
|
-Supramolecule #1: BbRAGL-31TIR synaptic complex with nicked DNA
| Supramolecule | Name: BbRAGL-31TIR synaptic complex with nicked DNA / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#5 |
|---|---|
| Source (natural) | Organism:  |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 350 KDa |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.4 mg/mL |
|---|---|
| Buffer | pH: 7.6 |
| Grid | Model: C-flat-1.2/1.3 4C / Material: COPPER / Mesh: 400 / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Atmosphere: AIR |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 283 K / Instrument: FEI VITROBOT MARK III |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Specialist optics | Energy filter - Name: GIF Quantum LS / Energy filter - Lower energy threshold: 0 eV / Energy filter - Upper energy threshold: 20 eV |
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: SUPER-RESOLUTION / Digitization - Dimensions - Width: 7676 pixel / Digitization - Dimensions - Height: 7420 pixel / Digitization - Sampling interval: 5.0 µm / Digitization - Frames/image: 3-40 / Number grids imaged: 1 / Number real images: 4429 / Average exposure time: 10.0 sec. / Average electron dose: 54.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 0.01 mm / Nominal defocus max: 2.5 µm / Nominal defocus min: 1.2 µm / Nominal magnification: 81000 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Refinement | Space: REAL / Protocol: FLEXIBLE FIT |
|---|
 Movie
Movie Controller
Controller














 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)