+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
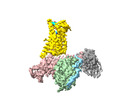
| Title | Cryo-EM structure of the pasireotide-bound SSTR5-Gi complex | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | pasireotide / SSTR5 / Cryo-EM / MEMBRANE PROTEIN/IMMUNE SYSTEM / MEMBRANE PROTEIN-IMMUNE SYSTEM complex | |||||||||
| Function / homology |  Function and homology information Function and homology informationsomatostatin receptor activity / neuropeptide binding / G-protein activation / Activation of the phototransduction cascade / Glucagon-type ligand receptors / Thromboxane signalling through TP receptor / Sensory perception of sweet, bitter, and umami (glutamate) taste / G beta:gamma signalling through PI3Kgamma / G beta:gamma signalling through CDC42 / Cooperation of PDCL (PhLP1) and TRiC/CCT in G-protein beta folding ...somatostatin receptor activity / neuropeptide binding / G-protein activation / Activation of the phototransduction cascade / Glucagon-type ligand receptors / Thromboxane signalling through TP receptor / Sensory perception of sweet, bitter, and umami (glutamate) taste / G beta:gamma signalling through PI3Kgamma / G beta:gamma signalling through CDC42 / Cooperation of PDCL (PhLP1) and TRiC/CCT in G-protein beta folding / Activation of G protein gated Potassium channels / Inhibition of voltage gated Ca2+ channels via Gbeta/gamma subunits / Ca2+ pathway / positive regulation of mini excitatory postsynaptic potential / G alpha (z) signalling events / beta2-adrenergic receptor activity / negative regulation of smooth muscle contraction / High laminar flow shear stress activates signaling by PIEZO1 and PECAM1:CDH5:KDR in endothelial cells / Glucagon-like Peptide-1 (GLP1) regulates insulin secretion / Vasopressin regulates renal water homeostasis via Aquaporins / AMPA selective glutamate receptor signaling pathway / positive regulation of autophagosome maturation / norepinephrine-epinephrine-mediated vasodilation involved in regulation of systemic arterial blood pressure / Adrenaline,noradrenaline inhibits insulin secretion / heat generation / ADP signalling through P2Y purinoceptor 12 / norepinephrine binding / G alpha (q) signalling events / Adrenoceptors / G alpha (i) signalling events / positive regulation of lipophagy / Thrombin signalling through proteinase activated receptors (PARs) / Activation of G protein gated Potassium channels / G-protein activation / G beta:gamma signalling through PI3Kgamma / Prostacyclin signalling through prostacyclin receptor / G beta:gamma signalling through PLC beta / ADP signalling through P2Y purinoceptor 1 / Thromboxane signalling through TP receptor / Presynaptic function of Kainate receptors / G beta:gamma signalling through CDC42 / Inhibition of voltage gated Ca2+ channels via Gbeta/gamma subunits / G alpha (12/13) signalling events / Glucagon-type ligand receptors / G beta:gamma signalling through BTK / ADP signalling through P2Y purinoceptor 12 / Adrenaline,noradrenaline inhibits insulin secretion / Cooperation of PDCL (PhLP1) and TRiC/CCT in G-protein beta folding / negative regulation of multicellular organism growth / negative regulation of G protein-coupled receptor signaling pathway / Ca2+ pathway / Thrombin signalling through proteinase activated receptors (PARs) / cellular response to glucocorticoid stimulus / G alpha (z) signalling events / Extra-nuclear estrogen signaling / photoreceptor outer segment membrane / G alpha (s) signalling events / G alpha (q) signalling events / endosome to lysosome transport / spectrin binding / response to psychosocial stress / adrenergic receptor signaling pathway / G alpha (i) signalling events / Glucagon-like Peptide-1 (GLP1) regulates insulin secretion / diet induced thermogenesis / High laminar flow shear stress activates signaling by PIEZO1 and PECAM1:CDH5:KDR in endothelial cells / Vasopressin regulates renal water homeostasis via Aquaporins / positive regulation of cytokinesis / alkylglycerophosphoethanolamine phosphodiesterase activity / positive regulation of cAMP/PKA signal transduction / adenylate cyclase binding / smooth muscle contraction / G protein-coupled receptor signaling pathway, coupled to cyclic nucleotide second messenger / photoreceptor outer segment / neuropeptide signaling pathway / bone resorption / positive regulation of bone mineralization / potassium