+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-30309 | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Human DMC1 post-synaptic complexes with mismatched dsDNA | ||||||||||||
 Map data Map data | |||||||||||||
 Sample Sample |
| ||||||||||||
 Keywords Keywords | meiotic homologous recombination / DNA repair / ATPase / RECOMBINATION / RECOMBINATION-DNA complex | ||||||||||||
| Function / homology |  Function and homology information Function and homology informationfemale gamete generation / DNA recombinase assembly / homologous chromosome pairing at meiosis / chromosome organization involved in meiotic cell cycle / double-strand break repair involved in meiotic recombination / DNA strand invasion / mitotic recombination / lateral element / DNA strand exchange activity / oocyte maturation ...female gamete generation / DNA recombinase assembly / homologous chromosome pairing at meiosis / chromosome organization involved in meiotic cell cycle / double-strand break repair involved in meiotic recombination / DNA strand invasion / mitotic recombination / lateral element / DNA strand exchange activity / oocyte maturation / reciprocal meiotic recombination / ATP-dependent DNA damage sensor activity / male meiosis I / spermatid development / ATP-dependent activity, acting on DNA / ovarian follicle development / condensed nuclear chromosome / meiotic cell cycle / Meiotic recombination / single-stranded DNA binding / chromosome / site of double-strand break / double-stranded DNA binding / spermatogenesis / chromosome, telomeric region / DNA binding / nucleoplasm / ATP binding / identical protein binding / nucleus Similarity search - Function | ||||||||||||
| Biological species |  Homo sapiens (human) / synthetic construct (others) Homo sapiens (human) / synthetic construct (others) | ||||||||||||
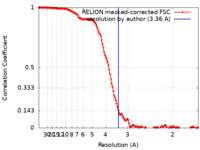
| Method | helical reconstruction / cryo EM / Resolution: 3.36 Å | ||||||||||||
 Authors Authors | Luo SC / Yeh HY / Chi P / Ho MC / Tsai MD | ||||||||||||
| Funding support |  Taiwan, 3 items Taiwan, 3 items
| ||||||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2021 Journal: Nat Commun / Year: 2021Title: Identification of fidelity-governing factors in human recombinases DMC1 and RAD51 from cryo-EM structures. Authors: Shih-Chi Luo / Hsin-Yi Yeh / Wei-Hsuan Lan / Yi-Min Wu / Cheng-Han Yang / Hao-Yen Chang / Guan-Chin Su / Chia-Yi Lee / Wen-Jin Wu / Hung-Wen Li / Meng-Chiao Ho / Peter Chi / Ming-Daw Tsai /  Abstract: Both high-fidelity and mismatch-tolerant recombination, catalyzed by RAD51 and DMC1 recombinases, respectively, are indispensable for genomic integrity. Here, we use cryo-EM, MD simulation and ...Both high-fidelity and mismatch-tolerant recombination, catalyzed by RAD51 and DMC1 recombinases, respectively, are indispensable for genomic integrity. Here, we use cryo-EM, MD simulation and functional analysis to elucidate the structural basis for the mismatch tolerance of DMC1. Structural analysis of DMC1 presynaptic and postsynaptic complexes suggested that the lineage-specific Loop 1 Gln244 (Met243 in RAD51) may help stabilize DNA backbone, whereas Loop 2 Pro274 and Gly275 (Val273/Asp274 in RAD51) may provide an open "triplet gate" for mismatch tolerance. In support, DMC1-Q244M displayed marked increase in DNA dynamics, leading to unobservable DNA map. MD simulation showed highly dispersive mismatched DNA ensemble in RAD51 but well-converged DNA in DMC1 and RAD51-V273P/D274G. Replacing Loop 1 or Loop 2 residues in DMC1 with RAD51 counterparts enhanced DMC1 fidelity, while reciprocal mutations in RAD51 attenuated its fidelity. Our results show that three Loop 1/Loop 2 residues jointly enact contrasting fidelities of DNA recombinases. | ||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_30309.map.gz emd_30309.map.gz | 202.1 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-30309-v30.xml emd-30309-v30.xml emd-30309.xml emd-30309.xml | 16.7 KB 16.7 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_30309_fsc.xml emd_30309_fsc.xml | 13.7 KB | Display |  FSC data file FSC data file |
| Images |  emd_30309.png emd_30309.png | 106.5 KB | ||
| Filedesc metadata |  emd-30309.cif.gz emd-30309.cif.gz | 6.6 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-30309 http://ftp.pdbj.org/pub/emdb/structures/EMD-30309 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-30309 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-30309 | HTTPS FTP |
-Related structure data
