+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | SARS-CoV-2 spike + IMCAS-123 | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | SARS-CoV-2 / broadly neutralizing antibodies / VIRUS / ANTIVIRAL PROTEIN / VIRAL PROTEIN-ANTIVIRAL PROTEIN complex / VIRAL PROTEIN/IMMUNE SYSTEM / VIRAL PROTEIN-IMMUNE SYSTEM complex | |||||||||
| Function / homology |  Function and homology information Function and homology informationMaturation of spike protein / viral translation / Translation of Structural Proteins / Virion Assembly and Release / host cell surface / host extracellular space / suppression by virus of host tetherin activity / Induction of Cell-Cell Fusion / structural constituent of virion / entry receptor-mediated virion attachment to host cell ...Maturation of spike protein / viral translation / Translation of Structural Proteins / Virion Assembly and Release / host cell surface / host extracellular space / suppression by virus of host tetherin activity / Induction of Cell-Cell Fusion / structural constituent of virion / entry receptor-mediated virion attachment to host cell / host cell endoplasmic reticulum-Golgi intermediate compartment membrane / membrane fusion / receptor-mediated endocytosis of virus by host cell / Attachment and Entry / positive regulation of viral entry into host cell / receptor-mediated virion attachment to host cell / receptor ligand activity / symbiont-mediated suppression of host innate immune response / host cell surface receptor binding / fusion of virus membrane with host plasma membrane / fusion of virus membrane with host endosome membrane / viral envelope / virion attachment to host cell / SARS-CoV-2 activates/modulates innate and adaptive immune responses / host cell plasma membrane / virion membrane / identical protein binding / membrane / plasma membrane Similarity search - Function | |||||||||
| Biological species |   Homo sapiens (human) Homo sapiens (human) | |||||||||
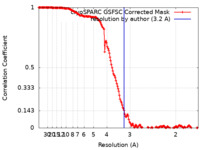
| Method | single particle reconstruction / cryo EM / Resolution: 3.2 Å | |||||||||
 Authors Authors | Tong Z / Cui Y / Xie Y / Tong J / Gao GF / Qi J | |||||||||
| Funding support |  China, 1 items China, 1 items
| |||||||||
 Citation Citation |  Journal: Cell Rep / Year: 2024 Journal: Cell Rep / Year: 2024Title: Deciphering a reliable synergistic bispecific strategy of rescuing antibodies for SARS-CoV-2 escape variants, including BA.2.86, EG.5.1, and JN.1. Authors: Zhou Tong / Jianyu Tong / Wenwen Lei / Yufeng Xie / Yingzi Cui / Guowen Jia / Shihua Li / Zezhong Zhang / Zhimin Cheng / Xiao Xing / Haiyun Ma / Lan Deng / Rong Zhang / Xin Zhao / Kefang Liu ...Authors: Zhou Tong / Jianyu Tong / Wenwen Lei / Yufeng Xie / Yingzi Cui / Guowen Jia / Shihua Li / Zezhong Zhang / Zhimin Cheng / Xiao Xing / Haiyun Ma / Lan Deng / Rong Zhang / Xin Zhao / Kefang Liu / Qihui Wang / Jianxun Qi / Haomin Huang / Rui Song / Zhaoming Su / Guizhen Wu / Jing Lou / George Fu Gao /  Abstract: The game between therapeutic monoclonal antibodies (mAbs) and continuously emerging severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) variants has favored the virus, as most therapeutic ...The game between therapeutic monoclonal antibodies (mAbs) and continuously emerging severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) variants has favored the virus, as most therapeutic mAbs have been evaded. Addressing this challenge, we systematically explored a reproducible bispecific antibody (bsAb)-dependent synergistic effect in this study. It could effectively