+ データを開く
データを開く
- 基本情報
基本情報
| 登録情報 |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| タイトル | ATR-ATRIP bound with ATPgammaS | |||||||||
 マップデータ マップデータ | ||||||||||
 試料 試料 |
| |||||||||
 キーワード キーワード | ATR-ATRIP ATPgammaS / CELL CYCLE | |||||||||
| 機能・相同性 |  機能・相同性情報 機能・相同性情報ATR-ATRIP complex / establishment of RNA localization to telomere / positive regulation of telomerase catalytic core complex assembly / MutSalpha complex binding / establishment of protein-containing complex localization to telomere / histone H2AXS139 kinase activity / nuclear membrane disassembly / MutLalpha complex binding / response to arsenic-containing substance / mitotic G2/M transition checkpoint ...ATR-ATRIP complex / establishment of RNA localization to telomere / positive regulation of telomerase catalytic core complex assembly / MutSalpha complex binding / establishment of protein-containing complex localization to telomere / histone H2AXS139 kinase activity / nuclear membrane disassembly / MutLalpha complex binding / response to arsenic-containing substance / mitotic G2/M transition checkpoint / regulation of double-strand break repair / nucleobase-containing compound metabolic process / positive regulation of DNA damage response, signal transduction by p53 class mediator / protein localization to chromosome, telomeric region / HDR through Single Strand Annealing (SSA) / K63-linked polyubiquitin modification-dependent protein binding / Impaired BRCA2 binding to RAD51 / negative regulation of DNA replication / replication fork processing / site of DNA damage / Presynaptic phase of homologous DNA pairing and strand exchange / replicative senescence / Regulation of HSF1-mediated heat shock response / response to mechanical stimulus / interstrand cross-link repair / Activation of ATR in response to replication stress / regulation of cellular response to heat / positive regulation of telomere maintenance via telomerase / telomere maintenance / Meiotic synapsis / DNA damage checkpoint signaling / TP53 Regulates Transcription of DNA Repair Genes / Fanconi Anemia Pathway / cellular response to gamma radiation / G2/M DNA damage checkpoint / PML body / cellular response to UV / nuclear envelope / double-strand break repair / chromosome / Processing of DNA double-strand break ends / Regulation of TP53 Activity through Phosphorylation / DNA replication / non-specific serine/threonine protein kinase / protein kinase activity / response to xenobiotic stimulus / protein serine kinase activity / DNA repair / protein serine/threonine kinase activity / DNA damage response / Golgi apparatus / DNA binding / nucleoplasm / ATP binding / nucleus 類似検索 - 分子機能 | |||||||||