| Related structure data |  7c99MC  7c98C  7c9aC  7c9cC  7cgyC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_30309.map.gz / Format: CCP4 / Size: 216 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_30309.map.gz / Format: CCP4 / Size: 216 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.84 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : DMC1-dsDNA filament with mismatched dsDNA
| Entire | Name: DMC1-dsDNA filament with mismatched dsDNA |
|---|---|
| Components |
|
-Supramolecule #1: DMC1-dsDNA filament with mismatched dsDNA
| Supramolecule | Name: DMC1-dsDNA filament with mismatched dsDNA / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#3 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Supramolecule #2: DMC1
| Supramolecule | Name: DMC1 / type: complex / ID: 2 / Parent: 1 / Macromolecule list: #1 |
|---|
-Supramolecule #3: DNA
| Supramolecule | Name: DNA / type: complex / ID: 3 / Parent: 1 / Macromolecule list: #2-#3 |
|---|
-Macromolecule #1: Meiotic recombination protein DMC1/LIM15 homolog
| Macromolecule | Name: Meiotic recombination protein DMC1/LIM15 homolog / type: protein_or_peptide / ID: 1 / Number of copies: 3 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 37.731031 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MKEDQVVAEE PGFQDEEESL FQDIDLLQKH GINVADIKKL KSVGICTIKG IQMTTRRALC NVKGLSEAKV DKIKEAANKL IEPGFLTAF EYSEKRKMVF HITTGSQEFD KLLGGGIESM AITEAFGEFR TGKTQLSHTL CVTAQLPGAG GYPGGKIIFI D TENTFRPD ...String: MKEDQVVAEE PGFQDEEESL FQDIDLLQKH GINVADIKKL KSVGICTIKG IQMTTRRALC NVKGLSEAKV DKIKEAANKL IEPGFLTAF EYSEKRKMVF HITTGSQEFD KLLGGGIESM AITEAFGEFR TGKTQLSHTL CVTAQLPGAG GYPGGKIIFI D TENTFRPD RLRDIADRFN VDHDAVLDNV LYARAYTSEH QMELLDYVAA KFHEEAGIFK LLIIDSIMAL FRVDFSGRGE LA ERQQKLA QMLSRLQKIS EEYNVAVFVT NQMTADPGAT MTFQADPKKP IGGHILAHAS TTRISLRKGR GELRIAKIYD SPE MPENEA TFAITAGGIG DAKE UniProtKB: Meiotic recombination protein DMC1/LIM15 homolog |
-Macromolecule #2: DNA (5'-D(P*TP*TP*TP*TP*TP*TP*TP*TP*T)-3')
| Macromolecule | Name: DNA (5'-D(P*TP*TP*TP*TP*TP*TP*TP*TP*T)-3') / type: dna / ID: 2 / Number of copies: 1 / Classification: DNA |
|---|---|
| Source (natural) | Organism: synthetic construct (others) |
| Molecular weight | Theoretical: 2.692778 KDa |
| Sequence | String: (DT)(DT)(DT)(DT)(DT)(DT)(DT)(DT)(DT) |
-Macromolecule #3: DNA (5'-D(P*AP*AP*AP*AP*AP*AP*AP*AP*A)-3')
| Macromolecule | Name: DNA (5'-D(P*AP*AP*AP*AP*AP*AP*AP*AP*A)-3') / type: dna / ID: 3 / Number of copies: 1 / Classification: DNA |
|---|---|
| Source (natural) | Organism: synthetic construct (others) |
| Molecular weight | Theoretical: 2.773904 KDa |
| Sequence | String: (DA)(DA)(DA)(DA)(DA)(DA)(DA)(DA)(DA) |
-Macromolecule #4: CALCIUM ION
| Macromolecule | Name: CALCIUM ION / type: ligand / ID: 4 / Number of copies: 3 / Formula: CA |
|---|---|
| Molecular weight | Theoretical: 40.078 Da |
-Macromolecule #5: PHOSPHOAMINOPHOSPHONIC ACID-ADENYLATE ESTER
| Macromolecule | Name: PHOSPHOAMINOPHOSPHONIC ACID-ADENYLATE ESTER / type: ligand / ID: 5 / Number of copies: 3 / Formula: ANP |
|---|---|
| Molecular weight | Theoretical: 506.196 Da |
| Chemical component information |  ChemComp-ANP: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | helical reconstruction |
| Aggregation state | filament |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 Details: 25 mM Tris-HCl, pH 7.5, 50 mM KCl and 1 mM dithiothreitol) containing 2 mM AMP-PNP and 5 mM CaCl2 |
|---|---|
| Grid | Model: Quantifoil R1.2/1.3 / Material: GRAPHENE OXIDE / Mesh: 200 / Support film - Material: GRAPHENE OXIDE / Support film - topology: CONTINUOUS |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 295 K / Instrument: FEI VITROBOT MARK IV Details: The grids were blotted for 1 sec at 22 degree C with 100% relative humidity and plunge-frozen in liquid ethane cooled by liquid nitrogen using a Vitrobot Mark IV (Thermo Fisher).. |
| Details | protein sample were applied onto a pre-glow-discharged graphene-oxide coated Quantifoil holey carbon grid (1.2/1.3, 200 mesh) using published protocol |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Alignment procedure | Coma free - Residual tilt: 10.0 mrad |
| Specialist optics | Energy filter - Name: GIF Quantum LS / Energy filter - Slit width: 30 eV |
| Image recording | Film or detector model: GATAN K2 QUANTUM (4k x 4k) / Detector mode: COUNTING / Digitization - Dimensions - Width: 3838 pixel / Digitization - Dimensions - Height: 3710 pixel / Digitization - Frames/image: 1-50 / Number grids imaged: 1 / Average exposure time: 5.0 sec. / Average electron dose: 50.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 70.0 µm / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal magnification: 165000 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller




















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)