+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM Structure of P74-26 tail-like tubes | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | tail tube protein / bacteriophage / VIRAL PROTEIN | |||||||||
| Function / homology | Phage tail tube protein-like / Phage tail tube protein / Major capsid protein Function and homology information Function and homology information | |||||||||
| Biological species |  Oshimavirus P7426 Oshimavirus P7426 | |||||||||
| Method | helical reconstruction / cryo EM / Resolution: 2.81 Å | |||||||||
 Authors Authors | Agnello E / Pajak J / Liu X / Kelch B | |||||||||
| Funding support |  United States, 2 items United States, 2 items
| |||||||||
 Citation Citation |  Journal: J Biol Chem / Year: 2023 Journal: J Biol Chem / Year: 2023Title: Conformational dynamics control assembly of an extremely long bacteriophage tail tube. Authors: Emily Agnello / Joshua Pajak / Xingchen Liu / Brian A Kelch /  Abstract: Tail tube assembly is an essential step in the lifecycle of long-tailed bacteriophages. Limited structural and biophysical information has impeded an understanding of assembly and stability of their ...Tail tube assembly is an essential step in the lifecycle of long-tailed bacteriophages. Limited structural and biophysical information has impeded an understanding of assembly and stability of their long, flexible tail tubes. The hyperthermophilic phage P74-26 is particularly intriguing as it has the longest tail of any known virus (nearly 1 μm) and is the most thermostable known phage. Here, we use structures of the P74-26 tail tube along with an in vitro system for studying tube assembly kinetics to propose the first molecular model for the tail tube assembly of long-tailed phages. Our high-resolution cryo-EM structure provides insight into how the P74-26 phage assembles through flexible loops that fit into neighboring rings through tight "ball-and-socket"-like interactions. Guided by this structure, and in combination with mutational, light scattering, and molecular dynamics simulations data, we propose a model for the assembly of conserved tube-like structures across phage and other entities possessing tail tube-like proteins. We propose that formation of a full ring promotes the adoption of a tube elongation-competent conformation among the flexible loops and their corresponding sockets, which is further stabilized by an adjacent ring. Tail assembly is controlled by the cooperative interaction of dynamic intraring and interring contacts. Given the structural conservation among tail tube proteins and tail-like structures, our model can explain the mechanism of high-fidelity assembly of long, stable tubes. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_28042.map.gz emd_28042.map.gz | 53.4 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-28042-v30.xml emd-28042-v30.xml emd-28042.xml emd-28042.xml | 16.3 KB 16.3 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_28042.png emd_28042.png | 125.4 KB | ||
| Filedesc metadata |  emd-28042.cif.gz emd-28042.cif.gz | 5.7 KB | ||
| Others |  emd_28042_half_map_1.map.gz emd_28042_half_map_1.map.gz emd_28042_half_map_2.map.gz emd_28042_half_map_2.map.gz | 95.1 MB 95.1 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-28042 http://ftp.pdbj.org/pub/emdb/structures/EMD-28042 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-28042 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-28042 | HTTPS FTP |
-Validation report
| Summary document |  emd_28042_validation.pdf.gz emd_28042_validation.pdf.gz | 931.8 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_28042_full_validation.pdf.gz emd_28042_full_validation.pdf.gz | 931.3 KB | Display | |
| Data in XML |  emd_28042_validation.xml.gz emd_28042_validation.xml.gz | 13.7 KB | Display | |
| Data in CIF |  emd_28042_validation.cif.gz emd_28042_validation.cif.gz | 16 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-28042 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-28042 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-28042 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-28042 | HTTPS FTP |