| 生物種 |  Homo sapiens (ヒト) Homo sapiens (ヒト) | |||||||||
| 手法 | 単粒子再構成法 / クライオ電子顕微鏡法 / 解像度: 6.22 Å | |||||||||
 データ登録者 データ登録者 | Wang G | |||||||||
| 資金援助 |  中国, 1件 中国, 1件
| |||||||||
 引用 引用 |  ジャーナル: Sci Bull (Beijing) / 年: 2025 ジャーナル: Sci Bull (Beijing) / 年: 2025タイトル: Molecular architecture and inhibition mechanism of human ATR-ATRIP. 著者: Guangxian Wang / Po Wang / Zexuan Zheng / Qingjun Zhang / Chenchen Xu / Xinyi Xu / Lingfei Jian / Zhanpeng Zhao / Gang Cai / Xuejuan Wang /  要旨: The ataxia telangiectasia-mutated and Rad3-related (ATR) kinase is a master regulator of DNA damage response and replication stress in humans. Targeting ATR is the focus of oncology drug pipelines ...The ataxia telangiectasia-mutated and Rad3-related (ATR) kinase is a master regulator of DNA damage response and replication stress in humans. Targeting ATR is the focus of oncology drug pipelines with a number of potent, selective ATR inhibitors currently in clinical development. Here, we determined the cryo-EM structures of the human ATR-ATRIP complex in the presence of VE-822 and RP-3500, two ATR inhibitors currently in Phase II clinical trials, achieving an overall resolution of approximately 3 Å. These structures yield a near-complete atomic model of the ATR-ATRIP complex, revealing subunit stoichiometry, intramolecular and intermolecular interactions, and critical regulatory sites including an insertion in the PIKK regulatory domain (PRD). Structural comparison provides insights into the modes of action and selectivity of ATR inhibitors. The divergent binding modes near the solvent side and in the rear pocket area of VE-822 and RP-3500, particularly their disparate binding orientations, lead to varying conformational changes in the active site. Surprisingly, one ATR-ATRIP complex binds four VE-822 molecules, with two in the ATR active site and two at the ATR-ATR dimer interface. The binding and selectivity of RP-3500 depend on two bound water molecules, which may be further enhanced by the substitution of these bound waters. Our study provides a structural framework for understanding ATR regulation and holds promise for assisting future efforts in rational drug design targeting ATR. | |||||||||
| 履歴 |
|
- 構造の表示
構造の表示
| 添付画像 |
|---|
- ダウンロードとリンク
ダウンロードとリンク
-EMDBアーカイブ
| マップデータ |  emd_62812.map.gz emd_62812.map.gz | 85.9 MB |  EMDBマップデータ形式 EMDBマップデータ形式 | |
|---|---|---|---|---|
| ヘッダ (付随情報) |  emd-62812-v30.xml emd-62812-v30.xml emd-62812.xml emd-62812.xml | 19.4 KB 19.4 KB | 表示 表示 |  EMDBヘッダ EMDBヘッダ |
| 画像 |  emd_62812.png emd_62812.png | 105.5 KB | ||
| Filedesc metadata |  emd-62812.cif.gz emd-62812.cif.gz | 7.9 KB | ||
| その他 |  emd_62812_half_map_1.map.gz emd_62812_half_map_1.map.gz emd_62812_half_map_2.map.gz emd_62812_half_map_2.map.gz | 84.7 MB 84.7 MB | ||