restore the neutralizing activity of the bsAb when any of its single mAbs is escaped by variants. This synergy is primarily attributed to the binding angle of receptor-binding domain (RBD)-5, facilitating inter-spike cross-linking and promoting cryptic epitope exposure that classical antibody cocktails cannot achieve. Furthermore, RBD-5 with RBD-2, RBD-6, and RBD-7, alongside RBD-8, also exhibit significantly enhanced effects. This study not only shifts the paradigm in understanding antibody interactions but paves the way for developing more effective therapeutic antibodies against rapidly mutating SARS-CoV-2, with Dia-19 already showing promise against emerging variants like BA.2.86, EG.5.1, and JN.1. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_38621.map.gz emd_38621.map.gz | 209.1 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-38621-v30.xml emd-38621-v30.xml emd-38621.xml emd-38621.xml | 17.9 KB 17.9 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_38621_fsc.xml emd_38621_fsc.xml | 15.9 KB | Display |  FSC data file FSC data file |
| Images |  emd_38621.png emd_38621.png | 86.8 KB | ||
| Filedesc metadata |  emd-38621.cif.gz emd-38621.cif.gz | 6.8 KB | ||
| Others |  emd_38621_half_map_1.map.gz emd_38621_half_map_1.map.gz emd_38621_half_map_2.map.gz emd_38621_half_map_2.map.gz | 391.2 MB 391.2 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-38621 http://ftp.pdbj.org/pub/emdb/structures/EMD-38621 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-38621 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-38621 | HTTPS FTP |
-Validation report
| Summary document |  emd_38621_validation.pdf.gz emd_38621_validation.pdf.gz | 921.1 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_38621_full_validation.pdf.gz emd_38621_full_validation.pdf.gz | 920.6 KB | Display | |
| Data in XML |  emd_38621_validation.xml.gz emd_38621_validation.xml.gz | 25.3 KB | Display | |
| Data in CIF |  emd_38621_validation.cif.gz emd_38621_validation.cif.gz | 33 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-38621 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-38621 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-38621 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-38621 | HTTPS FTP |
-Related structure data
| Related structure data |  8xslMC  8xseC  8xsfC  8xsiC  8xsjC  8y0yC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_38621.map.gz / Format: CCP4 / Size: 421.9 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_38621.map.gz / Format: CCP4 / Size: 421.9 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.85 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #2
| File | emd_38621_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #1
| File | emd_38621_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Cryo-EM structure of SARS-CoV-2 spike + IMCAS-123
| Entire | Name: Cryo-EM structure of SARS-CoV-2 spike + IMCAS-123 |
|---|---|
| Components |
|
-Supramolecule #1: Cryo-EM structure of SARS-CoV-2 spike + IMCAS-123
| Supramolecule | Name: Cryo-EM structure of SARS-CoV-2 spike + IMCAS-123 / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#3 |
|---|---|
| Source (natural) | Organism:  |
-Macromolecule #1: Spike glycoprotein