-Related structure data
| Related structure data |  8edxMC  8ed0C M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_28042.map.gz / Format: CCP4 / Size: 103 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_28042.map.gz / Format: CCP4 / Size: 103 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.87 Å | ||||||||||||||||||||||||||||||||||||
| Density |
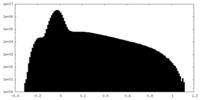
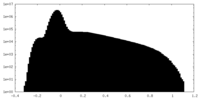
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #2
| File | emd_28042_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
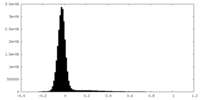
| Density Histograms |
-Half map: #1
| File | emd_28042_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
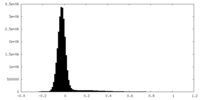
| Density Histograms |
- Sample components
Sample components
-Entire : In vitro-assembled tail-like tubes of tail tube protein gp93
| Entire | Name: In vitro-assembled tail-like tubes of tail tube protein gp93 |
|---|---|
| Components |
|
-Supramolecule #1: In vitro-assembled tail-like tubes of tail tube protein gp93
| Supramolecule | Name: In vitro-assembled tail-like tubes of tail tube protein gp93 type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  Oshimavirus P7426 Oshimavirus P7426 |
-Macromolecule #1: Tail Tube Protein gp93
| Macromolecule | Name: Tail Tube Protein gp93 / type: protein_or_peptide / ID: 1 / Number of copies: 6 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Oshimavirus P7426 Oshimavirus P7426 |
| Molecular weight | Theoretical: 37.955738 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MRGVDTFLAF KEQADLKTPA TLASLAAGDF LAFNSESLSG RQQVIQSRAI RRMPMRQIAY TANGTVEAGG AVEFTTSNYV LKKLLPLIF HSKTGQEDDP DGDGATFTLV NGGVLTPFTA FVGFDGPEGK YVRRFFGAKV NQATFSARVN DMLNLNLDVQ A IGKDILQP ...String: MRGVDTFLAF KEQADLKTPA TLASLAAGDF LAFNSESLSG RQQVIQSRAI RRMPMRQIAY TANGTVEAGG AVEFTTSNYV LKKLLPLIF HSKTGQEDDP DGDGATFTLV NGGVLTPFTA FVGFDGPEGK YVRRFFGAKV NQATFSARVN DMLNLNLDVQ A IGKDILQP GDPGWVNVTP VYPGGDEEYA YVFYQARVLI KAGDMADLAE LPVESFDLTI NHNLNTNRYR LGSIYRQSLD EG VTEVTGT FTLDAAVKSI SGPALNLTGG TAHDPAFLEK VALYGKYAAL KLEFIDPTRE VAEGVPCRLT IHLPFVRLEE PDF QVRDPG VITGSARFNA YETISVTHVA KF UniProtKB: Major capsid protein |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | helical reconstruction |
| Aggregation state | filament |
- Sample preparation
Sample preparation
| Concentration | 1.5 mg/mL | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 8 Component:
| |||||||||||||||
| Grid | Model: EMS Lacey Carbon / Material: COPPER / Mesh: 400 / Support film - Material: CARBON / Support film - topology: LACEY / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 60 sec. Details: Grids were glow discharged at 25 mA for 60 sec with negative polarity. | |||||||||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 90 % / Chamber temperature: 283.15 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TALOS ARCTICA |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: SUPER-RESOLUTION / Average electron dose: 38.4312 e/Å2 |
| Electron beam | Acceleration voltage: 200 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 1.5 µm / Nominal defocus min: 0.5 µm / Nominal magnification: 45000 |
| Sample stage | Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Talos Arctica / Image courtesy: FEI Company |
- Image processing
Image processing
| Final reconstruction | Number classes used: 1 Applied symmetry - Helical parameters - Δz: 40.0 Å Applied symmetry - Helical parameters - Δ&Phi: -44 ° Applied symmetry - Helical parameters - Axial symmetry: C3 (3 fold cyclic) Resolution.type: BY AUTHOR / Resolution: 2.81 Å / Resolution method: FSC 0.143 CUT-OFF / Software - Name: cryoSPARC / Number images used: 395357 |
|---|---|
| Segment selection | Number selected: 619907 |
| Startup model | Type of model: NONE / Details: Ab initio map generated in cryoSPARC |
| Final angle assignment | Type: NOT APPLICABLE |
 Movie
Movie Controller
Controller





 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)