+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Structure of a bacterial gasdermin slinky-like oligomer | |||||||||
 Map data Map data | EM map of Vitiosangium bGSDM slinky oligomer | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | viral mimicry / gasdermin / caspase / autoinhibition / pyroptosis / bats / immunity / cell death / IMMUNE SYSTEM | |||||||||
| Function / homology | : / Bacterial Gasdermin Family / defense response to virus / plasma membrane / cytoplasm / Gasdermin bGSDM Function and homology information Function and homology information | |||||||||
| Biological species |  Vitiosangium sp. GDMCC 1.1324 (bacteria) Vitiosangium sp. GDMCC 1.1324 (bacteria) | |||||||||
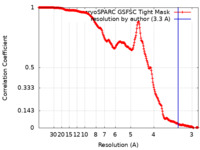
| Method | single particle reconstruction / cryo EM / Resolution: 3.3 Å | |||||||||
 Authors Authors | Johnson AG / Mayer ML / Kranzusch PJ | |||||||||
| Funding support |  United States, 1 items United States, 1 items
| |||||||||
 Citation Citation | Journal: bioRxiv / Year: 2023 Title: Structure and assembly of a bacterial gasdermin pore. Authors: Alex G Johnson / Megan L Mayer / Stefan L Schaefer / Nora K McNamara-Bordewick / Gerhard Hummer / Philip J Kranzusch /   Abstract: In response to pathogen infection, gasdermin (GSDM) proteins form membrane pores that induce a host cell death process called pyroptosis. Studies of human and mouse GSDM pores reveal the functions ...In response to pathogen infection, gasdermin (GSDM) proteins form membrane pores that induce a host cell death process called pyroptosis. Studies of human and mouse GSDM pores reveal the functions and architectures of 24-33 protomers assemblies, but the mechanism and evolutionary origin of membrane targeting and GSDM pore formation remain unknown. Here we determine a structure of a bacterial GSDM (bGSDM) pore and define a conserved mechanism of pore assembly. Engineering a panel of bGSDMs for site-specific proteolytic activation, we demonstrate that diverse bGSDMs form distinct pore sizes that range from smaller mammalian-like assemblies to exceptionally large pores containing >50 protomers. We determine a 3.3 Å cryo-EM structure of a bGSDM in an active slinky-like oligomeric conformation and analyze bGSDM pores in a native lipid environment to create an atomic-level model of a full 52-mer bGSDM pore. Combining our structural analysis with molecular dynamics simulations and cellular assays, our results support a stepwise model of GSDM pore assembly and suggest that a covalently bound palmitoyl can leave a hydrophobic sheath and insert into the membrane before formation of the membrane-spanning β-strand regions. These results reveal the diversity of GSDM pores found in nature and explain the function of an ancient post-translational modification in enabling programmed host cell death. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_40570.map.gz emd_40570.map.gz | 881.4 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-40570-v30.xml emd-40570-v30.xml emd-40570.xml emd-40570.xml | 20.8 KB 20.8 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_40570_fsc.xml emd_40570_fsc.xml | 21.1 KB | Display |  FSC data file FSC data file |
| Images |  emd_40570.png emd_40570.png | 46 KB | ||
| Filedesc metadata |  emd-40570.cif.gz emd-40570.cif.gz | 6.4 KB | ||
| Others |  emd_40570_half_map_1.map.gz emd_40570_half_map_1.map.gz emd_40570_half_map_2.map.gz emd_40570_half_map_2.map.gz | 929.2 MB 929.2 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-40570 http://ftp.pdbj.org/pub/emdb/structures/EMD-40570 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-40570 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-40570 | HTTPS FTP |
-Related structure data
| Related structure data |  8sl0MC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_40570.map.gz / Format: CCP4 / Size: 1000 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_40570.map.gz / Format: CCP4 / Size: 1000 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | EM map of Vitiosangium bGSDM slinky oligomer | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.431 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: EM half map A of Vitiosangium bGSDM slinky oligomer
| File | emd_40570_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | EM half map A of Vitiosangium bGSDM slinky oligomer | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: EM half map B of Vitiosangium bGSDM slinky oligomer
| File | emd_40570_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | EM half map B of Vitiosangium bGSDM slinky oligomer | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Vitiosangium bGSDM in an active slinky-like oligomeric conformation
| Entire | Name: Vitiosangium bGSDM in an active slinky-like oligomeric conformation |
|---|---|
| Components |
|
-Supramolecule #1: Vitiosangium bGSDM in an active slinky-like oligomeric conformation
| Supramolecule | Name: Vitiosangium bGSDM in an active slinky-like oligomeric conformation type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  Vitiosangium sp. GDMCC 1.1324 (bacteria) Vitiosangium sp. GDMCC 1.1324 (bacteria) |
-Macromolecule #1: Gasdermin bGSDM
| Macromolecule | Name: Gasdermin bGSDM / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Vitiosangium sp. GDMCC 1.1324 (bacteria) Vitiosangium sp. GDMCC 1.1324 (bacteria) |
| Molecular weight | Theoretical: 25.566246 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: SGLCSDPAIT YLKRLGYNVV RLPREGIQPL HLLGQQRGTV EYLGSLEKLI TQPPSEPPAI TRDQAAAGIN GQKTENLSFS IGINILKSV LAQFGAGAGI EAQYNQARKV RFEFSNVLAD SVEPLAVGQF LKMAEVDADN PVLKQYVLGN GRLYVITQVI K SNEFTVAA ...String: SGLCSDPAIT YLKRLGYNVV RLPREGIQPL HLLGQQRGTV EYLGSLEKLI TQPPSEPPAI TRDQAAAGIN GQKTENLSFS IGINILKSV LAQFGAGAGI EAQYNQARKV RFEFSNVLAD SVEPLAVGQF LKMAEVDADN PVLKQYVLGN GRLYVITQVI K SNEFTVAA EKSGGGSIQL DVPEIQKVVG GKLKVEASVS SQSTVTYKGE KQLVFGFKCF EIGVKNGEIT LFASQLVPR UniProtKB: Gasdermin bGSDM |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | filament |
- Sample preparation
Sample preparation
| Concentration | 1 mg/mL | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 7.5 Component:
Details: 150 mM NaCl, 20 mM HEPES-HOH (pH 7.5), 5.15 mM DDMAB | ||||||||||||
| Grid | Model: Quantifoil / Material: COPPER / Mesh: 200 / Support film - Material: CARBON / Support film - topology: HOLEY / Support film - Film thickness: 2 / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 30 sec. / Pretreatment - Atmosphere: AIR / Pretreatment - Pressure: 27.0 kPa | ||||||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 277.15 K / Instrument: FEI VITROBOT MARK IV | ||||||||||||
| Details | The sample was monodisperse |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Number real images: 8156 / Average electron dose: 51.8 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.0 µm / Nominal defocus min: 0.7000000000000001 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller









 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)