+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Outward_facing SLC15A4 monomer | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | endolysosomal transporter / PROTEIN TRANSPORT | |||||||||
| Function / homology |  Function and homology information Function and homology informationhistidine transport / mast cell homeostasis / L-histidine transmembrane export from vacuole / peptidoglycan transmembrane transporter activity / L-histidine transmembrane transporter activity / Proton/oligopeptide cotransporters / positive regulation of toll-like receptor 8 signaling pathway / positive regulation of toll-like receptor 7 signaling pathway / peptidoglycan transport / regulation of isotype switching to IgG isotypes ...histidine transport / mast cell homeostasis / L-histidine transmembrane export from vacuole / peptidoglycan transmembrane transporter activity / L-histidine transmembrane transporter activity / Proton/oligopeptide cotransporters / positive regulation of toll-like receptor 8 signaling pathway / positive regulation of toll-like receptor 7 signaling pathway / peptidoglycan transport / regulation of isotype switching to IgG isotypes / SLC15A4:TASL-dependent IRF5 activation / positive regulation of nucleotide-binding oligomerization domain containing 1 signaling pathway / dipeptide import across plasma membrane / positive regulation of toll-like receptor 9 signaling pathway / peptide:proton symporter activity / dipeptide transmembrane transporter activity / positive regulation of nucleotide-binding oligomerization domain containing 2 signaling pathway / regulation of nucleotide-binding domain, leucine rich repeat containing receptor signaling pathway / endolysosome membrane / positive regulation of innate immune response / specific granule membrane / monoatomic ion transport / protein transport / early endosome membrane / innate immune response / lysosomal membrane / Neutrophil degranulation / membrane / plasma membrane Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) / synthetic construct (others) Homo sapiens (human) / synthetic construct (others) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.25 Å | |||||||||
 Authors Authors | Zhang SS / Chen XD / Xie M | |||||||||
| Funding support |  China, 1 items China, 1 items
| |||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2023 Journal: Nat Commun / Year: 2023Title: Structural basis for recruitment of TASL by SLC15A4 in human endolysosomal TLR signaling. Authors: Xudong Chen / Min Xie / Sensen Zhang / Marta Monguió-Tortajada / Jian Yin / Chang Liu / Youqi Zhang / Maeva Delacrétaz / Mingyue Song / Yixue Wang / Lin Dong / Qiang Ding / Boda Zhou / ...Authors: Xudong Chen / Min Xie / Sensen Zhang / Marta Monguió-Tortajada / Jian Yin / Chang Liu / Youqi Zhang / Maeva Delacrétaz / Mingyue Song / Yixue Wang / Lin Dong / Qiang Ding / Boda Zhou / Xiaolin Tian / Haiteng Deng / Lina Xu / Xiaohui Liu / Zi Yang / Qing Chang / Jie Na / Wenwen Zeng / Giulio Superti-Furga / Manuele Rebsamen / Maojun Yang /    Abstract: Toll-like receptors (TLRs) are a class of proteins that play critical roles in recognizing pathogens and initiating innate immune responses. TASL, a recently identified innate immune adaptor protein ...Toll-like receptors (TLRs) are a class of proteins that play critical roles in recognizing pathogens and initiating innate immune responses. TASL, a recently identified innate immune adaptor protein for endolysosomal TLR7/8/9 signaling, is recruited by the lysosomal proton-coupled amino-acid transporter SLC15A4, and then activates IRF5, which in turn triggers the transcription of type I interferons and cytokines. Here, we report three cryo-electron microscopy (cryo-EM) structures of human SLC15A4 in the apo monomeric and dimeric state and as a TASL-bound complex. The apo forms are in an outward-facing conformation, with the dimeric form showing an extensive interface involving four cholesterol molecules. The structure of the TASL-bound complex reveals an unprecedented interaction mode with solute carriers. During the recruitment of TASL, SLC15A4 undergoes a conformational change from an outward-facing, lysosomal lumen-exposed state to an inward-facing state to form a binding pocket, allowing the N-terminal helix of TASL to be inserted into. Our findings provide insights into the molecular basis of regulatory switch involving a human solute carrier and offers an important framework for structure-guided drug discovery targeting SLC15A4-TASL-related human autoimmune diseases. