[English] 日本語
 Yorodumi
Yorodumi- EMDB-41771: S. thermodepolymerans KpsMT-KpsE with bound glycolipid - state 2 ... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | S. thermodepolymerans KpsMT-KpsE with bound glycolipid - state 2 - consensus map | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | ABC transporter / Capsular polysaccharide / MEMBRANE PROTEIN | |||||||||
| Biological species |  Caldimonas thermodepolymerans (bacteria) Caldimonas thermodepolymerans (bacteria) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.4 Å | |||||||||
 Authors Authors | Kuklewicz J / Zimmer J | |||||||||
| Funding support |  United States, 1 items United States, 1 items
| |||||||||
 Citation Citation |  Journal: Nature / Year: 2024 Journal: Nature / Year: 2024Title: Molecular insights into capsular polysaccharide secretion. Authors: Jeremi Kuklewicz / Jochen Zimmer /  Abstract: Capsular polysaccharides (CPSs) fortify the cell boundaries of many commensal and pathogenic bacteria. Through the ABC-transporter-dependent biosynthesis pathway, CPSs are synthesized intracellularly ...Capsular polysaccharides (CPSs) fortify the cell boundaries of many commensal and pathogenic bacteria. Through the ABC-transporter-dependent biosynthesis pathway, CPSs are synthesized intracellularly on a lipid anchor and secreted across the cell envelope by the KpsMT ABC transporter associated with the KpsE and KpsD subunits. Here we use structural and functional studies to uncover crucial steps of CPS secretion in Gram-negative bacteria. We show that KpsMT has broad substrate specificity and is sufficient for the translocation of CPSs across the inner bacterial membrane, and we determine the cell surface organization and localization of CPSs using super-resolution fluorescence microscopy. Cryo-electron microscopy analyses of the KpsMT-KpsE complex in six different states reveal a KpsE-encaged ABC transporter, rigid-body conformational rearrangements of KpsMT during ATP hydrolysis and recognition of a glycolipid inside a membrane-exposed electropositive canyon. In vivo CPS secretion assays underscore the functional importance of canyon-lining basic residues. Combined, our analyses suggest a molecular model of CPS secretion by ABC transporters. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_41771.map.gz emd_41771.map.gz | 168.3 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-41771-v30.xml emd-41771-v30.xml emd-41771.xml emd-41771.xml | 12.5 KB 12.5 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_41771_fsc.xml emd_41771_fsc.xml | 11.9 KB | Display |  FSC data file FSC data file |
| Images |  emd_41771.png emd_41771.png | 32.2 KB | ||
| Filedesc metadata |  emd-41771.cif.gz emd-41771.cif.gz | 3.8 KB | ||
| Others |  emd_41771_half_map_1.map.gz emd_41771_half_map_1.map.gz emd_41771_half_map_2.map.gz emd_41771_half_map_2.map.gz | 165.4 MB 165.4 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-41771 http://ftp.pdbj.org/pub/emdb/structures/EMD-41771 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-41771 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-41771 | HTTPS FTP |
-Validation report
| Summary document |  emd_41771_validation.pdf.gz emd_41771_validation.pdf.gz | 935.2 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_41771_full_validation.pdf.gz emd_41771_full_validation.pdf.gz | 934.8 KB | Display | |
| Data in XML |  emd_41771_validation.xml.gz emd_41771_validation.xml.gz | 20.5 KB | Display | |
| Data in CIF |  emd_41771_validation.cif.gz emd_41771_validation.cif.gz | 26.6 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-41771 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-41771 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-41771 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-41771 | HTTPS FTP |
-Related structure data
| Related structure data |  8tshC  8tsiC  8tslC  8tswC  8tt3C  8tunC  41603 C: citing same article ( |
|---|
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_41771.map.gz / Format: CCP4 / Size: 178 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_41771.map.gz / Format: CCP4 / Size: 178 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.08 Å | ||||||||||||||||||||||||||||||||||||
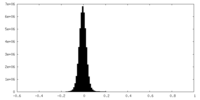
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #1
| File | emd_41771_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
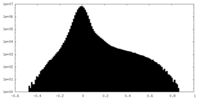
| Density Histograms |
-Half map: #2
| File | emd_41771_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
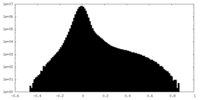
| Density Histograms |
- Sample components
Sample components
-Entire : ABC transporter KpsMT in complex with polysaccharide co-polymeras...
| Entire | Name: ABC transporter KpsMT in complex with polysaccharide co-polymerase KpsE bound to the glycolipid in state 2 |
|---|---|
| Components |
|
-Supramolecule #1: ABC transporter KpsMT in complex with polysaccharide co-polymeras...
| Supramolecule | Name: ABC transporter KpsMT in complex with polysaccharide co-polymerase KpsE bound to the glycolipid in state 2 type: complex / ID: 1 / Parent: 0 / Macromolecule list: #2-#3, #1 |
|---|---|
| Source (natural) | Organism:  Caldimonas thermodepolymerans (bacteria) Caldimonas thermodepolymerans (bacteria) |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: FEI FALCON III (4k x 4k) / Average electron dose: 50.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 1.8 µm / Nominal defocus min: 1.0 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller




















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)