| アーカイブディレクトリ |  http://ftp.pdbj.org/pub/emdb/structures/EMD-62812 http://ftp.pdbj.org/pub/emdb/structures/EMD-62812 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-62812 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-62812 | HTTPS FTP |
-検証レポート
| 文書・要旨 |  emd_62812_validation.pdf.gz emd_62812_validation.pdf.gz | 1.1 MB | 表示 |  EMDB検証レポート EMDB検証レポート |
|---|---|---|---|---|
| 文書・詳細版 |  emd_62812_full_validation.pdf.gz emd_62812_full_validation.pdf.gz | 1.1 MB | 表示 | |
| XML形式データ |  emd_62812_validation.xml.gz emd_62812_validation.xml.gz | 13.2 KB | 表示 | |
| CIF形式データ |  emd_62812_validation.cif.gz emd_62812_validation.cif.gz | 15.6 KB | 表示 | |
| アーカイブディレクトリ |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-62812 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-62812 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-62812 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-62812 | HTTPS FTP |
-関連構造データ
| 関連構造データ |  9l4fMC  9l40C  9l43C  9l45C  9l46C  9l4bC  9l4cC  9l4dC M: このマップから作成された原子モデル C: 同じ文献を引用 ( |
|---|---|
| 類似構造データ | 類似検索 - 機能・相同性  F&H 検索 F&H 検索 |
- リンク
リンク
| EMDBのページ |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| 「今月の分子」の関連する項目 |
- マップ
マップ
| ファイル |  ダウンロード / ファイル: emd_62812.map.gz / 形式: CCP4 / 大きさ: 91.1 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) ダウンロード / ファイル: emd_62812.map.gz / 形式: CCP4 / 大きさ: 91.1 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 投影像・断面図 | 画像のコントロール
画像は Spider により作成 | ||||||||||||||||||||||||||||||||||||
| ボクセルのサイズ | X=Y=Z: 1.07 Å | ||||||||||||||||||||||||||||||||||||
| 密度 |
| ||||||||||||||||||||||||||||||||||||
| 対称性 | 空間群: 1 | ||||||||||||||||||||||||||||||||||||
| 詳細 | EMDB XML:
|
-添付データ
-ハーフマップ: #2
| ファイル | emd_62812_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 投影像・断面図 |
| ||||||||||||
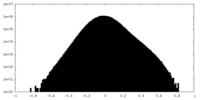
| 密度ヒストグラム |
-ハーフマップ: #1
| ファイル | emd_62812_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 投影像・断面図 |
| ||||||||||||
| 密度ヒストグラム |
- 試料の構成要素
試料の構成要素
-全体 : ATR-ATRIP
| 全体 | 名称: ATR-ATRIP |
|---|---|
| 要素 |
|
-超分子 #1: ATR-ATRIP
| 超分子 | 名称: ATR-ATRIP / タイプ: cell / ID: 1 / 親要素: 0 / 含まれる分子: #1-#2 |
|---|---|
| 由来(天然) | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
-分子 #1: Serine/threonine-protein kinase ATR
| 分子 | 名称: Serine/threonine-protein kinase ATR / タイプ: protein_or_peptide / ID: 1 / コピー数: 2 / 光学異性体: LEVO / EC番号: non-specific serine/threonine protein kinase |
|---|---|
| 由来(天然) | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
| 分子量 | 理論値: 301.756781 KDa |
| 組換発現 | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
