[English] 日本語
 Yorodumi
Yorodumi- EMDB-53529: Cytochrome bd II oxidase qOR-2 type from Mycobacterium smegmatis -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cytochrome bd II oxidase qOR-2 type from Mycobacterium smegmatis | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | MEMBRANE PROTEIN / ACTINOBACTERIA / ELECTRON TRANSPORT | |||||||||
| Function / homology |  Function and homology information Function and homology informationcytochrome complex / aerobic electron transport chain / oxidoreductase activity, acting on diphenols and related substances as donors, oxygen as acceptor / electron transfer activity / heme binding / metal ion binding / plasma membrane Similarity search - Function | |||||||||
| Biological species |  Mycolicibacterium smegmatis (bacteria) Mycolicibacterium smegmatis (bacteria) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 2.8 Å | |||||||||
 Authors Authors | Kovalova T / Janczak M / Adelroth P / Hogbom M | |||||||||
| Funding support |  Sweden, 1 items Sweden, 1 items
| |||||||||
 Citation Citation |  Journal: Proc Natl Acad Sci U S A / Year: 2026 Journal: Proc Natl Acad Sci U S A / Year: 2026Title: The -II terminal oxidase employs a carboxylate shift mechanism. Authors: Terezia Kovalova / Mateusz Janczak / Ana P Gamiz-Hernandez / Daniel Lundin / Soni Sharma / Johanna Vilhjálmsdóttir / Dan Sjöstrand / Ville R I Kaila / Martin Högbom / Pia Ädelroth /  Abstract: Cytochrome is a terminal oxidase expressed under low oxygen conditions and central for the survival of many pathogens. Here, we characterize the cyt -II from , a member of a hitherto uncharacterized ...Cytochrome is a terminal oxidase expressed under low oxygen conditions and central for the survival of many pathogens. Here, we characterize the cyt -II from , a member of a hitherto uncharacterized evolutionary group (qOR-2) of oxidases, by combining biochemical studies with cryo-electron microscopy (cryo-EM), and multiscale simulations. Overexpressing the operon in its native host led to production of a highly active -II ( = 30 e s) that together with a high-resolution (2.8 Å) cryo-EM structure and multiscale simulations reveal unique proton pathways and oxygen channels responsible for its function. We propose that a pH-dependent molecular switch, involving coordination changes of heme and surrounding bulky residues regulate substrate access into the active site. Taken together, our findings provide detailed mechanistic insight of qOR-2 type oxidases, and a basis for understanding the evolution of the superfamily. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_53529.map.gz emd_53529.map.gz | 87.7 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-53529-v30.xml emd-53529-v30.xml emd-53529.xml emd-53529.xml | 23.9 KB 23.9 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_53529.png emd_53529.png | 67.8 KB | ||
| Filedesc metadata |  emd-53529.cif.gz emd-53529.cif.gz | 6.9 KB | ||
| Others |  emd_53529_additional_1.map.gz emd_53529_additional_1.map.gz emd_53529_half_map_1.map.gz emd_53529_half_map_1.map.gz emd_53529_half_map_2.map.gz emd_53529_half_map_2.map.gz | 167.8 MB 165.1 MB 165.1 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-53529 http://ftp.pdbj.org/pub/emdb/structures/EMD-53529 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-53529 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-53529 | HTTPS FTP |
-Related structure data
| Related structure data |  9r2gMC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_53529.map.gz / Format: CCP4 / Size: 178 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_53529.map.gz / Format: CCP4 / Size: 178 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.65 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Additional map: #1
| File | emd_53529_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #2
| File | emd_53529_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #1
| File | emd_53529_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Cytochrome bd II oxidase
| Entire | Name: Cytochrome bd II oxidase |
|---|---|
| Components |
|
-Supramolecule #1: Cytochrome bd II oxidase
| Supramolecule | Name: Cytochrome bd II oxidase / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#2 |
