+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of the Rpd3S core complex | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | HDAC / TRANSCRIPTION | |||||||||
| Function / homology |  Function and homology information Function and homology informationSnt2C complex / negative regulation of silent mating-type cassette heterochromatin formation / negative regulation of reciprocal meiotic recombination / Rpd3L complex / protein localization to nucleolar rDNA repeats / negative regulation of rDNA heterochromatin formation / Rpd3L-Expanded complex / Rpd3S complex / rDNA chromatin condensation / nucleophagy ...Snt2C complex / negative regulation of silent mating-type cassette heterochromatin formation / negative regulation of reciprocal meiotic recombination / Rpd3L complex / protein localization to nucleolar rDNA repeats / negative regulation of rDNA heterochromatin formation / Rpd3L-Expanded complex / Rpd3S complex / rDNA chromatin condensation / nucleophagy / HDACs deacetylate histones / histone deacetylase activity, hydrolytic mechanism / histone deacetylase / SUMOylation of chromatin organization proteins / cellular response to nitrogen starvation / regulation of DNA-templated DNA replication initiation / histone deacetylase activity / negative regulation of transcription by RNA polymerase I / Sin3-type complex / NuA4 histone acetyltransferase complex / histone deacetylase complex / Estrogen-dependent gene expression / positive regulation of macroautophagy / nuclear periphery / meiotic cell cycle / transcription elongation by RNA polymerase II / G1/S transition of mitotic cell cycle / double-strand break repair via nonhomologous end joining / G2/M transition of mitotic cell cycle / structural constituent of chromatin / transcription corepressor activity / nucleosome / heterochromatin formation / nucleosome assembly / cellular response to heat / response to oxidative stress / transcription coactivator activity / chromatin remodeling / protein heterodimerization activity / cell division / DNA repair / regulation of DNA-templated transcription / regulation of transcription by RNA polymerase II / chromatin / negative regulation of transcription by RNA polymerase II / positive regulation of transcription by RNA polymerase II / DNA binding / metal ion binding / identical protein binding / nucleus / cytoplasm Similarity search - Function | |||||||||
| Biological species |  | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.37 Å | |||||||||
 Authors Authors | Zhang Y / Gang C | |||||||||
| Funding support | 1 items
| |||||||||
 Citation Citation |  Journal: Cell Res / Year: 2023 Journal: Cell Res / Year: 2023Title: Structural basis for nucleosome binding and catalysis by the yeast Rpd3S/HDAC holoenzyme. Authors: Yueyue Zhang / Mengxue Xu / Po Wang / Jiahui Zhou / Guangxian Wang / Shuailong Han / Gang Cai / Xuejuan Wang /  | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_35450.map.gz emd_35450.map.gz | 136.6 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-35450-v30.xml emd-35450-v30.xml emd-35450.xml emd-35450.xml | 22.5 KB 22.5 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_35450.png emd_35450.png | 63.8 KB | ||
| Filedesc metadata |  emd-35450.cif.gz emd-35450.cif.gz | 8.1 KB | ||
| Others |  emd_35450_half_map_1.map.gz emd_35450_half_map_1.map.gz emd_35450_half_map_2.map.gz emd_35450_half_map_2.map.gz | 134.2 MB 134.2 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-35450 http://ftp.pdbj.org/pub/emdb/structures/EMD-35450 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-35450 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-35450 | HTTPS FTP |
