+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Focused map of p97:UBXD1 open state (P1 protomer) | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | AAA+ / chaperone / hydrolase | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.51 Å | |||||||||
 Authors Authors | Braxton JR / Tucker MR / Tse E / Southworth DR | |||||||||
| Funding support |  United States, 1 items United States, 1 items
| |||||||||
 Citation Citation |  Journal: bioRxiv / Year: 2023 Journal: bioRxiv / Year: 2023Title: The p97/VCP adapter UBXD1 drives AAA+ remodeling and ring opening through multi-domain tethered interactions. Authors: Julian R Braxton / Chad R Altobelli / Maxwell R Tucker / Eric Tse / Aye C Thwin / Michelle R Arkin / Daniel R Southworth /  Abstract: p97/VCP is an essential cytosolic AAA+ ATPase hexamer that extracts and unfolds substrate polypeptides during protein homeostasis and degradation. Distinct sets of p97 adapters guide cellular ...p97/VCP is an essential cytosolic AAA+ ATPase hexamer that extracts and unfolds substrate polypeptides during protein homeostasis and degradation. Distinct sets of p97 adapters guide cellular functions but their roles in direct control of the hexamer are unclear. The UBXD1 adapter localizes with p97 in critical mitochondria and lysosome clearance pathways and contains multiple p97-interacting domains. We identify UBXD1 as a potent p97 ATPase inhibitor and report structures of intact p97:UBXD1 complexes that reveal extensive UBXD1 contacts across p97 and an asymmetric remodeling of the hexamer. Conserved VIM, UBX, and PUB domains tether adjacent protomers while a connecting strand forms an N-terminal domain lariat with a helix wedged at the interprotomer interface. An additional VIM-connecting helix binds along the second AAA+ domain. Together these contacts split the hexamer into a ring-open conformation. Structures, mutagenesis, and comparisons to other adapters further reveal how adapters containing conserved p97-remodeling motifs regulate p97 ATPase activity and structure. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_28985.map.gz emd_28985.map.gz | 230 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-28985-v30.xml emd-28985-v30.xml emd-28985.xml emd-28985.xml | 21.9 KB 21.9 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_28985.png emd_28985.png | 170 KB | ||
| Others |  emd_28985_additional_1.map.gz emd_28985_additional_1.map.gz emd_28985_half_map_1.map.gz emd_28985_half_map_1.map.gz emd_28985_half_map_2.map.gz emd_28985_half_map_2.map.gz | 120 MB 226.3 MB 226.3 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-28985 http://ftp.pdbj.org/pub/emdb/structures/EMD-28985 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-28985 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-28985 | HTTPS FTP |
-Validation report
| Summary document |  emd_28985_validation.pdf.gz emd_28985_validation.pdf.gz | 908.2 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_28985_full_validation.pdf.gz emd_28985_full_validation.pdf.gz | 907.7 KB | Display | |
| Data in XML |  emd_28985_validation.xml.gz emd_28985_validation.xml.gz | 15.5 KB | Display | |
| Data in CIF |  emd_28985_validation.cif.gz emd_28985_validation.cif.gz | 18 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-28985 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-28985 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-28985 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-28985 | HTTPS FTP |
-Related structure data
| Related structure data | C: citing same article ( |
|---|
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_28985.map.gz / Format: CCP4 / Size: 244.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_28985.map.gz / Format: CCP4 / Size: 244.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Voxel size | X=Y=Z: 0.834 Å | ||||||||||||||||||||
| Density |
| ||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Additional map: Unsharpened map
| File | emd_28985_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Unsharpened map | ||||||||||||
| Projections & Slices |
| ||||||||||||
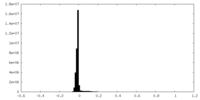
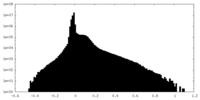
| Density Histograms |
-Half map: #1
| File | emd_28985_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
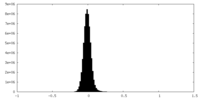
| Density Histograms |
-Half map: #2
