[English] 日本語
 Yorodumi
Yorodumi- EMDB-52782: Nitrogenase maturase NifEN in complex with the cofactor chaperone NifX -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Nitrogenase maturase NifEN in complex with the cofactor chaperone NifX | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | nitrogenase cofactor maturase / METAL BINDING PROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationnitrogenase activity / 4 iron, 4 sulfur cluster binding / protein-containing complex assembly / metal ion binding Similarity search - Function | |||||||||
| Biological species |  Azotobacter vinelandii DJ (bacteria) Azotobacter vinelandii DJ (bacteria) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 2.16 Å | |||||||||
 Authors Authors | Schneider FF / Martin del Campo JS / Zhang L / Dean DR / Einsle O | |||||||||
| Funding support | European Union,  Germany, 2 items Germany, 2 items
| |||||||||
 Citation Citation |  Journal: Nat Chem Biol / Year: 2026 Journal: Nat Chem Biol / Year: 2026Title: Trafficking of a nitrogenase FeMo-cofactor assembly intermediate. Authors: Florian F Schneider / Julia S Martin Del Campo / Lin Zhang / Dennis R Dean / Oliver Einsle /    Abstract: The maturation of the unique FeMo-cofactor of molybdenum nitrogenase is a multistep process requiring the sequential action of a series of maturase complexes. As a final step, the NifEN complex forms ...The maturation of the unique FeMo-cofactor of molybdenum nitrogenase is a multistep process requiring the sequential action of a series of maturase complexes. As a final step, the NifEN complex forms FeMo-cofactor from the precursor NifB-co, also called L-cluster, through replacement of an apical iron ion by molybdenum and the attachment of an organic homocitrate ligand. NifB-co is delivered by a small cofactor chaperone, NifX, and initially bound near the surface of the maturase NifEN. Here, we report high-resolution cryo-electron microscopy structures of NifEN in complex with NifX, showing NifB-co binding to NifEN in full detail, capturing both interacting partners in the act of cluster transfer. In a dynamic transfer complex, the large metal cluster is coordinated by single residues from both NifEN and NifX. In silico studies concur with these structures but suggest a third, internal conversion site where cluster maturation likely takes place. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_52782.map.gz emd_52782.map.gz | 49.1 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-52782-v30.xml emd-52782-v30.xml emd-52782.xml emd-52782.xml | 26.2 KB 26.2 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_52782.png emd_52782.png | 73.7 KB | ||
| Masks |  emd_52782_msk_1.map emd_52782_msk_1.map | 52.7 MB |  Mask map Mask map | |
| Filedesc metadata |  emd-52782.cif.gz emd-52782.cif.gz | 6.9 KB | ||
| Others |  emd_52782_additional_1.map.gz emd_52782_additional_1.map.gz emd_52782_half_map_1.map.gz emd_52782_half_map_1.map.gz emd_52782_half_map_2.map.gz emd_52782_half_map_2.map.gz | 49.1 MB 49 MB 49 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-52782 http://ftp.pdbj.org/pub/emdb/structures/EMD-52782 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-52782 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-52782 | HTTPS FTP |
-Related structure data
| Related structure data |  9ianMC  9iaoC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_52782.map.gz / Format: CCP4 / Size: 52.7 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_52782.map.gz / Format: CCP4 / Size: 52.7 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.7627 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Mask #1
| File |  emd_52782_msk_1.map emd_52782_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Additional map: #1
| File | emd_52782_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #2
| File | emd_52782_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #1
| File | emd_52782_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Ternary complex of NifENX for nitrogenase ion-molybdenum cofactor...
| Entire | Name: Ternary complex of NifENX for nitrogenase ion-molybdenum cofactor biosynthesis |
|---|---|
| Components |
|
-Supramolecule #1: Ternary complex of NifENX for nitrogenase ion-molybdenum cofactor...
| Supramolecule | Name: Ternary complex of NifENX for nitrogenase ion-molybdenum cofactor biosynthesis type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#3 |
|---|---|
| Source (natural) | Organism:  Azotobacter vinelandii DJ (bacteria) Azotobacter vinelandii DJ (bacteria) |
-Macromolecule #1: Nitrogenase iron-molybdenum cofactor biosynthesis protein NifE