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_36751.map.gz emd_36751.map.gz | 59.7 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-36751-v30.xml emd-36751-v30.xml emd-36751.xml emd-36751.xml | 21.3 KB 21.3 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_36751.png emd_36751.png | 99.5 KB | ||
| Filedesc metadata |  emd-36751.cif.gz emd-36751.cif.gz | 7 KB | ||
| Others |  emd_36751_half_map_1.map.gz emd_36751_half_map_1.map.gz emd_36751_half_map_2.map.gz emd_36751_half_map_2.map.gz | 59.4 MB 59.4 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-36751 http://ftp.pdbj.org/pub/emdb/structures/EMD-36751 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-36751 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-36751 | HTTPS FTP |
-Related structure data
| Related structure data |  8jzrMC  8jzsC  8jzuC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_36751.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_36751.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.8374 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #1
| File | emd_36751_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
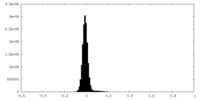
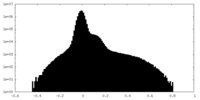
| Density Histograms |
-Half map: #2
| File | emd_36751_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
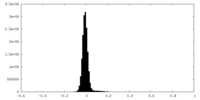
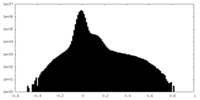
| Density Histograms |
- Sample components
Sample components
-Entire : endolysosomal transporter
| Entire | Name: endolysosomal transporter |
|---|---|
| Components |
|
-Supramolecule #1: endolysosomal transporter
| Supramolecule | Name: endolysosomal transporter / type: organelle_or_cellular_component / ID: 1 / Parent: 0 / Macromolecule list: #1 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: SLC15A4-ALFA tag-SLC15A4-twin-strep tag fusion protein
| Macromolecule | Name: SLC15A4-ALFA tag-SLC15A4-twin-strep tag fusion protein type: protein_or_peptide / ID: 1 Details: SLC15A4 (1-252 residues) + ALFA tag (SRLEEELRRRLTE) + SLC15A4(304-577 resides)+ twin-strep tag sequence Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism: synthetic construct (others) |
| Molecular weight | Theoretical: 62.274246 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: MEGSGGGAGE RAPLLGARRA AAAAAAAGAF AGRRAACGAV LLTELLERAA FYGITSNLVL FLNGAPFCWE GAQASEALLL FMGLTYLGS PFGGWLADAR LGRARAILLS LALYLLGMLA FPLLAAPATR AALCGSARLL NCTAPGPDAA ARCCSPATFA G LVLVGLGV ...String: MEGSGGGAGE RAPLLGARRA AAAAAAAGAF AGRRAACGAV LLTELLERAA FYGITSNLVL FLNGAPFCWE GAQASEALLL FMGLTYLGS PFGGWLADAR LGRARAILLS LALYLLGMLA FPLLAAPATR AALCGSARLL NCTAPGPDAA ARCCSPATFA G LVLVGLGV ATVKANITPF GADQVKDRGP EATRRFFNWF YWSINLGAIL SLGGIAYIQQ NVSFVTGYAI PTVCVGLAFV VF LCGQSVF ITKPPSRLEE ELRRRLTEGG PFTEEKVEDV KALVKIVPVF LALIPYWTVY FQMQTTYVLQ SLHLRIPEIS NIT TTPHTL PAAWLTMFDA VLILLLIPLK DKLVDPILRR HGLLPSSLKR IAVGMFFVMC SAFAAGILES KRLNLVKEKT INQT IGNVV YHAADLSLWW QVPQYLLIGI SEIFASIAGL EFAYSAAPKS MQSAIMGLFF FFSGVGSFVG SGLLALVSIK AIGWM SSHT DFGNINGCYL NYYFFLLAAI QGATLLLFLI ISVKYDHHRD HQRSRANGVP TSRRAGSLEV LFQGPGGGSG GGSWSH PQF EKGGGSGGGS WSHPQFEK UniProtKB: Solute carrier family 15 member 4, Solute carrier family 15 member 4 |
-Macromolecule #2: 2-acetamido-2-deoxy-beta-D-glucopyranose
| Macromolecule | Name: 2-acetamido-2-deoxy-beta-D-glucopyranose / type: ligand / ID: 2 / Number of copies: 1 / Formula: NAG |
|---|---|
| Molecular weight | Theoretical: 221.208 Da |
| Chemical component information |  ChemComp-NAG: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.2 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Average electron dose: 50.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.0 µm / Nominal defocus min: 1.2 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller









 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)