channel regulator activity / neuronal dense core vesicle / brown fat cell differentiation / adenylate cyclase inhibitor activity / intercellular bridge / positive regulation of protein localization to cell cortex / regulation of sodium ion transport / Adenylate cyclase inhibitory pathway / T cell migration / adenylate cyclase-activating adrenergic receptor signaling pathway / D2 dopamine receptor binding / response to prostaglandin E / adenylate cyclase regulator activity / G protein-coupled serotonin receptor binding / adenylate cyclase-inhibiting serotonin receptor signaling pathway / cardiac muscle cell apoptotic process / photoreceptor inner segment / regulation of insulin secretion / cellular response to forskolin / receptor-mediated endocytosis / regulation of mitotic spindle organization / response to cold / Peptide ligand-binding receptors Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) / Homo sapiens (human) /    Oplophorus gracilirostris (crustacean) / synthetic construct (others) Oplophorus gracilirostris (crustacean) / synthetic construct (others) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.09 Å | |||||||||
 Authors Authors | Li YG / Meng XY / Yang XR / Ling SL / Shi P / Tian CL / Yang F | |||||||||
| Funding support | 1 items
| |||||||||
 Citation Citation |  Journal: Acta Pharmacol Sin / Year: 2024 Journal: Acta Pharmacol Sin / Year: 2024Title: Structural insights into somatostatin receptor 5 bound with cyclic peptides. Authors: Ying-Ge Li / Xian-Yu Meng / Xiru Yang / Sheng-Long Ling / Pan Shi / Chang-Lin Tian / Fan Yang /  Abstract: Somatostatin receptor 5 (SSTR5) is highly expressed in ACTH-secreting pituitary adenomas and is an important drug target for the treatment of Cushing's disease. Two cyclic SST analog peptides ...Somatostatin receptor 5 (SSTR5) is highly expressed in ACTH-secreting pituitary adenomas and is an important drug target for the treatment of Cushing's disease. Two cyclic SST analog peptides (pasireotide and octreotide) both can activate SSTR5 and SSTR2. Pasireotide is preferential binding to SSTR5 than octreotide, while octreotide is biased to SSTR2 than SSTR5. The lack of selectivity of both pasireotide and octreotide causes side effects, such as hyperglycemia, gastrointestinal disturbance, and abnormal glucose homeostasis. However, little is known about the binding and selectivity mechanisms of pasireotide and octreotide with SSTR5, limiting the development of subtype-selective SST analog drugs specifically targeting SSTR5. Here, we report two cryo-electron microscopy (cryo-EM) structures of SSTR5-Gi complexes activated by pasireotide and octreoitde at resolutions of 3.09 Å and 3.24 Å, respectively. In combination with structural analysis and functional experiments, our results reveal the molecular mechanisms of ligand recognition and receptor activation. We also demonstrate that pasireotide preferentially binds to SSTR5 through the interactions between Tyr(Bzl)/Trp of pasireotide and SSTR5. Moreover, we find that the Q, N, F and ECL2 of SSTR2 play a crucial role in octreotide biased binding of SSTR2. Our results will provide structural insights and offer new opportunities for the drug discovery of better selective pharmaceuticals targeting specific SSTR subtypes. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_39931.map.gz emd_39931.map.gz | 28.7 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-39931-v30.xml emd-39931-v30.xml emd-39931.xml emd-39931.xml | 19.5 KB 19.5 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_39931.png emd_39931.png | 52.1 KB | ||
| Filedesc metadata |  emd-39931.cif.gz emd-39931.cif.gz | 6.9 KB | ||
| Others |  emd_39931_half_map_1.map.gz emd_39931_half_map_1.map.gz emd_39931_half_map_2.map.gz emd_39931_half_map_2.map.gz | 28.2 MB 28.2 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-39931 http://ftp.pdbj.org/pub/emdb/structures/EMD-39931 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-39931 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-39931 | HTTPS FTP |
-Validation report
| Summary document |  emd_39931_validation.pdf.gz emd_39931_validation.pdf.gz | 752.9 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_39931_full_validation.pdf.gz emd_39931_full_validation.pdf.gz | 752.5 KB | Display | |
| Data in XML |  emd_39931_validation.xml.gz emd_39931_validation.xml.gz | 10.7 KB | Display | |