| Macromolecule | Name: Nitrogenase iron-molybdenum cofactor biosynthesis protein NifE type: protein_or_peptide / ID: 1 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Azotobacter vinelandii DJ (bacteria) Azotobacter vinelandii DJ (bacteria) |
| Molecular weight | Theoretical: 50.551762 KDa |
| Recombinant expression | Organism:  Azotobacter vinelandii DJ (bacteria) Azotobacter vinelandii DJ (bacteria) |
| Sequence | String: MKAKDIAELL DEPACSHNKK EKSGCAKPKP GATDGGCSFD GAQIALLPVA DVAHIVHGPI ACAGSSWDNR GTRSSGPDLY RIGMTTDLT ENDVIMGRAE KRLFHAIRQA VESYSPPAVF VYNTCVPALI GDDVDAVCKA AAERFGTPVI PVDSAGFYGT K NLGNRIAG ...String: MKAKDIAELL DEPACSHNKK EKSGCAKPKP GATDGGCSFD GAQIALLPVA DVAHIVHGPI ACAGSSWDNR GTRSSGPDLY RIGMTTDLT ENDVIMGRAE KRLFHAIRQA VESYSPPAVF VYNTCVPALI GDDVDAVCKA AAERFGTPVI PVDSAGFYGT K NLGNRIAG EAMLKYVIGT REPDPLPVGS ERPGIRVHDV NLIGEYNIAG EFWHVLPLLD ELGLRVLCTL AGDARYREVQ TM HRAEVNM MVCSKAMLNV ARKLQETYGT PWFEGSFYGI TDTSQALRDF ARLLDDPDLT ARTEALIARE EAKVRAALEP WRA RLEGKR VLLYTGGVKS WSVVSALQDL GMKVVATGTK KSTEEDKARI RELMGDDVKM LDEGNARVLL KTVDEYQADI LIAG GRNMY TALKGRVPFL DINQEREFGY AGYDGMLELV RQLCITLECP VWEAVRRPAP WDIPA UniProtKB: Nitrogenase iron-molybdenum cofactor biosynthesis protein NifE |
-Macromolecule #2: Nitrogenase iron-molybdenum cofactor biosynthesis protein NifN
| Macromolecule | Name: Nitrogenase iron-molybdenum cofactor biosynthesis protein NifN type: protein_or_peptide / ID: 2 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Azotobacter vinelandii DJ (bacteria) Azotobacter vinelandii DJ (bacteria) |
| Molecular weight | Theoretical: 48.463977 KDa |
| Recombinant expression | Organism:  Azotobacter vinelandii DJ (bacteria) Azotobacter vinelandii DJ (bacteria) |
| Sequence | String: AEIINRNKAL AVSPLKASQT MGAALAILGL ARSMPLFHGS QGCTAFAKVF FVRHFREPVP LQTTAMDQVS SVMGADENVV EALKTICER QNPSVIGLLT TGLSETQGCD LHTALHEFRT QYEEYKDVPI VPVNTPDFSG CFESGFAAAV KAIVETLVPE R RDQVGKRP ...String: AEIINRNKAL AVSPLKASQT MGAALAILGL ARSMPLFHGS QGCTAFAKVF FVRHFREPVP LQTTAMDQVS SVMGADENVV EALKTICER QNPSVIGLLT TGLSETQGCD LHTALHEFRT QYEEYKDVPI VPVNTPDFSG CFESGFAAAV KAIVETLVPE R RDQVGKRP RQVNVLCSAN LTPGDLEYIA ESIESFGLRP LLIPDLSGSL DGHLDENRFN ALTTGGLSVA ELATAGQSVA TL VVGQSLA GAADALAERT GVPDRRFGML YGLDAVDAWL MALAEISGNP VPDRYKRQRA QLQDAMLDTH FMLSSARTAI AAD PDLLLG FDALLRSMGA HTVAAVVPAR AAALVDSPLP SVRVGDLEDL EHAARAGQAQ LVIGNSHALA SARRLGVPLL RAGF PQYDL LGGFQRCWSG YRGSSQVLFD LANLLVEHHQ GIQPYHSIYA QKPATEQ UniProtKB: Nitrogenase iron-molybdenum cofactor biosynthesis protein NifN |
-Macromolecule #3: Nitrogenase MoFe cofactor biosynthesis protein NifX
| Macromolecule | Name: Nitrogenase MoFe cofactor biosynthesis protein NifX / type: protein_or_peptide / ID: 3 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Azotobacter vinelandii DJ (bacteria) Azotobacter vinelandii DJ (bacteria) |
| Molecular weight | Theoretical: 17.30751 KDa |
| Recombinant expression | Organism:  Azotobacter vinelandii DJ (bacteria) Azotobacter vinelandii DJ (bacteria) |
| Sequence | String: MSSPTRQLQV LDSEDDGTLL KVAFASSDRE LVDQHFGSSR SFAIYGVNPE RSQLLSVVEF GELEQDGNED KLARKIDLLD GCVAVYCCA CGASAVRQLM AIGVQPIKVS EGARIAELIE ALQVELREGP SAWLAKAIQR TRGPDMRRFD AMAAEGWDE UniProtKB: Nitrogenase MoFe cofactor biosynthesis protein NifX |
-Macromolecule #4: FeFe cofactor
| Macromolecule | Name: FeFe cofactor / type: ligand / ID: 4 / Number of copies: 2 / Formula: S5Q |
|---|---|
| Molecular weight | Theoretical: 747.356 Da |
| Chemical component information | 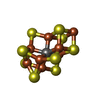 ChemComp-S5Q: |
-Macromolecule #5: IRON/SULFUR CLUSTER
| Macromolecule | Name: IRON/SULFUR CLUSTER / type: ligand / ID: 5 / Number of copies: 2 / Formula: SF4 |
|---|---|
| Molecular weight | Theoretical: 351.64 Da |
| Chemical component information |  ChemComp-FS1: |
-Macromolecule #6: MAGNESIUM ION
| Macromolecule | Name: MAGNESIUM ION / type: ligand / ID: 6 / Number of copies: 2 / Formula: MG |
|---|---|
| Molecular weight | Theoretical: 24.305 Da |
-Macromolecule #7: water
| Macromolecule | Name: water / type: ligand / ID: 7 / Number of copies: 779 / Formula: HOH |
|---|---|
| Molecular weight | Theoretical: 18.015 Da |
| Chemical component information |  ChemComp-HOH: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 8 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | TFS KRIOS |
|---|---|
| Image recording | Film or detector model: FEI FALCON IV (4k x 4k) / Average electron dose: 40.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: SPOT SCAN / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.0 µm / Nominal defocus min: 1.0 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller





 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)




















































