[English] 日本語
 Yorodumi
Yorodumi- EMDB-49800: CryoEM analysis of Phosphoglucose isomerase from P. aeruginosa re... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | CryoEM analysis of Phosphoglucose isomerase from P. aeruginosa reveals potential clinically relevant features | |||||||||
 Map data Map data | Sharpened Map for Model Building | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Phosphoglucose Isomerase / STRUCTURAL PROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationglucose-6-phosphate isomerase / glucose-6-phosphate isomerase activity / glucose 6-phosphate metabolic process / carbohydrate derivative binding / monosaccharide binding / glycolytic process / gluconeogenesis / cytosol Similarity search - Function | |||||||||
| Biological species |   Pseudomonas aeruginosa PAO1 (bacteria) Pseudomonas aeruginosa PAO1 (bacteria) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 1.45 Å | |||||||||
 Authors Authors | Sharma K / Borgnia JM | |||||||||
| Funding support |  United States, 1 items United States, 1 items
| |||||||||
 Citation Citation |  Journal: To Be Published Journal: To Be PublishedTitle: CryoEM analysis of Phosphoglucose isomerase from P. aeruginosa reveals potential clinically relevant features Authors: Sharma K / Borgnia JM | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_49800.map.gz emd_49800.map.gz | 51 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-49800-v30.xml emd-49800-v30.xml emd-49800.xml emd-49800.xml | 21.9 KB 21.9 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_49800_fsc.xml emd_49800_fsc.xml | 20.8 KB | Display |  FSC data file FSC data file |
| Images |  emd_49800.png emd_49800.png | 86.5 KB | ||
| Masks |  emd_49800_msk_1.map emd_49800_msk_1.map | 1000 MB |  Mask map Mask map | |
| Filedesc metadata |  emd-49800.cif.gz emd-49800.cif.gz | 6.5 KB | ||
| Others |  emd_49800_additional_1.map.gz emd_49800_additional_1.map.gz emd_49800_half_map_1.map.gz emd_49800_half_map_1.map.gz emd_49800_half_map_2.map.gz emd_49800_half_map_2.map.gz | 503.5 MB 928.8 MB 928.8 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-49800 http://ftp.pdbj.org/pub/emdb/structures/EMD-49800 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-49800 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-49800 | HTTPS FTP |
-Related structure data
| Related structure data |  9nucMC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_49800.map.gz / Format: CCP4 / Size: 55.4 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_49800.map.gz / Format: CCP4 / Size: 55.4 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Sharpened Map for Model Building | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. generated in cubic-lattice coordinate | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.675 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Mask #1
| File |  emd_49800_msk_1.map emd_49800_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Additional map: Unsharpened Map
| File | emd_49800_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Unsharpened Map | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: Half Map A
| File | emd_49800_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half Map A | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: Half Map B
| File | emd_49800_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half Map B | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Oligomeric structure of PGI
| Entire | Name: Oligomeric structure of PGI |
|---|---|
| Components |
|
-Supramolecule #1: Oligomeric structure of PGI
| Supramolecule | Name: Oligomeric structure of PGI / type: organelle_or_cellular_component / ID: 1 / Parent: 0 / Macromolecule list: #1 |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 650 KDa |
-Macromolecule #1: Glucose-6-phosphate isomerase
| Macromolecule | Name: Glucose-6-phosphate isomerase / type: protein_or_peptide / ID: 1 / Number of copies: 10 / Enantiomer: LEVO / EC number: glucose-6-phosphate isomerase |
|---|---|
| Source (natural) | Organism:  Pseudomonas aeruginosa PAO1 (bacteria) Pseudomonas aeruginosa PAO1 (bacteria) |
| Molecular weight | Theoretical: 62.275504 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: GSMKHHLTPL DATQLDSWRA LAAHRQELQD FRMRQAFIDD PERFKRFSFS ACGLFLDFSK NLIRQDTIDL LVKLAEEARL SDAIRAMFD GEAINASERR PVLHTALRRP IGDKVLVDGV DVMPEVHRVL HQMTELVGYV HNGLWRGYTE KPITDVVNIG I GGSFLGPQ ...String: GSMKHHLTPL DATQLDSWRA LAAHRQELQD FRMRQAFIDD PERFKRFSFS ACGLFLDFSK NLIRQDTIDL LVKLAEEARL SDAIRAMFD GEAINASERR PVLHTALRRP IGDKVLVDGV DVMPEVHRVL HQMTELVGYV HNGLWRGYTE KPITDVVNIG I GGSFLGPQ LVSEALLPFA QKGVRCHYLA NIDGSEFHEL ASRLNAETTL FIVSSKSFGT LETLKNAQAA RAWYLAQGGT EE ELYRHFI AVSSNKEAAI AFGIREENIF PMWDWVGGRY SLWSAIGLPI AMSIGISNFK ELLSGAYNMD QHFQTAPFER NIP VLLGLL GVWYGDFWGA NSHAILPYDY YLRNITDHLQ QLDMESNGKS VRQDGTPVTS GTGPVIWGGV GCNGQHAYHQ LLHQ GTQLI PADFIVPVSS YNPVADHHQW LYANCLSQSQ ALMLGKSREE AEAELRAKGL PEAEVQRLAP HKVIPGNRPS NTLVV ERIS ARRLGALIAM YEHKVYVQSI LWGINAFDQW GVELGKELGK GVYSRLVGSE ETPAEDASTQ GLIDFFRGRH RGL UniProtKB: Glucose-6-phosphate isomerase |
-Macromolecule #2: 6-PHOSPHOGLUCONIC ACID
| Macromolecule | Name: 6-PHOSPHOGLUCONIC ACID / type: ligand / ID: 2 / Number of copies: 10 / Formula: 6PG |
|---|---|
| Molecular weight | Theoretical: 276.135 Da |
| Chemical component information | 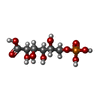 ChemComp-6PG: |
-Macromolecule #3: water
| Macromolecule | Name: water / type: ligand / ID: 3 / Number of copies: 6750 / Formula: HOH |
|---|---|
| Molecular weight | Theoretical: 18.015 Da |
| Chemical component information |  ChemComp-HOH: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 1.3 mg/mL | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 8 Component:
| |||||||||
| Grid | Model: Quantifoil / Material: GOLD / Support film - Material: GOLD / Support film - topology: HOLEY / Pretreatment - Type: PLASMA CLEANING / Pretreatment - Time: 75 sec. | |||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 95 % / Chamber temperature: 283 K / Instrument: LEICA EM GP |
- Electron microscopy
Electron microscopy
| Microscope | TFS KRIOS |
|---|---|
| Temperature | Min: 79.0 K / Max: 79.0 K |
| Specialist optics | Energy filter - Name: GIF Bioquantum / Energy filter - Slit width: 20 eV |
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Digitization - Dimensions - Width: 5760 pixel / Digitization - Dimensions - Height: 4092 pixel / Average exposure time: 0.75 sec. / Average electron dose: 60.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 50.0 µm / Calibrated magnification: 215000 / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 1.6 µm / Nominal defocus min: 0.2 µm / Nominal magnification: 215000 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Refinement | Protocol: AB INITIO MODEL |
|---|---|
| Output model |  PDB-9nuc: |
 Movie
Movie Controller
Controller





 X (Sec.)
X (Sec.) Y (Row.)
Y (Row.) Z (Col.)
Z (Col.)





















































