[English] 日本語
 Yorodumi
Yorodumi- EMDB-49207: Cryo-EM structure of quaternary complex of human phosphoribosylgl... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of quaternary complex of human phosphoribosylglycinamidine synthase with thioester intermediate bound (at glutaminase site) and AMPPNP and FGAR (at synthase site). | |||||||||
 Map data Map data | structure of quaternary complex of human phosphoribosylglycinamidine synthase with thioester intermediate bound (at glutaminase site) and AMPPNP and FGAR (at synthase site). | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | quaternary complex phosphoribosylglycinamidine synthase FGAR Thioester AMPPNP / LIGASE | |||||||||
| Function / homology |  Function and homology information Function and homology informationanterior head development / phosphoribosylformylglycinamidine synthase / phosphoribosylformylglycinamidine synthase activity / purine ribonucleoside monophosphate biosynthetic process / 'de novo' XMP biosynthetic process / Purine ribonucleoside monophosphate biosynthesis / 'de novo' AMP biosynthetic process / L-glutamine metabolic process / purine nucleotide biosynthetic process / GMP biosynthetic process ...anterior head development / phosphoribosylformylglycinamidine synthase / phosphoribosylformylglycinamidine synthase activity / purine ribonucleoside monophosphate biosynthetic process / 'de novo' XMP biosynthetic process / Purine ribonucleoside monophosphate biosynthesis / 'de novo' AMP biosynthetic process / L-glutamine metabolic process / purine nucleotide biosynthetic process / GMP biosynthetic process / 'de novo' IMP biosynthetic process / response to xenobiotic stimulus / extracellular exosome / ATP binding / metal ion binding / cytoplasm / cytosol Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.19 Å | |||||||||
 Authors Authors | Sharma N / French JB | |||||||||
| Funding support |  United States, 2 items United States, 2 items
| |||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2026 Journal: Nat Commun / Year: 2026Title: Structural and molecular basis for allosteric regulation and catalytic coupling of human phosphoribosylformylglycinamidine synthase. Authors: Nandini Sharma / Weijie Zhou / Jarrod B French /  Abstract: Purine nucleotides are ubiquitous molecules essential for all life. The de novo biosynthesis of purines is a metabolic dependency that is frequently reprogrammed in cancers and is a well-established ...Purine nucleotides are ubiquitous molecules essential for all life. The de novo biosynthesis of purines is a metabolic dependency that is frequently reprogrammed in cancers and is a well-established target for chemotherapies, immune modulation and antivirals. Here, we report cryo-electron microscopy structures of the multi-domain human phosphoribosylformylglycinamidine synthase, a central purine biosynthetic enzyme and foundational feature of the purinosome metabolon. These data capture, the proposed iminophosphate intermediate and provide the structural elucidation of an ammonia channel connecting the active sites of the glutaminase and synthase domains. Analysis of this series of structures and the accompanying biochemical data also reveal the molecular features and transient conformational changes that underlie allosteric regulation and catalytic coupling of the domains. This data resolves several longstanding mechanistic questions about this enzyme class and provides a strong foundation for therapeutic development. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_49207.map.gz emd_49207.map.gz | 109.4 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-49207-v30.xml emd-49207-v30.xml emd-49207.xml emd-49207.xml | 24.3 KB 24.3 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_49207_fsc.xml emd_49207_fsc.xml | 12 KB | Display |  FSC data file FSC data file |
| Images |  emd_49207.png emd_49207.png | 81.5 KB | ||
| Filedesc metadata |  emd-49207.cif.gz emd-49207.cif.gz | 7.2 KB | ||
| Others |  emd_49207_additional_1.map.gz emd_49207_additional_1.map.gz emd_49207_half_map_1.map.gz emd_49207_half_map_1.map.gz emd_49207_half_map_2.map.gz emd_49207_half_map_2.map.gz | 117.9 MB 116 MB 116 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-49207 http://ftp.pdbj.org/pub/emdb/structures/EMD-49207 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-49207 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-49207 | HTTPS FTP |
-Related structure data
| Related structure data |  9nb3MC  9n9wC  9nalC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_49207.map.gz / Format: CCP4 / Size: 125 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_49207.map.gz / Format: CCP4 / Size: 125 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | structure of quaternary complex of human phosphoribosylglycinamidine synthase with thioester intermediate bound (at glutaminase site) and AMPPNP and FGAR (at synthase site). | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.652 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Additional map: Additional Map
| File | emd_49207_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Additional Map | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: Half Map B
| File | emd_49207_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half Map B | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: Half Map A
| File | emd_49207_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half Map A | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Cryo-EM structure of quaternary complex of human phosphoribosylgl...
