[English] 日本語
 Yorodumi
Yorodumi- EMDB-34813: Structure of transforming growth factor beta induced protein (TGF... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Structure of transforming growth factor beta induced protein (TGFBIp) G623R fibril | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Pathological fibrils / TGFBI related corneal dystrophy / PROTEIN FIBRIL | |||||||||
| Function / homology |  Function and homology information Function and homology informationnegative regulation of cell adhesion / extracellular matrix binding / extracellular matrix structural constituent / basement membrane / chondrocyte differentiation / collagen binding / extracellular matrix organization / visual perception / cell adhesion molecule binding / trans-Golgi network ...negative regulation of cell adhesion / extracellular matrix binding / extracellular matrix structural constituent / basement membrane / chondrocyte differentiation / collagen binding / extracellular matrix organization / visual perception / cell adhesion molecule binding / trans-Golgi network / integrin binding / extracellular matrix / angiogenesis / cell adhesion / cell population proliferation / Amyloid fiber formation / : / extracellular exosome / extracellular region / identical protein binding / plasma membrane Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | helical reconstruction / cryo EM / Resolution: 4.9 Å | |||||||||
 Authors Authors | Low JYK / Pervushin K | |||||||||
| Funding support |  Singapore, 2 items Singapore, 2 items
| |||||||||
 Citation Citation |  Journal: Commun Biol / Year: 2023 Journal: Commun Biol / Year: 2023Title: Release of frustration drives corneal amyloid disaggregation by brain chaperone. Authors: Jia Yi Kimberly Low / Xiangyan Shi / Venkatraman Anandalakshmi / Dawn Neo / Gary Swee Lim Peh / Siew Kwan Koh / Lei Zhou / M K Abdul Rahim / Ketti Boo / JiaXuan Lee / Harini Mohanram / Reema ...Authors: Jia Yi Kimberly Low / Xiangyan Shi / Venkatraman Anandalakshmi / Dawn Neo / Gary Swee Lim Peh / Siew Kwan Koh / Lei Zhou / M K Abdul Rahim / Ketti Boo / JiaXuan Lee / Harini Mohanram / Reema Alag / Yuguang Mu / Jodhbir S Mehta / Konstantin Pervushin /   Abstract: TGFBI-related corneal dystrophy (CD) is characterized by the accumulation of insoluble protein deposits in the corneal tissues, eventually leading to progressive corneal opacity. Here we show that ...TGFBI-related corneal dystrophy (CD) is characterized by the accumulation of insoluble protein deposits in the corneal tissues, eventually leading to progressive corneal opacity. Here we show that ATP-independent amyloid-β chaperone L-PGDS can effectively disaggregate corneal amyloids in surgically excised human cornea of TGFBI-CD patients and release trapped amyloid hallmark proteins. Since the mechanism of amyloid disassembly by ATP-independent chaperones is unknown, we reconstructed atomic models of the amyloids self-assembled from TGFBIp-derived peptides and their complex with L-PGDS using cryo-EM and NMR. We show that L-PGDS specifically recognizes structurally frustrated regions in the amyloids and releases those frustrations. The released free energy increases the chaperone's binding affinity to amyloids, resulting in local restructuring and breakage of amyloids to protofibrils. Our mechanistic model provides insights into the alternative source of energy utilized by ATP-independent disaggregases and highlights the possibility of using these chaperones as treatment strategies for different types of amyloid-related diseases. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_34813.map.gz emd_34813.map.gz | 3 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-34813-v30.xml emd-34813-v30.xml emd-34813.xml emd-34813.xml | 14 KB 14 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_34813.png emd_34813.png | 44.1 KB | ||
| Filedesc metadata |  emd-34813.cif.gz emd-34813.cif.gz | 5 KB | ||
| Others |  emd_34813_half_map_1.map.gz emd_34813_half_map_1.map.gz emd_34813_half_map_2.map.gz emd_34813_half_map_2.map.gz | 81 MB 81.1 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-34813 http://ftp.pdbj.org/pub/emdb/structures/EMD-34813 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-34813 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-34813 | HTTPS FTP |
-Related structure data
| Related structure data |  8hiaMC  8hgaC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_34813.map.gz / Format: CCP4 / Size: 103 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_34813.map.gz / Format: CCP4 / Size: 103 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.85 Å | ||||||||||||||||||||||||||||||||||||
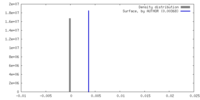
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #2
| File | emd_34813_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
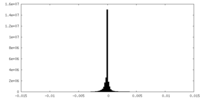
| Density Histograms |
-Half map: #1
| File | emd_34813_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Mutant of TGFBIp peptide derived from TGFBIp
| Entire | Name: Mutant of TGFBIp peptide derived from TGFBIp |
|---|---|
| Components |
|
-Supramolecule #1: Mutant of TGFBIp peptide derived from TGFBIp
| Supramolecule | Name: Mutant of TGFBIp peptide derived from TGFBIp / type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 21.43 kDa/nm |
-Macromolecule #1: Transforming growth factor-beta-induced protein ig-h3
| Macromolecule | Name: Transforming growth factor-beta-induced protein ig-h3 / type: protein_or_peptide / ID: 1 / Number of copies: 20 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 2.547922 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: EPVAEPDIMA TNRVVHVITN VLQ UniProtKB: Transforming growth factor-beta-induced protein ig-h3 |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | helical reconstruction |
| Aggregation state | filament |
- Sample preparation
Sample preparation
| Buffer | pH: 8 / Details: 20mM TRIS |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Average electron dose: 8.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: OTHER / Imaging mode: BRIGHT FIELD / Nominal defocus max: 3.0 µm / Nominal defocus min: 1.0 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Final reconstruction | Applied symmetry - Helical parameters - Δz: 2.75 Å Applied symmetry - Helical parameters - Δ&Phi: -179.4 ° Applied symmetry - Helical parameters - Axial symmetry: C1 (asymmetric) Resolution.type: BY AUTHOR / Resolution: 4.9 Å / Resolution method: FSC 0.143 CUT-OFF / Number images used: 48522 |
|---|---|
| Startup model | Type of model: OTHER / Details: Featureless cylinder |
| Final angle assignment | Type: NOT APPLICABLE |
 Movie
Movie Controller
Controller



 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)