[English] 日本語
 Yorodumi
Yorodumi- EMDB-35033: OutLHCIs plus Lhcb9 moiety of Large Photosystem I from Physcomitr... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | OutLHCIs plus Lhcb9 moiety of Large Photosystem I from Physcomitrium patens | |||||||||||||||
 Map data Map data | Focused refinement map of the outLHCIs plus Lhcb9 moiety of PpPSI-L | |||||||||||||||
 Sample Sample |
| |||||||||||||||
 Keywords Keywords | Photosystem I / Lhcb9 / PHOTOSYNTHESIS | |||||||||||||||
| Biological species |  Physcomitrium patens (plant) Physcomitrium patens (plant) | |||||||||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.29 Å | |||||||||||||||
 Authors Authors | Sun HY / Li M / Pan XW | |||||||||||||||
| Funding support |  China, 4 items China, 4 items
| |||||||||||||||
 Citation Citation |  Journal: Nat Plants / Year: 2023 Journal: Nat Plants / Year: 2023Title: Structural insights into the assembly and energy transfer of the Lhcb9-dependent photosystem I from moss Physcomitrium patens. Authors: Haiyu Sun / Hui Shang / Xiaowei Pan / Mei Li /  Abstract: In plants and green algae, light-harvesting complexes I and II (LHCI and LHCII) constitute the antennae of photosystem I (PSI), thus effectively increasing the cross-section of the PSI core. The moss ...In plants and green algae, light-harvesting complexes I and II (LHCI and LHCII) constitute the antennae of photosystem I (PSI), thus effectively increasing the cross-section of the PSI core. The moss Physcomitrium patens (P. patens) represents a well-studied primary land-dwelling photosynthetic autotroph branching from the common ancestor of green algae and land plants at the early stage of evolution. P. patens possesses at least three types of PSI with different antenna sizes. The largest PSI form (PpPSI-L) exhibits a unique organization found neither in flowering plants nor in algae. Its formation is mediated by the P. patens-specific LHC protein, Lhcb9. While previous studies have revealed the overall architecture of PpPSI-L, its assembly details and the relationship between different PpPSI types remain unclear. Here we report the high-resolution structure of PpPSI-L. We identified 14 PSI core subunits, one Lhcb9, one phosphorylated LHCII trimer and eight LHCI monomers arranged as two belts. Our structural analysis established the essential role of Lhcb9 and the phosphorylated LHCII in stabilizing the complex. In addition, our results suggest that PpPSI switches between different types, which share identical modules. This feature may contribute to the dynamic adjustment of the light-harvesting capability of PSI under different light conditions. | |||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_35033.map.gz emd_35033.map.gz | 7.5 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-35033-v30.xml emd-35033-v30.xml emd-35033.xml emd-35033.xml | 12.4 KB 12.4 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_35033.png emd_35033.png | 89.2 KB | ||
| Others |  emd_35033_half_map_1.map.gz emd_35033_half_map_1.map.gz emd_35033_half_map_2.map.gz emd_35033_half_map_2.map.gz | 98.5 MB 98.5 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-35033 http://ftp.pdbj.org/pub/emdb/structures/EMD-35033 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-35033 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-35033 | HTTPS FTP |
-Validation report
| Summary document |  emd_35033_validation.pdf.gz emd_35033_validation.pdf.gz | 771.3 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_35033_full_validation.pdf.gz emd_35033_full_validation.pdf.gz | 770.9 KB | Display | |
| Data in XML |  emd_35033_validation.xml.gz emd_35033_validation.xml.gz | 13.6 KB | Display | |
| Data in CIF |  emd_35033_validation.cif.gz emd_35033_validation.cif.gz | 15.7 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-35033 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-35033 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-35033 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-35033 | HTTPS FTP |
-Related structure data
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_35033.map.gz / Format: CCP4 / Size: 125 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_35033.map.gz / Format: CCP4 / Size: 125 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Focused refinement map of the outLHCIs plus Lhcb9 moiety of PpPSI-L | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.04 Å | ||||||||||||||||||||||||||||||||||||
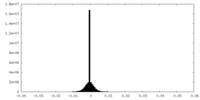
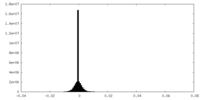
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #2
| File | emd_35033_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
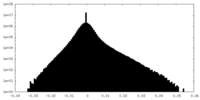
| Density Histograms |
-Half map: #1
| File | emd_35033_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
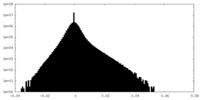
| Density Histograms |
- Sample components
Sample components
-Entire : Outer LHCIs plus Lhcb9 moiety of Large Photosystem I from Physcom...
| Entire | Name: Outer LHCIs plus Lhcb9 moiety of Large Photosystem I from Physcomitrium patens |
|---|---|
| Components |
|
-Supramolecule #1: Outer LHCIs plus Lhcb9 moiety of Large Photosystem I from Physcom...
| Supramolecule | Name: Outer LHCIs plus Lhcb9 moiety of Large Photosystem I from Physcomitrium patens type: complex / ID: 1 / Parent: 0 |
|---|---|
| Source (natural) | Organism:  Physcomitrium patens (plant) Physcomitrium patens (plant) |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 6.5 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Average electron dose: 60.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source: OTHER |
| Electron optics | Illumination mode: OTHER / Imaging mode: OTHER / Nominal defocus max: 2.5 µm / Nominal defocus min: 1.5 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: PDB ENTRY PDB model - PDB ID: Details: 7KSQ |
|---|---|
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 3.29 Å / Resolution method: FSC 0.143 CUT-OFF / Number images used: 89945 |
| Initial angle assignment | Type: PROJECTION MATCHING |
| Final angle assignment | Type: PROJECTION MATCHING |
 Movie
Movie Controller
Controller









 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)