+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of mink variant Y453F trimeric spike protein | |||||||||
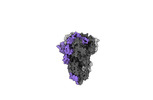
 Map data Map data | cryo-EM map of the global map of mink variant SARS-CoV-2 trimeric spike with no ACE2 bound. | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Coronavirus / Spike / Mink / SARS-CoV-2 / PROTEIN BINDING / VIRAL PROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationpositive regulation of viral entry into host cell / host cell endoplasmic reticulum-Golgi intermediate compartment membrane / receptor-mediated virion attachment to host cell / host cell surface receptor binding / endocytosis involved in viral entry into host cell / fusion of virus membrane with host plasma membrane / fusion of virus membrane with host endosome membrane / viral envelope / host cell plasma membrane / virion membrane ...positive regulation of viral entry into host cell / host cell endoplasmic reticulum-Golgi intermediate compartment membrane / receptor-mediated virion attachment to host cell / host cell surface receptor binding / endocytosis involved in viral entry into host cell / fusion of virus membrane with host plasma membrane / fusion of virus membrane with host endosome membrane / viral envelope / host cell plasma membrane / virion membrane / membrane / identical protein binding Similarity search - Function | |||||||||
| Biological species |  | |||||||||
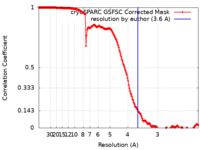
| Method | single particle reconstruction / cryo EM / Resolution: 3.6 Å | |||||||||
 Authors Authors | Ahn HM / Calderon B / Fan X / Gao Y / Horgan N / Zhou B / Liang B | |||||||||
| Funding support |  United States, 1 items United States, 1 items
| |||||||||
 Citation Citation |  Journal: J Med Virol / Year: 2023 Journal: J Med Virol / Year: 2023Title: Structural basis of the American mink ACE2 binding by Y453F trimeric spike glycoproteins of SARS-CoV-2. Authors: Hyunjun Ahn / Brenda M Calderon / Xiaoyu Fan / Yunrong Gao / Natalie L Horgan / Nannan Jiang / Dylan S Blohm / Jaber Hossain / Nicole Wedad K Rayyan / Sarah H Osman / Xudong Lin / Michael ...Authors: Hyunjun Ahn / Brenda M Calderon / Xiaoyu Fan / Yunrong Gao / Natalie L Horgan / Nannan Jiang / Dylan S Blohm / Jaber Hossain / Nicole Wedad K Rayyan / Sarah H Osman / Xudong Lin / Michael Currier / John Steel / David E Wentworth / Bin Zhou / Bo Liang /  Abstract: Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) enters the host cell by binding to angiotensin-converting enzyme 2 (ACE2). While evolutionarily conserved, ACE2 receptors differ across ...Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) enters the host cell by binding to angiotensin-converting enzyme 2 (ACE2). While evolutionarily conserved, ACE2 receptors differ across various species and differential interactions with Spike (S) glycoproteins of SARS-CoV-2 viruses impact species specificity. Reverse zoonoses led to SARS-CoV-2 outbreaks on multiple American mink (Mustela vison) farms during the pandemic and gave rise to mink-associated S substitutions known for transmissibility between mink and zoonotic transmission to humans. In this study, we used bio-layer interferometry (BLI) to discern the differences in binding affinity between multiple human and mink-derived S glycoproteins of SARS-CoV-2 and their respective ACE2 receptors. Further, we conducted a structural analysis of a mink variant S glycoprotein and American mink ACE2 (mvACE2) using cryo-electron microscopy (cryo-EM), revealing four distinct conformations. We discovered a novel intermediary conformation where the mvACE2 receptor is bound to the receptor-binding domain (RBD) of the S glycoprotein in a "down" position, approximately 34° lower than previously reported "up" RBD. Finally, we compared residue interactions in the S-ACE2 complex interface of S glycoprotein conformations with varying RBD orientations. These findings provide valuable insights into the molecular mechanisms of SARS-CoV-2 entry. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_40977.map.gz emd_40977.map.gz | 483.6 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-40977-v30.xml emd-40977-v30.xml emd-40977.xml emd-40977.xml | 17 KB 17 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_40977_fsc.xml emd_40977_fsc.xml | 16.9 KB | Display |  FSC data file FSC data file |
| Images |  emd_40977.png emd_40977.png | 24.4 KB | ||
| Filedesc metadata |  emd-40977.cif.gz emd-40977.cif.gz | 6.3 KB | ||
| Others |  emd_40977_half_map_1.map.gz emd_40977_half_map_1.map.gz emd_40977_half_map_2.map.gz emd_40977_half_map_2.map.gz | 475.8 MB 475.8 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-40977 http://ftp.pdbj.org/pub/emdb/structures/EMD-40977 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-40977 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-40977 | HTTPS FTP |
-Related structure data
| Related structure data |  8t21MC  8t20C  8t22C  8t23C  8t25C  8tazC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_40977.map.gz / Format: CCP4 / Size: 512 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_40977.map.gz / Format: CCP4 / Size: 512 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | cryo-EM map of the global map of mink variant SARS-CoV-2 trimeric spike with no ACE2 bound. | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.11 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: Half map of the global map of mink...