| 配列 | 文字列: MGEHGLELAS MIPALRELGS ATPEEYNTVV QKPRQILCQF IDRILTDVNV VAVELVKKTD SQPTSVMLLD FIQHIMKSSP LMFVNVSGS HEAKGSCIEF SNWIITRLLR IAATPSCHLL HKKICEVICS LLFLFKSKSP AIFGVLTKEL LQLFEDLVYL H RRNVMGHA ...文字列: MGEHGLELAS MIPALRELGS ATPEEYNTVV QKPRQILCQF IDRILTDVNV VAVELVKKTD SQPTSVMLLD FIQHIMKSSP LMFVNVSGS HEAKGSCIEF SNWIITRLLR IAATPSCHLL HKKICEVICS LLFLFKSKSP AIFGVLTKEL LQLFEDLVYL H RRNVMGHA VEWPVVMSRF LSQLDEHMGY LQSAPLQLMS MQNLEFIEVT LLMVLTRIIA IVFFRRQELL LWQIGCVLLE YG SPKIKSL AISFLTELFQ LGGLPAQPAS TFFSSFLELL KHLVEMDTDQ LKLYEEPLSK LIKTLFPFEA EAYRNIEPVY LNM LLEKLC VMFEDGVLMR LKSDLLKAAL CHLLQYFLKF VPAGYESALQ VRKVYVRNIC KALLDVLGIE VDAEYLLGPL YAAL KMESM EIIEEIQCQT QQENLSSNSD GISPKRRRLS SSLNPSKRAP KQTEEIKHVD MNQKSILWSA LKQKAESLQI SLEYS GLKN PVIEMLEGIA VVLQLTALCT VHCSHQNMNC RTFKDCQHKS KKKPSVVITW MSLDFYTKVL KSCRSLLESV QKLDLE ATI DKVVKIYDAL IYMQVNSSFE DHILEDLCGM LSLPWIYSHS DDGCLKLTTF AANLLTLSCR ISDSYSPQAQ SRCVFLL TL FPRRIFLEWR TAVYNWALQS SHEVIRASCV SGFFILLQQQ NSCNRVPKIL IDKVKDDSDI VKKEFASILG QLVCTLHG M FYLTSSLTEP FSEHGHVDLF CRNLKATSQH ECSSSQLKAS VCKPFLFLLK KKIPSPVKLA FIDNLHHLCK HLDFREDET DVKAVLGTLL NLMEDPDKDV RVAFSGNIKH ILESLDSEDG FIKELFVLRM KEAYTHAQIS RNNELKDTLI LTTGDIGRAA KGDLVPFAL LHLLHCLLSK SASVSGAAYT EIRALVAAKS VKLQSFFSQY KKPICQFLVE SLHSSQMTAL PNTPCQNADV R KQDVAHQR EMALNTLSEI ANVFDFPDLN RFLTRTLQVL LPDLAAKASP AASALIRTLG KQLNVNRREI LINNFKYIFS HL VCSCSKD ELERALHYLK NETEIELGSL LRQDFQGLHN ELLLRIGEHY QQVFNGLSIL ASFASSDDPY QGPRDIISPE LMA DYLQPK LLGILAFFNM QLLSSSVGIE DKKMALNSLM SLMKLMGPKH VSSVRVKMMT TLRTGLRFKD DFPELCCRAW DCFV RCLDH ACLGSLLSHV IVALLPLIHI QPKETAAIFH YLIIENRDAV QDFLHEIYFL PDHPELKKIK AVLQEYRKET SESTD LQTT LQLSMKAIQH ENVDVRIHAL TSLKETLYKN QEKLIKYATD SETVEPIISQ LVTVLLKGCQ DANSQARLLC GECLGE LGA IDPGRLDFST TETQGKDFTF VTGVEDSSFA YGLLMELTRA YLAYADNSRA QDSAAYAIQE LLSIYDCREM ETNGPGH QL WRRFPEHVRE ILEPHLNTRY KSSQKSTDWS GVKKPIYLSK LGSNFAEWSA SWAGYLITKV RHDLASKIFT CCSIMMKH D FKVTIYLLPH ILVYVLLGCN QEDQQEVYAE IMAVLKHDDQ HTINTQDIAS DLCQLSTQTV FSMLDHLTQW ARHKFQALK AEKCPHSKSN RNKVDSMVST VDYEDYQSVT RFLDLIPQDT LAVASFRSKA YTRAVMHFES FITEKKQNIQ EHLGFLQKLY AAMHEPDGV AGVSAIRKAE PSLKEQILEH ESLGLLRDAT ACYDRAIQLE PDQIIHYHGV VKSMLGLGQL STVITQVNGV H ANRSEWTD ELNTYRVEAA WKLSQWDLVE NYLAADGKST TWSVRLGQLL LSAKKRDITA FYDSLKLVRA EQIVPLSAAS FE RGSYQRG YEYIVRLHML CELEHSIKPL FQHSPGDSSQ EDSLNWVARL EMTQNSYRAK EPILALRRAL LSLNKRPDYN EMV GECWLQ SARVARKAGH HQTAYNALLN AGESRLAELY VERAKWLWSK GDVHQALIVL QKGVELCFPE NETPPEGKNM LIHG RAMLL VGRFMEETAN FESNAIMKKY KDVTACLPEW EDGHFYLAKY YDKLMPMVTD NKMEKQGDLI RYIVLHFGRS LQYGN QFIY QSMPRMLTLW LDYGTKAYEW EKAGRSDRVQ MRNDLGKINK VITEHTNYLA PYQFLTAFSQ LISRICHSHD EVFVVL MEI IAKVFLAYPQ QAMWMMTAVS KSSYPMRVNR CKEILNKAIH MKKSLEKFVG DATRLTDKLL ELCNKPVDGS SSTLSMS TH FKMLKKLVEE ATFSEILIPL QSVMIPTLPS