[English] 日本語
 Yorodumi
Yorodumi- EMDB-42026: Human retinal variant phosphomimetic IMPDH1(546)-S477D filament b... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Human retinal variant phosphomimetic IMPDH1(546)-S477D filament bound by ATP, IMP, and NAD+, octamer-centered | ||||||||||||||||||
 Map data Map data | Sharpened map from deepEMhancer | ||||||||||||||||||
 Sample Sample |
| ||||||||||||||||||
 Keywords Keywords | dehydrogenase / nucleotide synthesis / filament / phosphomimetic / OXIDOREDUCTASE | ||||||||||||||||||
| Function / homology |  Function and homology information Function and homology informationlymphocyte proliferation / 'de novo' XMP biosynthetic process / Purine ribonucleoside monophosphate biosynthesis / IMP dehydrogenase / IMP dehydrogenase activity / GMP biosynthetic process / Azathioprine ADME / GTP biosynthetic process / azurophil granule lumen / secretory granule lumen ...lymphocyte proliferation / 'de novo' XMP biosynthetic process / Purine ribonucleoside monophosphate biosynthesis / IMP dehydrogenase / IMP dehydrogenase activity / GMP biosynthetic process / Azathioprine ADME / GTP biosynthetic process / azurophil granule lumen / secretory granule lumen / Potential therapeutics for SARS / nucleic acid binding / ficolin-1-rich granule lumen / nucleotide binding / Neutrophil degranulation / DNA binding / RNA binding / extracellular region / metal ion binding / identical protein binding / nucleus / cytoplasm / cytosol Similarity search - Function | ||||||||||||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||||||||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 2.4 Å | ||||||||||||||||||
 Authors Authors | Calise SJ / Kollman JM | ||||||||||||||||||
| Funding support |  United States, 5 items United States, 5 items
| ||||||||||||||||||
 Citation Citation |  Journal: J Cell Biol / Year: 2024 Journal: J Cell Biol / Year: 2024Title: Light-sensitive phosphorylation regulates retinal IMPDH1 activity and filament assembly. Authors: S John Calise / Audrey G O'Neill / Anika L Burrell / Miles S Dickinson / Josephine Molfino / Charlie Clarke / Joel Quispe / David Sokolov / Rubén M Buey / Justin M Kollman /   Abstract: Inosine monophosphate dehydrogenase (IMPDH) is the rate-limiting enzyme in guanosine triphosphate (GTP) synthesis and assembles into filaments in cells, which desensitizes the enzyme to feedback ...Inosine monophosphate dehydrogenase (IMPDH) is the rate-limiting enzyme in guanosine triphosphate (GTP) synthesis and assembles into filaments in cells, which desensitizes the enzyme to feedback inhibition and boosts nucleotide production. The vertebrate retina expresses two splice variants IMPDH1(546) and IMPDH1(595). In bovine retinas, residue S477 is preferentially phosphorylated in the dark, but the effects on IMPDH1 activity and regulation are unclear. Here, we generated phosphomimetic mutants to investigate structural and functional consequences of S477 phosphorylation. The S477D mutation resensitized both variants to GTP inhibition but only blocked assembly of IMPDH1(595) filaments. Cryo-EM structures of both variants showed that S477D specifically blocks assembly of a high-activity assembly interface, still allowing assembly of low-activity IMPDH1(546) filaments. Finally, we discovered that S477D exerts a dominant-negative effect in cells, preventing endogenous IMPDH filament assembly. By modulating the structure and higher-order assembly of IMPDH, S477 phosphorylation acts as a mechanism for downregulating retinal GTP synthesis in the dark when nucleotide turnover is decreased. | ||||||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_42026.map.gz emd_42026.map.gz | 222.3 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-42026-v30.xml emd-42026-v30.xml emd-42026.xml emd-42026.xml | 20.3 KB 20.3 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_42026.png emd_42026.png | 115.4 KB | ||
| Filedesc metadata |  emd-42026.cif.gz emd-42026.cif.gz | 6.6 KB | ||
| Others |  emd_42026_additional_1.map.gz emd_42026_additional_1.map.gz emd_42026_half_map_1.map.gz emd_42026_half_map_1.map.gz emd_42026_half_map_2.map.gz emd_42026_half_map_2.map.gz | 229.9 MB 225.2 MB 225.2 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-42026 http://ftp.pdbj.org/pub/emdb/structures/EMD-42026 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-42026 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-42026 | HTTPS FTP |
-Related structure data