| Data in CIF |  emd_39931_validation.cif.gz emd_39931_validation.cif.gz | 12.4 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-39931 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-39931 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-39931 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-39931 | HTTPS FTP |
-Related structure data
| Related structure data |  8zcjMC  8zbeC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_39931.map.gz / Format: CCP4 / Size: 30.5 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_39931.map.gz / Format: CCP4 / Size: 30.5 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.07 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #2
| File | emd_39931_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #1
| File | emd_39931_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : pasireotide-SSTR5-Gi complex
| Entire | Name: pasireotide-SSTR5-Gi complex |
|---|---|
| Components |
|
-Supramolecule #1: pasireotide-SSTR5-Gi complex
| Supramolecule | Name: pasireotide-SSTR5-Gi complex / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#5 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: Guanine nucleotide-binding protein G(i) subunit alpha-1
| Macromolecule | Name: Guanine nucleotide-binding protein G(i) subunit alpha-1 type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 40.445059 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MGCTLSAEDK AAVERSKMID RNLREDGEKA AREVKLLLLG AGESGKSTIV KQMKIIHEAG YSEEECKQYK AVVYSNTIQS IIAIIRAMG RLKIDFGDSA RADDARQLFV LAGAAEEGFM TAELAGVIKR LWKDSGVQAC FNRSREYQLN DSAAYYLNDL D RIAQPNYI ...String: MGCTLSAEDK AAVERSKMID RNLREDGEKA AREVKLLLLG AGESGKSTIV KQMKIIHEAG YSEEECKQYK AVVYSNTIQS IIAIIRAMG RLKIDFGDSA RADDARQLFV LAGAAEEGFM TAELAGVIKR LWKDSGVQAC FNRSREYQLN DSAAYYLNDL D RIAQPNYI PTQQDVLRTR VKTTGIVETH FTFKDLHFKM FDVGAQRSER KKWIHCFEGV TAIIFCVALS DYDLVLAEDE EM NRMHESM KLFDSICNNK WFTDTSIILF LNKKDLFEEK IKKSPLTICY PEYAGSNTYE EAAAYIQCQF EDLNKRKDTK EIY THFTCS TDTKNVQFVF DAVTDVIIKN NLKDCGLF UniProtKB: Guanine nucleotide-binding protein G(i) subunit alpha-1 |
-Macromolecule #2: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1
| Macromolecule | Name: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1 type: protein_or_peptide / ID: 2 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 41.055867 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MHHHHHHGSL LQSELDQLRQ EAEQLKNQIR DARKACADAT LSQITNNIDP VGRIQMRTRR TLRGHLAKIY AMHWGTDSRL LVSASQDGK LIIWDSYTTN KVHAIPLRSS WVMTCAYAPS GNYVACGGLD NICSIYNLKT REGNVRVSRE LAGHTGYLSC C RFLDDNQI ...String: MHHHHHHGSL LQSELDQLRQ EAEQLKNQIR DARKACADAT LSQITNNIDP VGRIQMRTRR TLRGHLAKIY AMHWGTDSRL LVSASQDGK LIIWDSYTTN KVHAIPLRSS WVMTCAYAPS GNYVACGGLD NICSIYNLKT REGNVRVSRE LAGHTGYLSC C RFLDDNQI VTSSGDTTCA LWDIETGQQT TTFTGHTGDV MSLSLAPDTR LFVSGACDAS AKLWDVREGM CRQTFTGHES DI NAICFFP NGNAFATGSD DATCRLFDLR ADQELMTYSH DNIICGITSV SFSKSGRLLL AGYDDFNCNV WDALKADRAG VLA GHDNRV SCLGVTDDGM AVATGSWDSF LKIWNGSSGG GGSGGGGSSG VSGWRLFKKI S UniProtKB: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1 |
-Macromolecule #3: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2
| Macromolecule | Name: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2 type: protein_or_peptide / ID: 3 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 7.861143 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MASNNTASIA QARKLVEQLK MEANIDRIKV SKAAADLMAY CEAHAKEDPL LTPVPASENP FREKKFFCAI L UniProtKB: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2 |
-Macromolecule #4: ScFv16
| Macromolecule | Name: ScFv16 / type: protein_or_peptide / ID: 4 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 32.724473 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MLLVNQSHQG FNKEHTSKMV SAIVLYVLLA AAAHSAFAVQ LVESGGGLVQ PGGSRKLSCS ASGFAFSSFG MHWVRQAPEK GLEWVAYIS SGSGTIYYAD TVKGRFTISR DDPKNTLFLQ MTSLRSEDTA MYYCVRSIYY YGSSPFDFWG QGTTLTVSSG G GGSGGGGS ...String: MLLVNQSHQG FNKEHTSKMV SAIVLYVLLA AAAHSAFAVQ LVESGGGLVQ PGGSRKLSCS ASGFAFSSFG MHWVRQAPEK GLEWVAYIS SGSGTIYYAD TVKGRFTISR DDPKNTLFLQ MTSLRSEDTA MYYCVRSIYY YGSSPFDFWG QGTTLTVSSG G GGSGGGGS GGGGSSDIVM TQATSSVPVT PGESVSISCR SSKSLLHSNG NTYLYWFLQR PGQSPQLLIY RMSNLASGVP DR FSGSGSG TAFTLTISRL EAEDVGVYYC MQHLEYPLTF GAGTKLELVD ENLYFQGASH HHHHHHH |
-Macromolecule #5: Beta-2 adrenergic receptor,Somatostatin receptor type 5,lgbit (fu...