-Related structure data
| Related structure data |  8ihnMC  8ihmC  8ihtC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_35450.map.gz / Format: CCP4 / Size: 144.7 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_35450.map.gz / Format: CCP4 / Size: 144.7 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.07 Å | ||||||||||||||||||||||||||||||||||||
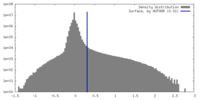
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #2
| File | emd_35450_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #1
| File | emd_35450_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Eaf3 CHD domain bound to the nucleosome
| Entire | Name: Eaf3 CHD domain bound to the nucleosome |
|---|---|
| Components |
|
-Supramolecule #1: Eaf3 CHD domain bound to the nucleosome
| Supramolecule | Name: Eaf3 CHD domain bound to the nucleosome / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#5 |
|---|---|
| Source (natural) | Organism: |
-Macromolecule #1: Histone H3
| Macromolecule | Name: Histone H3 / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism: |
| Molecular weight | Theoretical: 2.563012 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: ARTKQTARKS TGGKAPRKQL ATKA UniProtKB: Histone H3 |
-Macromolecule #2: Transcriptional regulatory protein SIN3
| Macromolecule | Name: Transcriptional regulatory protein SIN3 / type: protein_or_peptide / ID: 2 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 175.047266 KDa |
| Sequence | String: MSQVWHNSNS QSNDVATSND ATGSNERNEK EPSLQGNKPG FVQQQQRITL PSLSALSTKE EDRRDSNGQQ ALTSHAAHIL GYPPPHSNA MPSIATDSAL KQPHEYHPRP KSSSSSPSIN ASLMNAGPAP LPTVGAASFS LSRFDNPLPI KAPVHTEEPK S YNGLQEEE ...String: MSQVWHNSNS QSNDVATSND ATGSNERNEK EPSLQGNKPG FVQQQQRITL PSLSALSTKE EDRRDSNGQQ ALTSHAAHIL GYPPPHSNA MPSIATDSAL KQPHEYHPRP KSSSSSPSIN ASLMNAGPAP LPTVGAASFS LSRFDNPLPI KAPVHTEEPK S YNGLQEEE KATQRPQDCK EVPAGVQPAD APDPSSNHAD ANDDNNNNEN SHDEDADYRP LNVKDALSYL EQVKFQFSSR PD IYNLFLD IMKDFKSQAI DTPGVIERVS TLFRGYPILI QGFNTFLPQG YRIECSSNPD DPIRVTTPMG TTTVNNNISP SGR GTTDAQ ELGSFPESDG NGVQQPSNVP MVPSSVYQSE QNQDQQQSLP LLATSSGLPS IQQPEMPAHR QIPQSQSLVP QEDA KKNVD VEFSQAISYV NKIKTRFADQ PDIYKHFLEI LQTYQREQKP INEVYAQVTH LFQNAPDLLE DFKKFLPDSS ASANQ QVQH AQQHAQQQHE AQMHAQAQAQ AQAQAQVEQQ KQQQQFLYPA SGYYGHPSNR GIPQQNLPPI GSFSPPTNGS TVHEAY QDQ QHMQPPHFMP LPSIVQHGPN MVHQGIANEN PPLSDLRTSL TEQYAPSSIQ HQQQHPQSIS PIANTQYGDI PVRPEID LD PSIVPVVPEP TEPIENNISL NEEVTFFEKA KRYIGNKHLY TEFLKILNLY SQDILDLDDL VEKVDFYLGS NKELFTWF K NFVGYQEKTK CIENIVHEKH RLDLDLCEAF GPSYKRLPKS DTFMPCSGRD DMCWEVLNDE WVGHPVWASE DSGFIAHRK NQYEETLFKI EEERHEYDFY IESNLRTIQC LETIVNKIEN MTENEKANFK LPPGLGHTSM TIYKKVIRKV YDKERGFEII DALHEHPAV TAPVVLKRLK QKDEEWRRAQ REWNKVWREL EQKVFFKSLD HLGLTFKQAD KKLLTTKQLI SEISSIKVDQ T NKKIHWLT PKPKSQLDFD FPDKNIFYDI LCLADTFITH TTAYSNPDKE RLKDLLKYFI SLFFSISFEK IEESLYSHKQ NV SESSGSD DGSSIASRKR PYQQEMSLLD ILHRSRYQKL KRSNDEDGKV PQLSEPPEEE PNTIEEEELI DEEAKNPWLT GNL VEEANS QGIIQNRSIF NLFANTNIYI FFRHWTTIYE RLLEIKQMNE RVTKEINTRS TVTFAKDLDL LSSQLSEMGL DFVG EDAYK QVLRLSRRLI NGDLEHQWFE ESLRQAYNNK AFKLYTIDKV TQSLVKHAHT LMTDAKTAEI MALFVKDRNA STTSA KDQI IYRLQVRSHM SNTENMFRIE FDKRTLHVSI QYIALDDLTL KEPKADEDKW KYYVTSYALP HPTEGIPHEK LKIPFL ERL IEFGQDIDGT EVDEEFSPEG ISVSTLKIKI QPITYQLHIE NGSYDVFTRK ATNKYPTIAN DNTQKGMVSQ KKELISK FL DCAVGLRNNL DEAQKLSMQK KWENLKDSIA KTSAGNQGIE SETEKGKITK QEQSDNLDSS TASVLPASIT TVPQDDNI E TTGNTESSDK GAKIQ UniProtKB: Transcriptional regulatory protein SIN3 |
-Macromolecule #3: Histone deacetylase RPD3
| Macromolecule | Name: Histone deacetylase RPD3 / type: protein_or_peptide / ID: 3 / Number of copies: 1 / Enantiomer: LEVO / EC number: histone deacetylase |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 48.961957 KDa |