| Related structure data |  8u8oMC  8u7mC  8u7qC  8u7vC  8u8yC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_42026.map.gz / Format: CCP4 / Size: 244.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_42026.map.gz / Format: CCP4 / Size: 244.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Sharpened map from deepEMhancer | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.843 Å | ||||||||||||||||||||||||||||||||||||
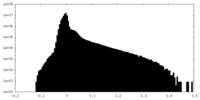
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Additional map: Sharpened map from CryoSPARC
| File | emd_42026_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Sharpened map from CryoSPARC | ||||||||||||
| Projections & Slices |
| ||||||||||||
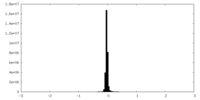
| Density Histograms |
-Half map: Half map 1
| File | emd_42026_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half map 1 | ||||||||||||
| Projections & Slices |
| ||||||||||||
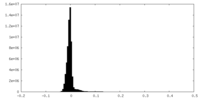
| Density Histograms |
-Half map: Half map 2
| File | emd_42026_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half map 2 | ||||||||||||
| Projections & Slices |
| ||||||||||||
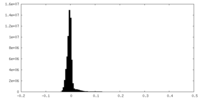
| Density Histograms |
- Sample components
Sample components
-Entire : IMPDH1(546)-S477D filament bound by ATP, IMP, and NAD+
| Entire | Name: IMPDH1(546)-S477D filament bound by ATP, IMP, and NAD+ |
|---|---|
| Components |
|
-Supramolecule #1: IMPDH1(546)-S477D filament bound by ATP, IMP, and NAD+
| Supramolecule | Name: IMPDH1(546)-S477D filament bound by ATP, IMP, and NAD+ type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 468 KDa |
-Macromolecule #1: Inosine-5'-monophosphate dehydrogenase 1
| Macromolecule | Name: Inosine-5'-monophosphate dehydrogenase 1 / type: protein_or_peptide / ID: 1 / Number of copies: 8 / Enantiomer: LEVO / EC number: IMP dehydrogenase |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 58.542906 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: GSMADYLISG GTGYVPEDGL TAQQLFASAD GLTYNDFLIL PGFIDFIADE VDLTSALTRK ITLKTPLISS PMDTVTEADM AIAMALMGG IGFIHHNCTP EFQANEVRKV KKFEQGFITD PVVLSPSHTV GDVLEAKMRH GFSGIPITET GTMGSKLVGI V TSRDIDFL ...String: GSMADYLISG GTGYVPEDGL TAQQLFASAD GLTYNDFLIL PGFIDFIADE VDLTSALTRK ITLKTPLISS PMDTVTEADM AIAMALMGG IGFIHHNCTP EFQANEVRKV KKFEQGFITD PVVLSPSHTV GDVLEAKMRH GFSGIPITET GTMGSKLVGI V TSRDIDFL AEKDHTTLLS EVMTPRIELV VAPAGVTLKE ANEILQRSKK GKLPIVNDCD ELVAIIARTD LKKNRDYPLA SK DSQKQLL CGAAVGTRED DKYRLDLLTQ AGVDVIVLDS SQGNSVYQIA MVHYIKQKYP HLQVIGGNVV TAAQAKNLID AGV DGLRVG MGCGSICITQ EVMACGRPQG TAVYKVAEYA RRFGVPIIAD GGIQTVGHVV KALALGASTV MMGSLLAATT EAPG EYFFS DGVRLKKYRG MGSLDAMEKS SSSQKRYFSE GDKVKIAQGV SGSIQDKGSI QKFVPYLIAG IQHGCQDIGA RSLDV LRSM MYSGELKFEK RTMSAQIEGG VHGLHSYTFL PFTKSGCTED SGGGRGGGGD APQCPLLGTA SLHN UniProtKB: Inosine-5'-monophosphate dehydrogenase 1 |
-Macromolecule #2: ADENOSINE-5'-TRIPHOSPHATE
| Macromolecule | Name: ADENOSINE-5'-TRIPHOSPHATE / type: ligand / ID: 2 / Number of copies: 16 / Formula: ATP |
|---|---|
| Molecular weight | Theoretical: 507.181 Da |
| Chemical component information |  ChemComp-ATP: |
-Macromolecule #3: INOSINIC ACID
| Macromolecule | Name: INOSINIC ACID / type: ligand / ID: 3 / Number of copies: 8 / Formula: IMP |
|---|---|
| Molecular weight | Theoretical: 348.206 Da |
| Chemical component information |  ChemComp-I: |
-Macromolecule #4: NICOTINAMIDE-ADENINE-DINUCLEOTIDE
| Macromolecule | Name: NICOTINAMIDE-ADENINE-DINUCLEOTIDE / type: ligand / ID: 4 / Number of copies: 8 / Formula: NAD |
|---|---|
| Molecular weight | Theoretical: 663.425 Da |
| Chemical component information |  ChemComp-NAD: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | filament |
- Sample preparation
Sample preparation
| Buffer | pH: 7 |
|---|---|
| Grid | Model: C-flat-2/2 / Material: COPPER / Pretreatment - Type: GLOW DISCHARGE |
| Vitrification | Cryogen name: ETHANE / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | TFS KRIOS |
|---|---|
| Specialist optics | Energy filter - Name: GIF Bioquantum / Energy filter - Slit width: 20 eV |
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Detector mode: COUNTING / Number real images: 4632 / Average electron dose: 50.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 1.8 µm / Nominal defocus min: 1.2 µm / Nominal magnification: 105000 |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller








 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)