[English] 日本語
 Yorodumi
Yorodumi- EMDB-27225: Cryo-EM 3D map of the Mycobacterium tuberculosis 20S proteasome b... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
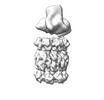
| Title | Cryo-EM 3D map of the Mycobacterium tuberculosis 20S proteasome bound to the ATP-bound Mpa ATPase | |||||||||
 Map data Map data | Cryo-EM 3D map of the Mycobacterium tuberculosis 20S proteasome bound to the ATP-bound Mpa ATPase | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Mpa / proteasome / ANTIMICROBIAL PROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationproteasome-activating nucleotidase complex / proteasome endopeptidase complex / proteasome core complex, beta-subunit complex / threonine-type endopeptidase activity / proteasome core complex, alpha-subunit complex / proteasomal protein catabolic process / modification-dependent protein catabolic process / ATP hydrolysis activity / ATP binding / cytoplasm Similarity search - Function | |||||||||
| Biological species |  | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.2 Å | |||||||||
 Authors Authors | Xiao X / Li H | |||||||||
| Funding support |  United States, 1 items United States, 1 items
| |||||||||
 Citation Citation |  Journal: mSphere / Year: 2022 Journal: mSphere / Year: 2022Title: The β-Grasp Domain of Proteasomal ATPase Mpa Makes Critical Contacts with the Mycobacterium tuberculosis 20S Core Particle to Facilitate Degradation. Authors: Xiansha Xiao / Xiang Feng / Jin Hee Yoo / Amanda Kovach / K Heran Darwin / Huilin Li /  Abstract: Mycobacterium tuberculosis possesses a Pup-proteasome system analogous to the eukaryotic ubiquitin-proteasome pathway. We have previously shown that the hexameric mycobacterial proteasome ATPase (Mpa) ...Mycobacterium tuberculosis possesses a Pup-proteasome system analogous to the eukaryotic ubiquitin-proteasome pathway. We have previously shown that the hexameric mycobacterial proteasome ATPase (Mpa) recruits pupylated protein substrates via interactions between amino-terminal coiled-coils in Mpa monomers and the degradation tag Pup. However, it is unclear how Mpa rings interact with a proteasome due to the presence of a carboxyl-terminal β-grasp domain unique to Mpa homologues that makes the interaction highly unstable. Here, we describe newly identified critical interactions between Mpa and 20S core proteasomes. Interestingly, the Mpa C-terminal GQYL motif binds the 20S core particle activation pocket differently than the same motif of the ATP-independent proteasome accessory factor PafE. We further found that the β-hairpin of the Mpa β-grasp domain interacts variably with the H0 helix on top of the 20S core particle via a series of ionic and hydrogen-bond interactions. Individually mutating several involved residues reduced Mpa-mediated protein degradation both and . The Pup-proteasome system in Mycobacterium tuberculosis is critical for this species to cause lethal infections in mice. Investigating the molecular mechanism of how the Mpa ATPase recruits and unfolds pupylated substrates to the 20S proteasomal core particle for degradation will be essential to fully understand how degradation is regulated, and the structural information we report may be useful for the development of new tuberculosis chemotherapies. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_27225.map.gz emd_27225.map.gz | 1.6 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-27225-v30.xml emd-27225-v30.xml emd-27225.xml emd-27225.xml | 17.3 KB 17.3 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_27225.png emd_27225.png | 45.2 KB | ||
| Filedesc metadata |  emd-27225.cif.gz emd-27225.cif.gz | 6.7 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-27225 http://ftp.pdbj.org/pub/emdb/structures/EMD-27225 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-27225 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-27225 | HTTPS FTP |
-Related structure data
| Related structure data |  8d6xMC  8d6vC  8d6wC  8d6yC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_27225.map.gz / Format: CCP4 / Size: 3.4 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_27225.map.gz / Format: CCP4 / Size: 3.4 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Cryo-EM 3D map of the Mycobacterium tuberculosis 20S proteasome bound to the ATP-bound Mpa ATPase | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 4.592 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
- Sample components
Sample components