| Entire | Name: Cryo-EM structure of quaternary complex of human phosphoribosylglycinamidine synthase with thioester intermediate bound (at glutaminase site) and AMPPNP and FGAR (at synthase site) |
|---|---|
| Components |
|
-Supramolecule #1: Cryo-EM structure of quaternary complex of human phosphoribosylgl...
| Supramolecule | Name: Cryo-EM structure of quaternary complex of human phosphoribosylglycinamidine synthase with thioester intermediate bound (at glutaminase site) and AMPPNP and FGAR (at synthase site) type: organelle_or_cellular_component / ID: 1 / Parent: 0 / Macromolecule list: #1 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: Phosphoribosylformylglycinamidine synthase
| Macromolecule | Name: Phosphoribosylformylglycinamidine synthase / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO / EC number: phosphoribosylformylglycinamidine synthase |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 148.495672 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: MSPVLHFYVR PSGHEGAAPG HTRRKLQGKL PELQGVETEL CYNVNWTAEA LPSAEETKKL MWLFGCPLLL DDVARESWLL PGSNDLLLE VGPRLNFSTP TSTNIVSVCR ATGLGPVDRV ETTRRYRLSF AHPPSAEVEA IALATLHDRM TEQHFPHPIQ S FSPESMPE ...String: MSPVLHFYVR PSGHEGAAPG HTRRKLQGKL PELQGVETEL CYNVNWTAEA LPSAEETKKL MWLFGCPLLL DDVARESWLL PGSNDLLLE VGPRLNFSTP TSTNIVSVCR ATGLGPVDRV ETTRRYRLSF AHPPSAEVEA IALATLHDRM TEQHFPHPIQ S FSPESMPE PLNGPINILG EGRLALEKAN QELGLALDSW DLDFYTKRFQ ELQRNPSTVE AFDLAQSNSE HSRHWFFKGQ LH VDGQKLV HSLFESIMST QESSNPNNVL KFCDNSSAIQ GKEVRFLRPE DPTRPSRFQQ QQGLRHVVFT AETHNFPTGV CPF SGATTG TGGRIRDVQC TGRGAHVVAG TAGYCFGNLH IPGYNLPWED PSFQYPGNFA RPLEVAIEAS NGASDYGNKF GEPV LAGFA RSLGLQLPDG QRREWIKPIM FSGGIGSMEA DHISKEAPEP GMEVVKVGGP VYRIGVGGGA ASSVQVQGDN TSDLD FGAV QRGDPEMEQK MNRVIRACVE APKGNPICSL HDQGAGGNGN VLKELSDPAG AIIYTSRFQL GDPTLNALEI WGAEYQ ESN ALLLRSPNRD FLTHVSARER CPACFVGTIT GDRRIVLVDD RECPVRRNGQ GDAPPTPLPT PVDLELEWVL GKMPRKE FF LQRKPPMLQP LALPPGLSVH QALERVLRLP AVASKRYLTN KVDRSVGGLV AQQQCVGPLQ TPLADVAVVA LSHEELIG A ATALGEQPVK SLLDPKVAAR LAVAEALTNL VFALVTDLRD VKCSGNWMWA AKLPGEGAAL ADACEAMVAV MAALGVAVD GGKDSLSMAA RVGTETVRAP GSLVISAYAV CPDITATVTP DLKHPEGRGH LLYVALSPGQ HRLGGTALAQ CFSQLGEHPP DLDLPENLV RAFSITQGLL KDRLLCSGHD VSDGGLVTCL LEMAFAGNCG LQVDVPVPRV DVLSVLFAEE PGLVLEVQEP D LAQVLKRY RDAGLHCLEL GHTGEAGPHA MVRVSVNGAV VLEEPVGELR ALWEETSFQL DRLQAEPRCV AEEERGLRER MG PSYCLPP TFPKASVPRE PGGPSPRVAI LREEGSNGDR EMADAFHLAG FEVWDVTMQD LCSGAIGLDT FRGVAFVGGF SYA DVLGSA KGWAAAVTFH PRAGAELRRF RKRPDTFSLG VCNGCQLLAL LGWVGGDPNE DAAEMGPDSQ PARPGLLLRH NLSG RYESR WASVRVGPGP ALMLRGMEGA VLPVWSAHGE GYVAFSSPEL QAQIEARGLA PLHWADDDGN PTEQYPLNPN GSPGG VAGI CSCDGRHLAV MPHPERAVRP WQWAWRPPPF DTLTTSPWLQ LFINARNWTL EGSCTRTRPL EQKLISEEDL AANDIL DYK DDDDKV UniProtKB: Phosphoribosylformylglycinamidine synthase |