|---|---|
| Source (natural) | Organism:  Mycolicibacterium smegmatis (bacteria) Mycolicibacterium smegmatis (bacteria) |
-Macromolecule #1: Cytochrome bd menaquinol oxidase subunit I
| Macromolecule | Name: Cytochrome bd menaquinol oxidase subunit I / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Mycolicibacterium smegmatis (bacteria) Mycolicibacterium smegmatis (bacteria) |
| Molecular weight | Theoretical: 50.327875 KDa |
| Recombinant expression | Organism:  Mycolicibacterium smegmatis (bacteria) Mycolicibacterium smegmatis (bacteria) |
| Sequence | String: MVFTETLLLL AADGEPPGLL PARQQMAFSL GWHIVLACFG VAFPTMIFVV HRRGIVRDDA VALGLAQRWA KVSAVLFAIG AVSGTVLSF EMGLLWPGLM GRFGDVLGLP FAFEGLSFFV EAIFLGIYLY GWGRMPPRRH LLTLIPMGLA GIVGTFCVVS V NAWMNNPA ...String: MVFTETLLLL AADGEPPGLL PARQQMAFSL GWHIVLACFG VAFPTMIFVV HRRGIVRDDA VALGLAQRWA KVSAVLFAIG AVSGTVLSF EMGLLWPGLM GRFGDVLGLP FAFEGLSFFV EAIFLGIYLY GWGRMPPRRH LLTLIPMGLA GIVGTFCVVS V NAWMNNPA GFRIVNGEVV DIDPWRAMFN SGVWLQFAHM WVAAFMLVGL VVSGVYAFGM LRGRVDTHHR LGFAVPFTFA SV AAVAQPL IGHVLGMRIH DTQPAKLAAF ELAQTTEGPA PLRLGGVLID GEVHWALTIP RLGSIIARNS LDAPVPGLDG VPR SDWPPV NITHLAFQSM VGIGTLLAAV AVVYWLARWR GRDLLANRWF LRLSVITGPL AVLAVESGWV ATEVGRQPWT VWKV LTTTE AASQSSGLWW SYVIVLVVYL GMTIGAVVVL RSMARRWRAG ETDLPSPYGP PRTEARS UniProtKB: Cytochrome bd menaquinol oxidase subunit I |
-Macromolecule #2: Cytochrome bd-I oxidase subunit II
| Macromolecule | Name: Cytochrome bd-I oxidase subunit II / type: protein_or_peptide / ID: 2 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Mycolicibacterium smegmatis (bacteria) Mycolicibacterium smegmatis (bacteria) |
| Molecular weight | Theoretical: 39.166078 KDa |
| Recombinant expression | Organism:  Mycolicibacterium smegmatis (bacteria) Mycolicibacterium smegmatis (bacteria) |
| Sequence | String: MTQATFVAMA MFLGVVIYAL FAGADFGSGF YDLTAGDARS GAKVRTLVDH SIGPVWEANH VWLIYILVIW WTGFPRTFAA ATTTLFIPL ALALTGIVLR GASFAFRKYS ATVSQARLFG AIFAASSLIS PFFLGTVAGA IASGRVPAEG YGDRIGSWLN P TSLVGGFL ...String: MTQATFVAMA MFLGVVIYAL FAGADFGSGF YDLTAGDARS GAKVRTLVDH SIGPVWEANH VWLIYILVIW WTGFPRTFAA ATTTLFIPL ALALTGIVLR GASFAFRKYS ATVSQARLFG AIFAASSLIS PFFLGTVAGA IASGRVPAEG YGDRIGSWLN P TSLVGGFL AVATCVFLAG VFLTADAARS GDNGLADSLR RRTLAVGVVT GLIVFAGLYP VAHDAPTLTA GLRTYAAPLL VI ALLAGVA TVWLVFRRRY AISRIPAAVA VAAVVTGWGV GQYPWLLVDE VTIADAAGAD ATLTGLLIVV VLAGVIVLPA LAY LLRLTQ TEEWSRKLSG GSAENLYFQG WSHPQFEKGG GSGGGSGGSA WSHPQFEK UniProtKB: Cytochrome bd-I oxidase subunit II |
-Macromolecule #3: CARDIOLIPIN
| Macromolecule | Name: CARDIOLIPIN / type: ligand / ID: 3 / Number of copies: 1 / Formula: CDL |
|---|---|
| Molecular weight | Theoretical: 1.464043 KDa |
| Chemical component information |  ChemComp-CDL: |
-Macromolecule #4: CIS-HEME D HYDROXYCHLORIN GAMMA-SPIROLACTONE
| Macromolecule | Name: CIS-HEME D HYDROXYCHLORIN GAMMA-SPIROLACTONE / type: ligand / ID: 4 / Number of copies: 1 / Formula: HDD |
|---|---|
| Molecular weight | Theoretical: 632.487 Da |
| Chemical component information |  ChemComp-HDD: |
-Macromolecule #5: HEME B/C
| Macromolecule | Name: HEME B/C / type: ligand / ID: 5 / Number of copies: 2 / Formula: HEB |
|---|---|
| Molecular weight | Theoretical: 618.503 Da |
| Chemical component information |  ChemComp-HEB: |
-Macromolecule #6: MENAQUINONE-9
| Macromolecule | Name: MENAQUINONE-9 / type: ligand / ID: 6 / Number of copies: 3 / Formula: MQ9 |
|---|---|
| Molecular weight | Theoretical: 785.233 Da |
| Chemical component information | 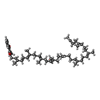 ChemComp-MQ9: |
-Macromolecule #7: water
| Macromolecule | Name: water / type: ligand / ID: 7 / Number of copies: 7 / Formula: HOH |
|---|---|
| Molecular weight | Theoretical: 18.015 Da |
| Chemical component information |  ChemComp-HOH: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.4 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | TFS KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Average electron dose: 93.4 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.2 µm / Nominal defocus min: 0.6 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller



 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)












