| File | emd_40977_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half map of the global map of mink variant SARS-CoV-2 trimeric spike with no ACE2 bound. | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: Half map of the global map of mink...
| File | emd_40977_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half map of the global map of mink variant SARS-CoV-2 trimeric spike with no ACE2 bound. | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Cryo-EM structure of mink variant Y453F trimeric spike protein
| Entire | Name: Cryo-EM structure of mink variant Y453F trimeric spike protein |
|---|---|
| Components |
|
-Supramolecule #1: Cryo-EM structure of mink variant Y453F trimeric spike protein
| Supramolecule | Name: Cryo-EM structure of mink variant Y453F trimeric spike protein type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  |
-Macromolecule #1: Spike glycoprotein
| Macromolecule | Name: Spike glycoprotein / type: protein_or_peptide / ID: 1 / Number of copies: 3 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 140.244109 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: MFVFLVLLPL VSSQCVNLTT RTQLPPAYTN SFTRGVYYPD KVFRSSVLHS TQDLFLPFFS NVTWFHAISG TNGTKRFDNP VLPFNDGVY FASTEKSNII RGWIFGTTLD SKTQSLLIVN NATNVVIKVC EFQFCNDPFL GVYYHKNNKS WMESEFRVYS S ANNCTFEY ...String: MFVFLVLLPL VSSQCVNLTT RTQLPPAYTN SFTRGVYYPD KVFRSSVLHS TQDLFLPFFS NVTWFHAISG TNGTKRFDNP VLPFNDGVY FASTEKSNII RGWIFGTTLD SKTQSLLIVN NATNVVIKVC EFQFCNDPFL GVYYHKNNKS WMESEFRVYS S ANNCTFEY VSQPFLMDLE GKQGNFKNLR EFVFKNIDGY FKIYSKHTPI NLVRDLPQGF SALEPLVDLP IGINITRFQT LL ALHRSYL TPGDSSSGWT AGAAAYYVGY LQPRTFLLKY NENGTITDAV DCALDPLSET KCTLKSFTVE KGIYQTSNFR VQP TESIVR FPNITNLCPF GEVFNATRFA SVYAWNRKRI SNCVADYSVL YNSASFSTFK CYGVSPTKLN DLCFTNVYAD SFVI RGDEV RQIAPGQTGK IADYNYKLPD DFTGCVIAWN SNNLDSKVGG NYNYLFRLFR KSNLKPFERD ISTEIYQAGS TPCNG VEGF NCYFPLQSYG FQPTNGVGYQ PYRVVVLSFE LLHAPATVCG PKKSTNLVKN KCVNFNFNGL TGTGVLTESN KKFLPF QQF GRDIADTTDA VRDPQTLEIL DITPCSFGGV SVITPGTNTS NQVAVLYQGV NCTEVPVAIH ADQLTPTWRV YSTGSNV FQ TRAGCLIGAE HVNNSYECDI PIGAGICASY QTQTNSPGSA SSVASQSIIA YTMSLGAENS VAYSNNSIAI PTNFTISV T TEILPVSMTK TSVDCTMYIC GDSTECSNLL LQYGSFCTQL NRALTGIAVE QDKNTQEVFA QVKQIYKTPP IKDFGGFNF SQILPDPSKP SKRSPIEDLL FNKVTLADAG FIKQYGDCLG DIAARDLICA QKFNGLTVLP PLLTDEMIAQ YTSALLAGTI TSGWTFGAG PALQIPFPMQ MAYRFNGIGV TQNVLYENQK LIANQFNSAI GKIQDSLSST PSALGKLQDV VNQNAQALNT L VKQLSSNF GAISSVLNDI LSRLDPPEAE VQIDRLITGR LQSLQTYVTQ QLIRAAEIRA SANLAATKMS ECVLGQSKRV DF CGKGYHL MSFPQSAPHG VVFLHVTYVP AQEKNFTTAP AICHDGKAHF PREGVFVSNG THWFVTQRNF YEPQIITTDN TFV SGNCDV VIGIVNNTVY DPLQPELDSF KEELDKYFKN HTSPDVDLGD ISGINASVVN IQKEIDRLNE VAKNLNESLI DLQE LGKYE QGSGSGSGSG YIPEAPRDGQ AYVRKDGEWV LLSTFLGSGS GSGHHHHHHG LNDIFEAQKI EWHE UniProtKB: Spike glycoprotein |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 1.5 mg/mL | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 8 Component:
| |||||||||
| Grid | Material: COPPER / Support film - Material: CARBON / Support film - topology: HOLEY / Pretreatment - Type: GLOW DISCHARGE | |||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | TFS KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Number real images: 11392 / Average exposure time: 4.0 sec. / Average electron dose: 49.98 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 100.0 µm / Illumination mode: OTHER / Imaging mode: OTHER / Nominal defocus max: 1.75 µm / Nominal defocus min: 0.75 µm / Nominal magnification: 81000 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Refinement | Space: REAL |
|---|---|
| Output model |  PDB-8t21: |
 Movie
Movie Controller
Controller








 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)