-Macromolecule #2: N-(N-FORMYLGLYCYL)-5-O-PHOSPHONO-BETA-D-RIBOFURANOSYLAMINE
| Macromolecule | Name: N-(N-FORMYLGLYCYL)-5-O-PHOSPHONO-BETA-D-RIBOFURANOSYLAMINE type: ligand / ID: 2 / Number of copies: 1 / Formula: FGR |
|---|---|
| Molecular weight | Theoretical: 314.186 Da |
| Chemical component information | 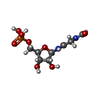 ChemComp-FGR: |
-Macromolecule #3: PHOSPHOAMINOPHOSPHONIC ACID-ADENYLATE ESTER
| Macromolecule | Name: PHOSPHOAMINOPHOSPHONIC ACID-ADENYLATE ESTER / type: ligand / ID: 3 / Number of copies: 1 / Formula: ANP |
|---|---|
| Molecular weight | Theoretical: 506.196 Da |
| Chemical component information |  ChemComp-ANP: |
-Macromolecule #4: ADENOSINE-5'-DIPHOSPHATE
| Macromolecule | Name: ADENOSINE-5'-DIPHOSPHATE / type: ligand / ID: 4 / Number of copies: 1 / Formula: ADP |
|---|---|
| Molecular weight | Theoretical: 427.201 Da |
| Chemical component information |  ChemComp-ADP: |
-Macromolecule #5: GLUTAMINE
| Macromolecule | Name: GLUTAMINE / type: ligand / ID: 5 / Number of copies: 1 / Formula: GLN |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 146.144 Da |
| Chemical component information | 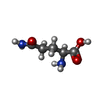 ChemComp-GLN: |
-Macromolecule #6: MAGNESIUM ION
| Macromolecule | Name: MAGNESIUM ION / type: ligand / ID: 6 / Number of copies: 3 / Formula: MG |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 24.305 Da |
-Macromolecule #7: water
| Macromolecule | Name: water / type: ligand / ID: 7 / Number of copies: 59 / Formula: HOH |
|---|---|
| Molecular weight | Theoretical: 18.015 Da |
| Chemical component information |  ChemComp-HOH: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.4 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | TFS KRIOS |
|---|---|
| Image recording | Film or detector model: FEI FALCON IV (4k x 4k) / Average electron dose: 45.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.5 µm / Nominal defocus min: 0.75 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller








 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)













