| Macromolecule | Name: Spike glycoprotein / type: protein_or_peptide / ID: 1 / Number of copies: 3 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 141.263344 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: MFVFLVLLPL VSSQCVNLTT RTQLPPAYTN SFTRGVYYPD KVFRSSVLHS TQDLFLPFFS NVTWFHAIHV SGTNGTKRFD NPVLPFNDG VYFASTEKSN IIRGWIFGTT LDSKTQSLLI VNNATNVVIK VCEFQFCNDP FLGVYYHKNN KSWMESEFRV Y SSANNCTF ...String: MFVFLVLLPL VSSQCVNLTT RTQLPPAYTN SFTRGVYYPD KVFRSSVLHS TQDLFLPFFS NVTWFHAIHV SGTNGTKRFD NPVLPFNDG VYFASTEKSN IIRGWIFGTT LDSKTQSLLI VNNATNVVIK VCEFQFCNDP FLGVYYHKNN KSWMESEFRV Y SSANNCTF EYVSQPFLMD LEGKQGNFKN LREFVFKNID GYFKIYSKHT PINLVRDLPQ GFSALEPLVD LPIGINITRF QT LLALHRS YLTPGDSSSG WTAGAAAYYV GYLQPRTFLL KYNENGTITD AVDCALDPLS ETKCTLKSFT VEKGIYQTSN FRV QPTESI VRFPNITNLC PFGEVFNATR FASVYAWNRK RISNCVADYS VLYNSASFST FKCYGVSPTK LNDLCFTNVY ADSF VIRGD EVRQIAPGQT GKIADYNYKL PDDFTGCVIA WNSNNLDSKV GGNYNYLYRL FRKSNLKPFE RDISTEIYQA GSTPC NGVE GFNCYFPLQS YGFQPTNGVG YQPYRVVVLS FELLHAPATV CGPKKSTNLV KNKCVNFNFN GLTGTGVLTE SNKKFL PFQ QFGRDIADTT DAVRDPQTLE ILDITPCSFG GVSVITPGTN TSNQVAVLYQ DVNCTEVPVA IHADQLTPTW RVYSTGS NV FQTRAGCLIG AEHVNNSYEC DIPIGAGICA SYQTQTNSPR RARSVASQSI IAYTMSLGAE NSVAYSNNSI AIPTNFTI S VTTEILPVSM TKTSVDCTMY ICGDSTECSN LLLQYGSFCT QLNRALTGIA VEQDKNTQEV FAQVKQIYKT PPIKDFGGF NFSQILPDPS KPSKRSFIED LLFNKVTLAD AGFIKQYGDC LGDIAARDLI CAQKFNGLTV LPPLLTDEMI AQYTSALLAG TITSGWTFG AGAALQIPFA MQMAYRFNGI GVTQNVLYEN QKLIANQFNS AIGKIQDSLS STASALGKLQ DVVNQNAQAL N TLVKQLSS NFGAISSVLN DILSRLDPPE AEVQIDRLIT GRLQSLQTYV TQQLIRAAEI RASANLAATK MSECVLGQSK RV DFCGKGY HLMSFPQSAP HGVVFLHVTY VPAQEKNFTT APAICHDGKA HFPREGVFVS NGTHWFVTQR NFYEPQIITT DNT FVSGNC DVVIGIVNNT VYDPLQPELD SFKEELDKYF KNHTSPDVDL GDISGINASV VNIQKEIDRL NEVAKNLNES LIDL QELGK YEQYIKWPWY IWLGFIAGLI AIVMVTIMLC CMTSCCSCLK GCCSCGSCCK FDEDDSEPVL KGVKLHYT UniProtKB: Spike glycoprotein |
-Macromolecule #2: IMCAS-123 heavy chain
| Macromolecule | Name: IMCAS-123 heavy chain / type: protein_or_peptide / ID: 2 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 23.917811 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: QVQLVESGGG LVQPGGSLRL SCAASGFTFS SYAMSWVRQA PGKGLEWVSA ISGSGGSTYY ADSVKGRFTI SRDNSKNTLY LQMNSLRAE DTAVYYCAKD HLITMVQPEY FHHWGQGTLV TVSSASTKGP SVFPLAPSSK STSGGTAALG CLVKDYFPEP V TVSWNSGA ...String: QVQLVESGGG LVQPGGSLRL SCAASGFTFS SYAMSWVRQA PGKGLEWVSA ISGSGGSTYY ADSVKGRFTI SRDNSKNTLY LQMNSLRAE DTAVYYCAKD HLITMVQPEY FHHWGQGTLV TVSSASTKGP SVFPLAPSSK STSGGTAALG CLVKDYFPEP V TVSWNSGA LTSGVHTFPA VLQSSGLYSL SSVVTVPSSS LGTQTYICNV NHKPSNTKVD KRVEPKSC |
-Macromolecule #3: IMCAS-123 light chain
| Macromolecule | Name: IMCAS-123 light chain / type: protein_or_peptide / ID: 3 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 23.261725 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: DIQMTQSPSS VSASVGDSVT ITCRASQGIS RWLAWYQQRP GKAPKLLIYA AGNLETGVPS RFSGSGSGTD FTLTISDLQA EDFATYYCQ QADSFPLTFG GGTKVDIKRT VAAPSVFIFP PSDEQLKSGT ASVVCLLNNF YPREAKVQWK VDNALQSGNS Q ESVTEQDS ...String: DIQMTQSPSS VSASVGDSVT ITCRASQGIS RWLAWYQQRP GKAPKLLIYA AGNLETGVPS RFSGSGSGTD FTLTISDLQA EDFATYYCQ QADSFPLTFG GGTKVDIKRT VAAPSVFIFP PSDEQLKSGT ASVVCLLNNF YPREAKVQWK VDNALQSGNS Q ESVTEQDS KDSTYSLSST LTLSKADYEK HKVYACEVTH QGLSSPVTKS FNRGECS |
-Macromolecule #5: 2-acetamido-2-deoxy-beta-D-glucopyranose
| Macromolecule | Name: 2-acetamido-2-deoxy-beta-D-glucopyranose / type: ligand / ID: 5 / Number of copies: 15 / Formula: NAG |
|---|---|
| Molecular weight | Theoretical: 221.208 Da |
| Chemical component information |  ChemComp-NAG: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.4 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Average electron dose: 40.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.0 µm / Nominal defocus min: 1.0 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller













 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)