+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
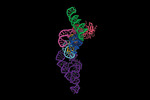
| Title | E.coli RNaseP Holoenzyme with Mg2+ | |||||||||
 Map data Map data | full map | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | ribozyme / protein-RNA complex / divalent ion / RNA / HYDROLASE-RNA complex | |||||||||
| Function / homology |  Function and homology information Function and homology information3'-tRNA processing endoribonuclease activity / ribonuclease P complex / ribonuclease P / ribonuclease P activity / tRNA 5'-leader removal / tRNA binding Similarity search - Function | |||||||||
| Biological species |  | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.1 Å | |||||||||
 Authors Authors | Huang W / Taylor DJ | |||||||||
| Funding support |  United States, 1 items United States, 1 items
| |||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2022 Journal: Nat Commun / Year: 2022Title: Structural and mechanistic basis for recognition of alternative tRNA precursor substrates by bacterial ribonuclease P. Authors: Jiaqiang Zhu / Wei Huang / Jing Zhao / Loc Huynh / Derek J Taylor / Michael E Harris /  Abstract: Binding of precursor tRNAs (ptRNAs) by bacterial ribonuclease P (RNase P) involves an encounter complex (ES) that isomerizes to a catalytic conformation (ES*). However, the structures of ...Binding of precursor tRNAs (ptRNAs) by bacterial ribonuclease P (RNase P) involves an encounter complex (ES) that isomerizes to a catalytic conformation (ES*). However, the structures of intermediates and the conformational changes that occur during binding are poorly understood. Here, we show that pairing between the 5' leader and 3'RCCA extending the acceptor stem of ptRNA inhibits ES* formation. Cryo-electron microscopy single particle analysis reveals a dynamic enzyme that becomes ordered upon formation of ES* in which extended acceptor stem pairing is unwound. Comparisons of structures with alternative ptRNAs reveals that once unwinding is completed RNase P primarily uses stacking interactions and shape complementarity to accommodate alternative sequences at its cleavage site. Our study reveals active site interactions and conformational changes that drive molecular recognition by RNase P and lays the foundation for understanding how binding interactions are linked to helix unwinding and catalysis. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_26640.map.gz emd_26640.map.gz | 96.2 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-26640-v30.xml emd-26640-v30.xml emd-26640.xml emd-26640.xml | 17.1 KB 17.1 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_26640.png emd_26640.png | 29.3 KB | ||
| Filedesc metadata |  emd-26640.cif.gz emd-26640.cif.gz | 5.8 KB | ||
| Others |  emd_26640_half_map_1.map.gz emd_26640_half_map_1.map.gz emd_26640_half_map_2.map.gz emd_26640_half_map_2.map.gz | 176.2 MB 176.2 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-26640 http://ftp.pdbj.org/pub/emdb/structures/EMD-26640 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-26640 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-26640 | HTTPS FTP |
-Related structure data
| Related structure data |  7uo5MC  7uo0C  7uo1C  7uo2C M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_26640.map.gz / Format: CCP4 / Size: 190.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_26640.map.gz / Format: CCP4 / Size: 190.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | full map | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.2132 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #1
| File | emd_26640_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
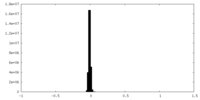
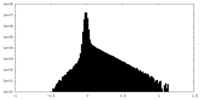
| Density Histograms |
-Half map: #2
| File | emd_26640_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
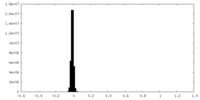
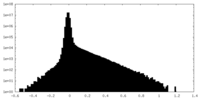
| Density Histograms |
- Sample components
Sample components
-Entire : E.coli RNase P holoenzyme with Mg2+
| Entire | Name: E.coli RNase P holoenzyme with Mg2+ |
|---|---|
| Components |
|
-Supramolecule #1: E.coli RNase P holoenzyme with Mg2+
| Supramolecule | Name: E.coli RNase P holoenzyme with Mg2+ / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#2 |
|---|---|
| Source (natural) | Organism:  |
-Macromolecule #1: Ribonuclease P protein component
| Macromolecule | Name: Ribonuclease P protein component / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO / EC number: ribonuclease P |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 13.10237 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: KLAFPRELRL LTPSQFTFVF QQPQRAGTPQ ITILGRLNSL GHPRIGLTVA KKNVRRAHER NRIKRLTRES FRLRQHELPA MDFVVVAKK GVADLDNRAL SEALEKLWRR HCR UniProtKB: Ribonuclease P protein component |
-Macromolecule #2: RNase P RNA
| Macromolecule | Name: RNase P RNA / type: rna / ID: 2 / Number of copies: 1 |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 121.191891 KDa |
| Sequence | String: GAAGCUGACC AGACAGUCGC CGCUUCGUCG UCGUCCUCUU CGGGGGAGAC GGGCGGAGGG GAGGAAAGUC CGGGCUCCAU AGGGCAGGG UGCCAGGUAA CGCCUGGGGG GGAAACCCAC GACCAGUGCA ACAGAGAGCA AACCGCCGAU GGCCCGCGCA A GCGGGAUC ...String: GAAGCUGACC AGACAGUCGC CGCUUCGUCG UCGUCCUCUU CGGGGGAGAC GGGCGGAGGG GAGGAAAGUC CGGGCUCCAU AGGGCAGGG UGCCAGGUAA CGCCUGGGGG GGAAACCCAC GACCAGUGCA ACAGAGAGCA AACCGCCGAU GGCCCGCGCA A GCGGGAUC AGGUAAGGGU GAAAGGGUGC GGUAAGAGCG CACCGCGCGG CUGGUAACAG UCCGUGGCAC GGUAAACUCC AC CCGGAGC AAGGCCAAAU AGGGGUUCAU AAGGUACGGC CCGUACUGAA CCCGGGUAGG CUGCUUGAGC CAGUGAGCGA UUG CUGGCC UAGAUGAAUG ACUGUCCACG ACAGAACCCG GCUUAUCGGU CAGUUUC GENBANK: GENBANK: CP053595.1 |
-Macromolecule #3: CALCIUM ION
| Macromolecule | Name: CALCIUM ION / type: ligand / ID: 3 / Number of copies: 7 / Formula: CA |
|---|---|
| Molecular weight | Theoretical: 40.078 Da |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.1 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Average electron dose: 40.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 3.0 µm / Nominal defocus min: 1.0 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller







 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)