ILGTHANHAS HEPFPGHWAY IAGFDDMVEI LASLQKPKKI SLKGSDGK F YIMMCKPKDD LRKDCRLMEF NSLINKCLRK DAESRRRELH IRTYAVIPLN DECGIIEWVN NTAGLRPILT KLYKEKGVY MTGKELRQCM LPKSAALSEK LKVFREFLLP RHPPIFHEWF LRTFPDPTSW YSSRSAYCRS TAVMSMVGYI LGLGDRHGEN ILFDSLTGE CVHVDFNCLF NKGETFEVPE IVPFRLTHNM VNGMGPMGTE GLFRRACEVT MRLMRDQREP LMSVLKTFLH D PLVEWSKP VKGHSKAPLN ETGEVVNEKA KTHVLDIEQR LQGVIKTRNR VTGLPLSIEG HVHYLIQEAT DENLLCQMYL GW TPYM UniProtKB: Serine/threonine-protein kinase ATR |
-分子 #2: ATR-interacting protein
| 分子 | 名称: ATR-interacting protein / タイプ: protein_or_peptide / ID: 2 / コピー数: 2 / 光学異性体: LEVO |
|---|---|
| 由来(天然) | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
| 分子量 | 理論値: 85.940664 KDa |
| 組換発現 | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
| 配列 | 文字列: MAGTSAPGSK RRSEPPAPRP GPPPGTGHPP SKRARGFSAA AAPDPDDPFG AHGDFTADDL EELDTLASQA LSQCPAAARD VSSDHKVHR LLDGMSKNPS GKNRETVPIK DNFELEVLQA QYKELKEKMK VMEEEVLIKN GEIKILRDSL HQTESVLEEQ R RSHFLLEQ ...文字列: MAGTSAPGSK RRSEPPAPRP GPPPGTGHPP SKRARGFSAA AAPDPDDPFG AHGDFTADDL EELDTLASQA LSQCPAAARD VSSDHKVHR LLDGMSKNPS GKNRETVPIK DNFELEVLQA QYKELKEKMK VMEEEVLIKN GEIKILRDSL HQTESVLEEQ R RSHFLLEQ EKTQALSDKE KEFSKKLQSL QSELQFKDAE MNELRTKLQT SERANKLAAP SVSHVSPRKN PSVVIKPEAC SP QFGKTSF PTKESFSANM SLPHPCQTES GYKPLVGRED SKPHSLRGDS IKQEEAQKSF VDSWRQRSNT QGSILINLLL KQP LIPGSS LSLCHLLSSS SESPAGTPLQ PPGFGSTLAG MSGLRTTGSY DGSFSLSALR EAQNLAFTGL NLVARNECSR DGDP AEGGR RAFPLCQLPG AVHFLPLVQF FIGLHCQALQ DLAAAKRSGA PGDSPTHSSC VSSGVETNPE DSVCILEGFS VTALS ILQH LVCHSGAVVS LLLSGVGADS AAGEGNRSLV HRLSDGDMTS ALRGVADDQG QHPLLKMLLH LLAFSSAATG HLQASV LTQ CLKVLVKLAE NTSCDFLPRF QCVFQVLPKC LSPETPLPSV LLAVELLSLL ADHDQLAPQL CSHSEGCLLL LLYMYIT SR PDRVALETQW LQLEQEVVWL LAKLGVQSPL PPVTGSNCQC NVEVVRALTV MLHRQWLTVR RAGGPPRTDQ QRRTVRCL R DTVLLLHGLS QKDKLFMMHC VEVLHQFDQV MPGVSMLIRG LPDVTDCEEA ALDDLCAAET DVEDPEVECG UniProtKB: ATR-interacting protein |
-分子 #3: PHOSPHOTHIOPHOSPHORIC ACID-ADENYLATE ESTER
| 分子 | 名称: PHOSPHOTHIOPHOSPHORIC ACID-ADENYLATE ESTER / タイプ: ligand / ID: 3 / コピー数: 2 / 式: AGS |
|---|---|
| 分子量 | 理論値: 523.247 Da |
| Chemical component information |  ChemComp-AGS: |
-実験情報
-構造解析
| 手法 | クライオ電子顕微鏡法 |
|---|---|
 解析 解析 | 単粒子再構成法 |
| 試料の集合状態 | particle |
- 試料調製
試料調製
| 緩衝液 | pH: 7.5 |
|---|---|
| 凍結 | 凍結剤: ETHANE |
- 電子顕微鏡法
電子顕微鏡法
| 顕微鏡 | TFS KRIOS |
|---|---|
| 撮影 | フィルム・検出器のモデル: GATAN K3 BIOQUANTUM (6k x 4k) 平均電子線量: 50.0 e/Å2 |
| 電子線 | 加速電圧: 300 kV / 電子線源:  FIELD EMISSION GUN FIELD EMISSION GUN |
| 電子光学系 | 照射モード: FLOOD BEAM / 撮影モード: BRIGHT FIELD / 最大 デフォーカス(公称値): 2.5 µm / 最小 デフォーカス(公称値): 1.5 µm |
| 実験機器 |  モデル: Titan Krios / 画像提供: FEI Company |
 ムービー
ムービー コントローラー
コントローラー

























 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)