[English] 日本語
 Yorodumi
Yorodumi- EMDB-42491: CryoEM Structure of Allosterically Switchable De Novo Protein sr3... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | CryoEM Structure of Allosterically Switchable De Novo Protein sr312, in Open State with Effector Peptide | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | de novo / allosterically switchable de novo protein / sr312 / effector peptide / DE NOVO PROTEIN | |||||||||
| Biological species | synthetic construct (others) | |||||||||
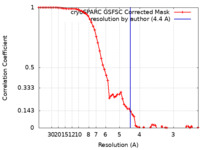
| Method | single particle reconstruction / cryo EM / Resolution: 4.4 Å | |||||||||
 Authors Authors | Weidle C / Skotheim R | |||||||||
| Funding support |  United States, 1 items United States, 1 items
| |||||||||
 Citation Citation |  Journal: Nature / Year: 2024 Journal: Nature / Year: 2024Title: De novo design of allosterically switchable protein assemblies. Authors: Arvind Pillai / Abbas Idris / Annika Philomin / Connor Weidle / Rebecca Skotheim / Philip J Y Leung / Adam Broerman / Cullen Demakis / Andrew J Borst / Florian Praetorius / David Baker /   Abstract: Allosteric modulation of protein function, wherein the binding of an effector to a protein triggers conformational changes at distant functional sites, plays a central part in the control of ...Allosteric modulation of protein function, wherein the binding of an effector to a protein triggers conformational changes at distant functional sites, plays a central part in the control of metabolism and cell signalling. There has been considerable interest in designing allosteric systems, both to gain insight into the mechanisms underlying such 'action at a distance' modulation and to create synthetic proteins whose functions can be regulated by effectors. However, emulating the subtle conformational changes distributed across many residues, characteristic of natural allosteric proteins, is a significant challenge. Here, inspired by the classic Monod-Wyman-Changeux model of cooperativity, we investigate the de novo design of allostery through rigid-body coupling of peptide-switchable hinge modules to protein interfaces that direct the formation of alternative oligomeric states. We find that this approach can be used to generate a wide variety of allosterically switchable systems, including cyclic rings that incorporate or eject subunits in response to peptide binding and dihedral cages that undergo effector-induced disassembly. Size-exclusion chromatography, mass photometry and electron microscopy reveal that these designed allosteric protein assemblies closely resemble the design models in both the presence and absence of peptide effectors and can have ligand-binding cooperativity comparable to classic natural systems such as haemoglobin. Our results indicate that allostery can arise from global coupling of the energetics of protein substructures without optimized side-chain-side-chain allosteric communication pathways and provide a roadmap for generating allosterically triggerable delivery systems, protein nanomachines and cellular feedback control circuitry. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_42491.map.gz emd_42491.map.gz | 27.3 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-42491-v30.xml emd-42491-v30.xml emd-42491.xml emd-42491.xml | 19.1 KB 19.1 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_42491_fsc.xml emd_42491_fsc.xml | 6.6 KB | Display |  FSC data file FSC data file |
| Images |  emd_42491.png emd_42491.png | 110.1 KB | ||
| Filedesc metadata |  emd-42491.cif.gz emd-42491.cif.gz | 6 KB | ||
| Others |  emd_42491_half_map_1.map.gz emd_42491_half_map_1.map.gz emd_42491_half_map_2.map.gz emd_42491_half_map_2.map.gz | 28.2 MB 28.2 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-42491 http://ftp.pdbj.org/pub/emdb/structures/EMD-42491 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-42491 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-42491 | HTTPS FTP |
-Related structure data
| Related structure data |  8ureMC  8up1C  8utmC M: atomic model generated by this map C: citing same article ( |
|---|
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_42491.map.gz / Format: CCP4 / Size: 30.5 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_42491.map.gz / Format: CCP4 / Size: 30.5 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.2375 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #2
| File | emd_42491_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #1
| File | emd_42491_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Complex is comprised of 4 sr312 proteins, each sr312 protein is b...
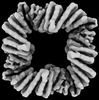
| Entire | Name: Complex is comprised of 4 sr312 proteins, each sr312 protein is bound to an effector peptide, putting the protein in an open state. Complex is formed with C4 symmetry. |
|---|---|
| Components |
|
-Supramolecule #1: Complex is comprised of 4 sr312 proteins, each sr312 protein is b...
| Supramolecule | Name: Complex is comprised of 4 sr312 proteins, each sr312 protein is bound to an effector peptide, putting the protein in an open state. Complex is formed with C4 symmetry. type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Molecular weight | Theoretical: 206.1 KDa |
-Supramolecule #2: sr312 protein
| Supramolecule | Name: sr312 protein / type: complex / ID: 2 / Parent: 1 / Macromolecule list: #1 |
|---|---|
| Source (natural) | Organism: synthetic construct (others) |
-Supramolecule #3: effector peptide
| Supramolecule | Name: effector peptide / type: complex / ID: 3 / Parent: 1 / Macromolecule list: #2 |
|---|---|
| Source (natural) | Organism: synthetic construct (others) / Synthetically produced: Yes |
-Macromolecule #1: sr312
| Macromolecule | Name: sr312 / type: protein_or_peptide / ID: 1 / Number of copies: 4 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism: synthetic construct (others) |
| Molecular weight | Theoretical: 47.297691 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: SGTVTFDITN IDWETAEWIM KHVYLIAKKE GTDVTFSFKE GELQITVKNL HEEAKREIEK WIRAAQLAQD PDAESKAEAR KILNELITE KAEELREKTK DEEVRELARE AARLALESDD IEVQRVVLKA LLAALKSKDE EVIRLLLLAA VLAAAAARSG S PEEKLEIA ...String: SGTVTFDITN IDWETAEWIM KHVYLIAKKE GTDVTFSFKE GELQITVKNL HEEAKREIEK WIRAAQLAQD PDAESKAEAR KILNELITE KAEELREKTK DEEVRELARE AARLALESDD IEVQRVVLKA LLAALKSKDE EVIRLLLLAA VLAAAAARSG S PEEKLEIA KKALELAMKS KDEEVIRLAL LAAVLAARSD DEEVLKKVKE ALEKMERIMD LEDVAREKSG SAEASQAVKE IA DIAEEAL REGLCEVARV ALKRLFKLAK DYPGSDVASL AKKALEKIAE TALRNGCKET AELAKLLLFL LLIIEVVLKM GVR MLTHRG GNAVIVVIEG LHPSQIVQLM QDVIKAAKKL GVTVTITVSG DIVVIMVVVG ASDEEQEEAR RLVQEIARAL QEAK RKGAN EEQLEQLLRE LLERAEREGG SG |
-Macromolecule #2: Effector peptide cs221B
| Macromolecule | Name: Effector peptide cs221B / type: protein_or_peptide / ID: 2 / Number of copies: 4 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism: synthetic construct (others) |
| Molecular weight | Theoretical: 3.091635 KDa |
| Sequence | String: EERKKELAKE VIETAKKLIE KLAKEE |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 1.0 mg/mL | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 8 Component:
Details: 150 mM NaCl, 40 mM Tris, pH 8.0 | |||||||||
| Grid | Model: C-flat-2/2 / Material: COPPER / Mesh: 200 / Support film - Material: CARBON / Support film - topology: HOLEY / Support film - Film thickness: 40 / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 25 sec. / Pretreatment - Atmosphere: AIR / Pretreatment - Pressure: 39.0 kPa | |||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 295.15 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Number real images: 3795 / Average electron dose: 43.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 50.0 µm / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 1.8 µm / Nominal defocus min: 0.8 µm |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller




 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)