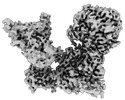
登録情報 データベース : EMDB / ID : EMD-34084タイトル Cryo-EM Structure of FGF23-FGFR4-aKlotho-HS Quaternary Complex 複合体 : 1:2:1:1 FGF23-FGFR4-aKlotho-HS Quaternary Complex複合体 : FGFR4-aKlotho-HSタンパク質・ペプチド : Fibroblast growth factor receptor 4タンパク質・ペプチド : Klotho複合体 : FGF23タンパク質・ペプチド : Fibroblast growth factor 23リガンド : COPPER (II) IONリガンド : ZINC ION / / / / 機能・相同性 分子機能 ドメイン・相同性 構成要素
/ / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / 生物種 Homo sapiens (ヒト)手法 / / 解像度 : 3.03 Å Mohammadi M / Chen L 資金援助 Organization Grant number 国 National Natural Science Foundation of China (NSFC) OJQD2022007
ジャーナル : Nature / 年 : 2023タイトル : Structural basis for FGF hormone signalling.著者 : Lingfeng Chen / Lili Fu / Jingchuan Sun / Zhiqiang Huang / Mingzhen Fang / Allen Zinkle / Xin Liu / Junliang Lu / Zixiang Pan / Yang Wang / Guang Liang / Xiaokun Li / Gaozhi Chen / Moosa Mohammadi / 要旨 : α/βKlotho coreceptors simultaneously engage fibroblast growth factor (FGF) hormones (FGF19, FGF21 and FGF23) and their cognate cell-surface FGF receptors (FGFR1-4) thereby stabilizing the endocrine ... α/βKlotho coreceptors simultaneously engage fibroblast growth factor (FGF) hormones (FGF19, FGF21 and FGF23) and their cognate cell-surface FGF receptors (FGFR1-4) thereby stabilizing the endocrine FGF-FGFR complex. However, these hormones still require heparan sulfate (HS) proteoglycan as an additional coreceptor to induce FGFR dimerization/activation and hence elicit their essential metabolic activities. To reveal the molecular mechanism underpinning the coreceptor role of HS, we solved cryo-electron microscopy structures of three distinct 1:2:1:1 FGF23-FGFR-αKlotho-HS quaternary complexes featuring the 'c' splice isoforms of FGFR1 (FGFR1c), FGFR3 (FGFR3c) or FGFR4 as the receptor component. These structures, supported by cell-based receptor complementation and heterodimerization experiments, reveal that a single HS chain enables FGF23 and its primary FGFR within a 1:1:1 FGF23-FGFR-αKlotho ternary complex to jointly recruit a lone secondary FGFR molecule leading to asymmetric receptor dimerization and activation. However, αKlotho does not directly participate in recruiting the secondary receptor/dimerization. We also show that the asymmetric mode of receptor dimerization is applicable to paracrine FGFs that signal solely in an HS-dependent fashion. Our structural and biochemical data overturn the current symmetric FGFR dimerization paradigm and provide blueprints for rational discovery of modulators of FGF signalling as therapeutics for human metabolic diseases and cancer. 履歴 登録 2022年8月13日 - ヘッダ(付随情報) 公開 2023年6月14日 - マップ公開 2023年6月14日 - 更新 2025年6月18日 - 現状 2025年6月18日 処理サイト : PDBj / 状態 : 公開
すべて表示 表示を減らす
 データを開く
データを開く 基本情報
基本情報
 マップデータ
マップデータ 試料
試料 キーワード
キーワード 機能・相同性情報
機能・相同性情報 Homo sapiens (ヒト)
Homo sapiens (ヒト) データ登録者
データ登録者 中国, 1件
中国, 1件  引用
引用 ジャーナル: Nature / 年: 2023
ジャーナル: Nature / 年: 2023

 構造の表示
構造の表示 ダウンロードとリンク
ダウンロードとリンク emd_34084.map.gz
emd_34084.map.gz EMDBマップデータ形式
EMDBマップデータ形式 emd-34084-v30.xml
emd-34084-v30.xml emd-34084.xml
emd-34084.xml EMDBヘッダ
EMDBヘッダ emd_34084.png
emd_34084.png emd_34084_msk_1.map
emd_34084_msk_1.map マスクマップ
マスクマップ emd-34084.cif.gz
emd-34084.cif.gz emd_34084_half_map_1.map.gz
emd_34084_half_map_1.map.gz emd_34084_half_map_2.map.gz
emd_34084_half_map_2.map.gz http://ftp.pdbj.org/pub/emdb/structures/EMD-34084
http://ftp.pdbj.org/pub/emdb/structures/EMD-34084 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-34084
ftp://ftp.pdbj.org/pub/emdb/structures/EMD-34084 emd_34084_validation.pdf.gz
emd_34084_validation.pdf.gz EMDB検証レポート
EMDB検証レポート emd_34084_full_validation.pdf.gz
emd_34084_full_validation.pdf.gz emd_34084_validation.xml.gz
emd_34084_validation.xml.gz emd_34084_validation.cif.gz
emd_34084_validation.cif.gz https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-34084
https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-34084 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-34084
ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-34084 リンク
リンク EMDB (EBI/PDBe) /
EMDB (EBI/PDBe) /  EMDataResource
EMDataResource マップ
マップ ダウンロード / ファイル: emd_34084.map.gz / 形式: CCP4 / 大きさ: 244.1 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES)
ダウンロード / ファイル: emd_34084.map.gz / 形式: CCP4 / 大きさ: 244.1 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) emd_34084_msk_1.map
emd_34084_msk_1.map 試料の構成要素
試料の構成要素 Homo sapiens (ヒト)
Homo sapiens (ヒト) Homo sapiens (ヒト)
Homo sapiens (ヒト) Homo sapiens (ヒト)
Homo sapiens (ヒト) Homo sapiens (ヒト)
Homo sapiens (ヒト) Homo sapiens (ヒト)
Homo sapiens (ヒト) Homo sapiens (ヒト)
Homo sapiens (ヒト)

 解析
解析 試料調製
試料調製 電子顕微鏡法
電子顕微鏡法 FIELD EMISSION GUN
FIELD EMISSION GUN
 ムービー
ムービー コントローラー
コントローラー














 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)