-Entire : binary complex of 20S proteasome with ATPase Mpa
| Entire | Name: binary complex of 20S proteasome with ATPase Mpa |
|---|---|
| Components |
|
-Supramolecule #1: binary complex of 20S proteasome with ATPase Mpa
| Supramolecule | Name: binary complex of 20S proteasome with ATPase Mpa / type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  |
-Macromolecule #1: AAA ATPase forming ring-shaped complexes
| Macromolecule | Name: AAA ATPase forming ring-shaped complexes / type: protein_or_peptide / ID: 1 / Number of copies: 6 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 67.48793 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MGESERSEAF GIPRDSPLSS GDAAELEQLR REAAVLREQL ENAVGSHAPT RSARDIHQLE ARIDSLAARN SKLMETLKEA RQQLLALRE EVDRLGQPPS GYGVLLATHD DDTVDVFTSG RKMRLTCSPN IDAASLKKGQ TVRLNEALTV VEAGTFEAVG E ISTLREIL ...String: MGESERSEAF GIPRDSPLSS GDAAELEQLR REAAVLREQL ENAVGSHAPT RSARDIHQLE ARIDSLAARN SKLMETLKEA RQQLLALRE EVDRLGQPPS GYGVLLATHD DDTVDVFTSG RKMRLTCSPN IDAASLKKGQ TVRLNEALTV VEAGTFEAVG E ISTLREIL ADGHRALVVG HADEERVVWL ADPLIAEDLP DGLPEALNDD TRPRKLRPGD SLLVDTKAGY AFERIPKAEV ED LVLEEVP DVSYADIGGL SRQIEQIRDA VELPFLHKEL YREYSLRPPK GVLLYGPPGC GKTLIAKAVA NSLAKKMAEV RGD DAHEAK SYFLNIKGPE LLNKFVGETE RHIRLIFQRA REKASEGTPV IVFFDEMDSI FRTRGTGVSS DVETTVVPQL LSEI DGVEG LENVIVIGAS NREDMIDPAI LRPGRLDVKI KIERPDAEAA QDIYSKYLTE FLPVHADDLA EFDGDRSACI KAMIE KVVD RMYAEIDDNR FLEVTYANGD KEVMYFKDFN SGAMIQNVVD RAKKNAIKSV LETGQPGLRI QHLLDSIVDE FAENED LPN TTNPDDWARI SGKKGERIVY IRTLVTGKSS SASRAIDTES NLGQYL UniProtKB: AAA ATPase forming ring-shaped complexes |
-Macromolecule #2: Proteasome subunit alpha
| Macromolecule | Name: Proteasome subunit alpha / type: protein_or_peptide / ID: 2 / Number of copies: 14 / Enantiomer: LEVO / EC number: proteasome endopeptidase complex |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 26.911039 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MSFPYFISPE QAMRERSELA RKGIARAKSV VALAYAGGVL FVAENPSRSL QKISELYDRV GFAAAGKFNE FDNLRRGGIQ FADTRGYAY DRRDVTGRQL ANVYAQTLGT IFTEQAKPYE VELCVAEVAH YGETKRPELY RITYDGSIAD EPHFVVMGGT T EPIANALK ...String: MSFPYFISPE QAMRERSELA RKGIARAKSV VALAYAGGVL FVAENPSRSL QKISELYDRV GFAAAGKFNE FDNLRRGGIQ FADTRGYAY DRRDVTGRQL ANVYAQTLGT IFTEQAKPYE VELCVAEVAH YGETKRPELY RITYDGSIAD EPHFVVMGGT T EPIANALK ESYAENASLT DALRIAVAAL RAGSADTSGG DQPTLGVASL EVAVLDANRP RRAFRRITGS ALQALLVDQE SP QSDGESS G UniProtKB: Proteasome subunit alpha |
-Macromolecule #3: Proteasome subunit beta
| Macromolecule | Name: Proteasome subunit beta / type: protein_or_peptide / ID: 3 / Number of copies: 14 / Enantiomer: LEVO / EC number: proteasome endopeptidase complex |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 30.332006 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MTWPLPDRLS INSLSGTPAV DLSSFTDFLR RQAPELLPAS ISGGAPLAGG DAQLPHGTTI VALKYPGGVV MAGDRRSTQG NMISGRDVR KVYITDDYTA TGIAGTAAVA VEFARLYAVE LEHYEKLEGV PLTFAGKINR LAIMVRGNLA AAMQGLLALP L LAGYDIHA ...String: MTWPLPDRLS INSLSGTPAV DLSSFTDFLR RQAPELLPAS ISGGAPLAGG DAQLPHGTTI VALKYPGGVV MAGDRRSTQG NMISGRDVR KVYITDDYTA TGIAGTAAVA VEFARLYAVE LEHYEKLEGV PLTFAGKINR LAIMVRGNLA AAMQGLLALP L LAGYDIHA SDPQSAGRIV SFDAAGGWNI EEEGYQAVGS GSLFAKSSMK KLYSQVTDGD SGLRVAVEAL YDAADDDSAT GG PDLVRGI FPTAVIIDAD GAVDVPESRI AELARAIIES RSGADTFGSD GGEK UniProtKB: Proteasome subunit beta |
-Macromolecule #4: Proteasome-associated ATPase
| Macromolecule | Name: Proteasome-associated ATPase / type: protein_or_peptide / ID: 4 / Number of copies: 7 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 479.527 Da |
| Recombinant expression | Organism:  |
| Sequence | String: GQYL UniProtKB: Proteasome-associated ATPase |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 8 |
|---|---|
| Grid | Model: Quantifoil R1.2/1.3 / Material: GOLD / Mesh: 300 / Support film - Material: CARBON |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 283.15 K / Instrument: FEI VITROBOT MARK II |
- Electron microscopy
Electron microscopy
| Microscope | FEI TALOS ARCTICA |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: SUPER-RESOLUTION / Average exposure time: 6.0 sec. / Average electron dose: 1.2 e/Å2 |
| Electron beam | Acceleration voltage: 200 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 5.0 µm / Nominal defocus min: 1.2 µm |
| Sample stage | Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Talos Arctica / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller










 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)





















