[English] 日本語
 Yorodumi
Yorodumi- EMDB-40050: Cryo-electron microscopy structure of the zinc transporter from B... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
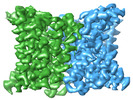
| Title | Cryo-electron microscopy structure of the zinc transporter from Bordetella bronchiseptica | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | zinc transporter / dimer / complex / metal binding / inward-open inhibited conformation / TRANSPORT PROTEIN | |||||||||
| Function / homology | Zinc/iron permease / ZIP Zinc transporter / zinc ion transmembrane transporter activity / plasma membrane / Zinc transporter ZIPB Function and homology information Function and homology information | |||||||||
| Biological species |  Bordetella bronchiseptica (bacteria) Bordetella bronchiseptica (bacteria) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.05 Å | |||||||||
 Authors Authors | Liu Q / Chai J / Pang CX / Shanklin J | |||||||||
| Funding support |  United States, 1 items United States, 1 items
| |||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2023 Journal: Nat Commun / Year: 2023Title: Structural mechanism of intracellular autoregulation of zinc uptake in ZIP transporters. Authors: Changxu Pang / Jin Chai / Ping Zhu / John Shanklin / Qun Liu /  Abstract: Zinc is an essential micronutrient that supports all living organisms through regulating numerous biological processes. However, the mechanism of uptake regulation by intracellular Zn status remains ...Zinc is an essential micronutrient that supports all living organisms through regulating numerous biological processes. However, the mechanism of uptake regulation by intracellular Zn status remains unclear. Here we report a cryo-electron microscopy structure of a ZIP-family transporter from Bordetella bronchiseptica at 3.05 Å resolution in an inward-facing, inhibited conformation. The transporter forms a homodimer, each protomer containing nine transmembrane helices and three metal ions. Two metal ions form a binuclear pore structure, and the third ion is located at an egress site facing the cytoplasm. The egress site is covered by a loop, and two histidine residues on the loop interact with the egress-site ion and regulate its release. Cell-based Zn uptake and cell growth viability assays reveal a negative regulation of Zn uptake through sensing intracellular Zn status using a built-in sensor. These structural and biochemical analyses provide mechanistic insight into the autoregulation of zinc uptake across membranes. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_40050.map.gz emd_40050.map.gz | 4 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-40050-v30.xml emd-40050-v30.xml emd-40050.xml emd-40050.xml | 14.2 KB 14.2 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_40050_fsc.xml emd_40050_fsc.xml | 4.4 KB | Display |  FSC data file FSC data file |
| Images |  emd_40050.png emd_40050.png | 164.1 KB | ||
| Masks |  emd_40050_msk_1.map emd_40050_msk_1.map | 8 MB |  Mask map Mask map | |
| Filedesc metadata |  emd-40050.cif.gz emd-40050.cif.gz | 5.3 KB | ||
| Others |  emd_40050_half_map_1.map.gz emd_40050_half_map_1.map.gz emd_40050_half_map_2.map.gz emd_40050_half_map_2.map.gz | 7.4 MB 7.4 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-40050 http://ftp.pdbj.org/pub/emdb/structures/EMD-40050 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-40050 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-40050 | HTTPS FTP |
-Related structure data
| Related structure data |  8ghtMC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_40050.map.gz / Format: CCP4 / Size: 8 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_40050.map.gz / Format: CCP4 / Size: 8 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.332 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Mask #1
| File |  emd_40050_msk_1.map emd_40050_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #1
| File | emd_40050_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #2
| File | emd_40050_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Zinc transporter dimer from Bordetella bronchiseptica
| Entire | Name: Zinc transporter dimer from Bordetella bronchiseptica |
|---|---|
| Components |
|
-Supramolecule #1: Zinc transporter dimer from Bordetella bronchiseptica
| Supramolecule | Name: Zinc transporter dimer from Bordetella bronchiseptica / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1 |
|---|---|
| Source (natural) | Organism:  Bordetella bronchiseptica (bacteria) Bordetella bronchiseptica (bacteria) |
-Macromolecule #1: Putative membrane protein
| Macromolecule | Name: Putative membrane protein / type: protein_or_peptide / ID: 1 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Bordetella bronchiseptica (bacteria) Bordetella bronchiseptica (bacteria) |
| Molecular weight | Theoretical: 31.291566 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: GSHMNQPSSL AADLRGAWHA QAQSHPLITL GLAASAAGVV LLLVAGIVNA LTGENRVHVG YAVLGGAAGF AATALGALMA LGLRAISAR TQDAMLGFAA GMMLAASAFS LILPGLDAAG TIVGPGPAAA AVVALGLGLG VLLMLGLDYF TPHEHERTGH Q GPEAARVN ...String: GSHMNQPSSL AADLRGAWHA QAQSHPLITL GLAASAAGVV LLLVAGIVNA LTGENRVHVG YAVLGGAAGF AATALGALMA LGLRAISAR TQDAMLGFAA GMMLAASAFS LILPGLDAAG TIVGPGPAAA AVVALGLGLG VLLMLGLDYF TPHEHERTGH Q GPEAARVN RVWLFVLTII LHNLPEGMAI GVSFATGDLR IGLPLTSAIA IQDVPEGLAV ALALRAVGLP IGRAVLVAVA SG LMEPLGA LVGVGISSGF ALAYPISMGL AAGAMIFVVS HEVIPETHRN GHETTATVGL MAGFALMMFL DTALG UniProtKB: Zinc transporter ZIPB |
-Macromolecule #2: CADMIUM ION
| Macromolecule | Name: CADMIUM ION / type: ligand / ID: 2 / Number of copies: 6 / Formula: CD |
|---|---|
| Molecular weight | Theoretical: 112.411 Da |
-Macromolecule #3: PHOSPHATIDYLETHANOLAMINE
| Macromolecule | Name: PHOSPHATIDYLETHANOLAMINE / type: ligand / ID: 3 / Number of copies: 2 / Formula: PTY |
|---|---|
| Molecular weight | Theoretical: 734.039 Da |
| Chemical component information |  ChemComp-PTY: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 4 mg/mL |
|---|---|
| Buffer | pH: 7.5 |
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Average electron dose: 64.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.4 µm / Nominal defocus min: 0.7000000000000001 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller


 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)