| File | emd_28985_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
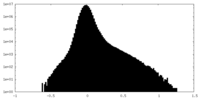
| Density Histograms |
- Sample components
Sample components
-Entire : Complex of p97 and UBXD1
| Entire | Name: Complex of p97 and UBXD1 |
|---|---|
| Components |
|
-Supramolecule #1: Complex of p97 and UBXD1
| Supramolecule | Name: Complex of p97 and UBXD1 / type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: Transitional endoplasmic reticulum ATPase
| Macromolecule | Name: Transitional endoplasmic reticulum ATPase / type: protein_or_peptide / ID: 1 / Enantiomer: LEVO / EC number: vesicle-fusing ATPase |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Recombinant expression | Organism:  |
| Sequence | String: MASGADSKGD DLSTAILKQK NRPNRLIVDE AINEDNSVVS LSQPKMDELQ LFRGDTVLLK GKKRREAVCI VLSDDTCSDE KIRMNRVVRN NLRVRLGDVI SIQPCPDVKY GKRIHVLPID DTVEGITGNL FEVYLKPYFL EAYRPIRKGD IFLVRGGMRA VEFKVVETDP ...String: MASGADSKGD DLSTAILKQK NRPNRLIVDE AINEDNSVVS LSQPKMDELQ LFRGDTVLLK GKKRREAVCI VLSDDTCSDE KIRMNRVVRN NLRVRLGDVI SIQPCPDVKY GKRIHVLPID DTVEGITGNL FEVYLKPYFL EAYRPIRKGD IFLVRGGMRA VEFKVVETDP SPYCIVAPDT VIHCEGEPIK REDEEESLNE VGYDDIGGCR KQLAQIKEMV ELPLRHPALF KAIGVKPPRG ILLYGPPGTG KTLIARAVAN ETGAFFFLIN GPEIMSKLAG ESESNLRKAF EEAEKNAPAI IFIDELDAIA PKREKTHGEV ERRIVSQLLT LMDGLKQRAH VIVMAATNRP NSIDPALRRF GRFDREVDIG IPDATGRLEI LQIHTKNMKL ADDVDLEQVA NETHGHVGAD LAALCSEAAL QAIRKKMDLI DLEDETIDAE VMNSLAVTMD DFRWALSQSN PSALRETVVE VPQVTWEDIG GLEDVKRELQ ELVQYPVEHP DKFLKFGMTP SKGVLFYGPP GCGKTLLAKA IANECQANFI SIKGPELLTM WFGESEANVR EIFDKARQAA PCVLFFDELD SIAKARGGNI GDGGGAADRV INQILTEMDG MSTKKNVFII GATNRPDIID PAILRPGRLD QLIYIPLPDE KSRVAILKAN LRKSPVAKDV DLEFLAKMTN GFSGADLTEI CQRACKLAIR ESIESEIRRE RERQTNPSAM EVEEDDPVPE IRRDHFEEAM RFARRSVSDN DIRKYEMFAQ TLQQSRGFGS FRFPSGNQGG AGPSQGSGGG TGGSVYTEDN DDDLYG |
-Macromolecule #2: UBX domain-containing protein 6
| Macromolecule | Name: UBX domain-containing protein 6 / type: protein_or_peptide / ID: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Recombinant expression | Organism:  |
| Sequence | String: MKKFFQEFKA DIKFKSAGPG QKLKESVGEK AHKEKPNQPA PRPPRQGPTN EAQMAAAAAL ARLEQKQSRA WGPTSQDTIR NQVRKELQAE ATVSGSPEAP GTNVVSEPRE EGSAHLAVPG VYFTCPLTGA TLRKDQRDAC IKEAILLHFS TDPVAASIMK IYTFNKDQDR ...String: MKKFFQEFKA DIKFKSAGPG QKLKESVGEK AHKEKPNQPA PRPPRQGPTN EAQMAAAAAL ARLEQKQSRA WGPTSQDTIR NQVRKELQAE ATVSGSPEAP GTNVVSEPRE EGSAHLAVPG VYFTCPLTGA TLRKDQRDAC IKEAILLHFS TDPVAASIMK IYTFNKDQDR VKLGVDTIAK YLDNIHLHPE EEKYRKIKLQ NKVFQERINC LEGTHEFFEA IGFQKVLLPA QDQEDPEEFY VLSETTLAQP QSLERHKEQL LAAEPVRAKL DRQRRVFQPS PLASQFELPG DFFNLTAEEI KREQRLRSEA VERLSVLRTK AMREKEEQRG LRKYNYTLLR VRLPDGCLLQ GTFYARERLG AVYGFVREAL QSDWLPFELL ASGGQKLSED ENLALNECGL VPSALLTFSW DMAVLEDIKA AGAEPDSILK PELLSAIEKL L |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 1.89 mg/mL |
|---|---|
| Buffer | pH: 7.4 |
| Grid | Model: Quantifoil R1.2/1.3 / Material: GOLD / Mesh: 200 / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 120 sec. / Pretreatment - Atmosphere: AIR / Pretreatment - Pressure: 0.03 kPa / Details: Pelco easiGlow, 15 mA |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 277 K / Instrument: FEI VITROBOT MARK IV / Details: Wait time 10 sec, blot time 3 sec, blot force 0. |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Specialist optics | Energy filter - Name: GIF Bioquantum / Energy filter - Slit width: 20 eV |
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Number grids imaged: 1 / Number real images: 22536 / Average exposure time: 2.024 sec. / Average electron dose: 43.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 70.0 µm / Calibrated magnification: 59952 / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 1.8 µm / Nominal defocus min: 0.8 µm / Nominal magnification: 105000 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: OTHER / Details: Ab initio reconstruction from cryoSPARC |
|---|---|
| Final reconstruction | Applied symmetry - Point group: C1 (asymmetric) / Resolution.type: BY AUTHOR / Resolution: 3.51 Å / Resolution method: FSC 0.143 CUT-OFF / Software - Name: cryoSPARC / Number images used: 208495 |
| Initial angle assignment | Type: RANDOM ASSIGNMENT / Software - Name: cryoSPARC |
| Final angle assignment | Type: MAXIMUM LIKELIHOOD / Software - Name: cryoSPARC |
-Atomic model buiding 1
| Refinement | Protocol: FLEXIBLE FIT |
|---|
 Movie
Movie Controller
Controller






















 Z
Z Y
Y X
X