| Macromolecule | Name: Beta-2 adrenergic receptor,Somatostatin receptor type 5,lgbit (fusion protein) type: protein_or_peptide / ID: 5 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Oplophorus gracilirostris (crustacean) Oplophorus gracilirostris (crustacean) |
| Molecular weight | Theoretical: 62.01593 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MKTIIALSYI FCLVFADYKD DDDKMGQPGN GSAFLLAPNG SHAPDHDVME PLFPASTPSW NASSPGAASG GGDNRTLVGP APSAGARAV LVPVLYLLVC AAGLGGNTLV IYVVLRFAKM KTVTNIYILN LAVADVLYML GLPFLATQNA ASFWPFGPVL C RLVMTLDG ...String: MKTIIALSYI FCLVFADYKD DDDKMGQPGN GSAFLLAPNG SHAPDHDVME PLFPASTPSW NASSPGAASG GGDNRTLVGP APSAGARAV LVPVLYLLVC AAGLGGNTLV IYVVLRFAKM KTVTNIYILN LAVADVLYML GLPFLATQNA ASFWPFGPVL C RLVMTLDG VNQFTSVFCL TVMSVDRYLA VVHPLSSARW RRPRVAKLAS AAAWVLSLCM SLPLLVFADV QEGGTCNASW PE PVGLWGA VFIIYTAVLG FFAPLLVICL CYLLIVVKVR AAGVRVGCVR RRSERKVTRM VLVVVLVFAG CWLPFFTVNI VNL AVALPQ EPASAGLYFF VVILSYANSC ANPVLYGFLS DNFRQSFQKV LCLRKGSGAK DADATEPRPD RIRQQQEATP PAHR AAANG LMQTSKLVFT LEDFVGDWEQ TAAYNLDQVL EQGGVSSLLQ NLAVSVTPIQ RIVRSGENAL KIDIHVIIPY EGLSA DQMA QIEEVFKVVY PVDDHHFKVI LPYGTLVIDG VTPNMLNYFG RPYEGIAVFD GKKITVTGTL WNGNKIIDER LITPDG SML FRVTINS UniProtKB: Beta-2 adrenergic receptor, Somatostatin receptor type 5 |
-Macromolecule #6: 004-DTR-LYS-TYR-PHA-HYP
| Macromolecule | Name: 004-DTR-LYS-TYR-PHA-HYP / type: protein_or_peptide / ID: 6 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism: synthetic construct (others) |
| Molecular weight | Theoretical: 874.016 Da |
| Sequence | String: (004)(DTR)KY(PHA)(HYP) |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Average electron dose: 55.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: SPOT SCAN / Imaging mode: DIFFRACTION / Nominal defocus max: 2.0 µm / Nominal defocus min: 1.4000000000000001 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: PDB ENTRY PDB model - PDB ID: |
|---|---|
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 3.09 Å / Resolution method: FSC 0.143 CUT-OFF / Number images used: 1227503 |
| Initial angle assignment | Type: ANGULAR RECONSTITUTION |
| Final angle assignment | Type: ANGULAR RECONSTITUTION |
 Movie
Movie Controller
Controller




























 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)