| Sequence | String: MVYEATPFDP ITVKPSDKRR VAYFYDADVG NYAYGAGHPM KPHRIRMAHS LIMNYGLYKK MEIYRAKPAT KQEMCQFHTD EYIDFLSRV TPDNLEMFKR ESVKFNVGDD CPVFDGLYEY CSISGGGSME GAARLNRGKC DVAVNYAGGL HHAKKSEASG F CYLNDIVL ...String: MVYEATPFDP ITVKPSDKRR VAYFYDADVG NYAYGAGHPM KPHRIRMAHS LIMNYGLYKK MEIYRAKPAT KQEMCQFHTD EYIDFLSRV TPDNLEMFKR ESVKFNVGDD CPVFDGLYEY CSISGGGSME GAARLNRGKC DVAVNYAGGL HHAKKSEASG F CYLNDIVL GIIELLRYHP RVLYIDIDVH HGDGVEEAFY TTDRVMTCSF HKYGEFFPGT GELRDIGVGA GKNYAVNVPL RD GIDDATY RSVFEPVIKK IMEWYQPSAV VLQCGGDSLS GDRLGCFNLS MEGHANCVNY VKSFGIPMMV VGGGGYTMRN VAR TWCFET GLLNNVVLDK DLPYNEYYEY YGPDYKLSVR PSNMFNVNTP EYLDKVMTNI FANLENTKYA PSVQLNHTPR DAED LGDVE EDSAEAKDTK GGSQYARDLH VEHDNEFY UniProtKB: Histone deacetylase RPD3 |
-Macromolecule #4: RCO1 isoform 1
| Macromolecule | Name: RCO1 isoform 1 / type: protein_or_peptide / ID: 4 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 78.951305 KDa |
| Sequence | String: MDTSKKDTTR SPSHSNSSSP SSSSLSSSSS KEKKRPKRLS SQNVNYDLKR RKIITSEGIE RSFKNEHSNL AVEDNIPEEE PKELLEKDS KGNIIKLNEP STISEDSKVS VTGLPLNKGP SEKIKRESLW NYRKNLGGQS NNSEMTLVPS KRFTQVPKNF Q DLNRNDLK ...String: MDTSKKDTTR SPSHSNSSSP SSSSLSSSSS KEKKRPKRLS SQNVNYDLKR RKIITSEGIE RSFKNEHSNL AVEDNIPEEE PKELLEKDS KGNIIKLNEP STISEDSKVS VTGLPLNKGP SEKIKRESLW NYRKNLGGQS NNSEMTLVPS KRFTQVPKNF Q DLNRNDLK TFLTENMTEE SNIRSTIGWN GDIINRTRDR EPESDRDNKK LSNIRTKIIL STNATYDSKS KLFGQNSIKS TS NASEKIF RDKNNSTIDF ENEDFCSACN QSGSFLCCDT CPKSFHFLCL DPPIDPNNLP KGDWHCNECK FKIFINNSMA TLK KIESNF IKQNNNVKIF AKLLFNIDSH NPKQFQLPNY IKETFPAVKT GSRGQYSDEN DKIPLTDRQL FNTSYGQSIT KLDS YNPDT HIDSNSGKFL ICYKCNQTRL GSWSHPENSR LIMTCDYCQT PWHLDCVPRA SFKNLGSKWK CPLHSPTKVY KKIHH CQED NSVNYKVWKK QRLINKKNQL YYEPLQKIGY QNNGNIQIIP TTSHTDYDFN QDFKITQIDE NSIKYDFFDK IYKSKM VQK RKLFQFQESL IDKLVSNGSQ NGNSEDNMVK DIASLIYFQV SNNDKSSNNK SASKSNNLRK LWDLKELTNV VVPNELD SI QFNDFSSDEI KHLLYLKKII ESKPKEELLK FLNIENPENQ SE UniProtKB: RCO1 isoform 1 |
-Macromolecule #5: Chromatin modification-related protein EAF3
| Macromolecule | Name: Chromatin modification-related protein EAF3 / type: protein_or_peptide / ID: 5 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 45.266406 KDa |
| Sequence | String: MVDLEQEFAL GGRCLAFHGP LMYEAKILKI WDPSSKMYTS IPNDKPGGSS QATKEIKPQK LGEDESIPEE IINGKCFFIH YQGWKSSWD EWVGYDRIRA YNEENIAMKK RLANEAKEAK KSLLEQQKKK KLSTSLGGPS NGGKRKGDSR SNASISKSTS Q SFLTSSVS ...String: MVDLEQEFAL GGRCLAFHGP LMYEAKILKI WDPSSKMYTS IPNDKPGGSS QATKEIKPQK LGEDESIPEE IINGKCFFIH YQGWKSSWD EWVGYDRIRA YNEENIAMKK RLANEAKEAK KSLLEQQKKK KLSTSLGGPS NGGKRKGDSR SNASISKSTS Q SFLTSSVS GRKSGRSSAN SLHPGSSLRS SSDQNGNDDR RRSSSLSPNM LHHIAGYPTP KISLQIPIKL KSVLVDDWEY VT KDKKICR LPADVTVEMV LNKYEHEVSQ ELESPGSQSQ LSEYCAGLKL YFDKCLGNML LYRLERLQYD ELLKKSSKDQ KPL VPIRIY GAIHLLRLIS VLPELISSTT MDLQSCQLLI KQTEDFLVWL LMHVDEYFND KDPNRSDDAL YVNTSSQYEG VALG M UniProtKB: Chromatin modification-related protein EAF3 |
-Macromolecule #6: ZINC ION
| Macromolecule | Name: ZINC ION / type: ligand / ID: 6 / Number of copies: 1 / Formula: ZN |
|---|---|
| Molecular weight | Theoretical: 65.409 Da |
-Macromolecule #7: CALCIUM ION
| Macromolecule | Name: CALCIUM ION / type: ligand / ID: 7 / Number of copies: 1 / Formula: CA |
|---|---|
| Molecular weight | Theoretical: 40.078 Da |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.6 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Average electron dose: 53.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.8000000000000003 µm / Nominal defocus min: 1.8 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller














 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)