[English] 日本語
 Yorodumi
Yorodumi- EMDB-25834: CryoEM Structure of sFab COP-2 Complex with human claudin-4 and C... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-25834 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | CryoEM Structure of sFab COP-2 Complex with human claudin-4 and Clostridium perfringens enterotoxin C-terminal domain | |||||||||
 Map data Map data | main map | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Claudin / cell adhesion / enterotoxin / Fab / antibody fragment / MEMBRANE PROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationparacellular transport / calcium-independent cell-cell adhesion / Tight junction interactions / apicolateral plasma membrane / bicellular tight junction assembly / regulation of cell morphogenesis / tight junction / positive regulation of wound healing / renal absorption / chloride channel activity ...paracellular transport / calcium-independent cell-cell adhesion / Tight junction interactions / apicolateral plasma membrane / bicellular tight junction assembly / regulation of cell morphogenesis / tight junction / positive regulation of wound healing / renal absorption / chloride channel activity / establishment of skin barrier / lateral plasma membrane / bicellular tight junction / chloride channel complex / response to progesterone / basal plasma membrane / female pregnancy / circadian rhythm / cell-cell junction / transmembrane signaling receptor activity / toxin activity / cell adhesion / apical plasma membrane / positive regulation of cell migration / structural molecule activity / extracellular region / identical protein binding / plasma membrane Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) / Homo sapiens (human) /  | |||||||||
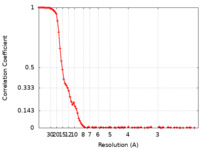
| Method | single particle reconstruction / cryo EM / Resolution: 6.9 Å | |||||||||
 Authors Authors | Vecchio AJ / Orlando BJ | |||||||||
| Funding support |  United States, 2 items United States, 2 items
| |||||||||
 Citation Citation |  Journal: J Biol Chem / Year: 2022 Journal: J Biol Chem / Year: 2022Title: Development, structure, and mechanism of synthetic antibodies that target claudin and Clostridium perfringens enterotoxin complexes. Authors: Benjamin J Orlando / Pawel K Dominik / Sourav Roy / Chinemerem P Ogbu / Satchal K Erramilli / Anthony A Kossiakoff / Alex J Vecchio /  Abstract: Strains of Clostridium perfringens produce a two-domain enterotoxin (CpE) that afflicts humans and domesticated animals, causing prevalent gastrointestinal illnesses. CpE's C-terminal domain (cCpE) ...Strains of Clostridium perfringens produce a two-domain enterotoxin (CpE) that afflicts humans and domesticated animals, causing prevalent gastrointestinal illnesses. CpE's C-terminal domain (cCpE) binds cell surface receptors, followed by a restructuring of its N-terminal domain to form a membrane-penetrating β-barrel pore, which is toxic to epithelial cells of the gut. The claudin family of membrane proteins are known receptors for CpE and also control the architecture and function of cell-cell contacts (tight junctions) that create barriers to intercellular molecular transport. CpE binding and assembly disables claudin barrier function and induces cytotoxicity via β-pore formation, disrupting gut homeostasis; however, a structural basis of this process and strategies to inhibit the claudin-CpE interactions that trigger it are both lacking. Here, we used a synthetic antigen-binding fragment (sFab) library to discover two sFabs that bind claudin-4 and cCpE complexes. We established these sFabs' mode of molecular recognition and binding properties and determined structures of each sFab bound to claudin-4-cCpE complexes using cryo-EM. The structures reveal that the sFabs bind a shared epitope, but conform distinctly, which explains their unique binding equilibria. Mutagenesis of antigen/sFab interfaces observed therein result in binding changes, validating the structures, and uncovering the sFab's targeting mechanism. From these insights, we generated a model for CpE's claudin-bound β-pore that predicted sFabs would not prevent cytotoxicity, which we then verified in vivo. Taken together, this work demonstrates the development and mechanism of claudin/cCpE-binding sFabs that provide a framework and strategy for obstructing claudin/CpE assembly to treat CpE-linked gastrointestinal diseases. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_25834.map.gz emd_25834.map.gz | 59.2 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-25834-v30.xml emd-25834-v30.xml emd-25834.xml emd-25834.xml | 19.8 KB 19.8 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_25834_fsc.xml emd_25834_fsc.xml | 11.9 KB | Display |  FSC data file FSC data file |
| Images |  emd_25834.png emd_25834.png | 63.3 KB | ||
| Filedesc metadata |  emd-25834.cif.gz emd-25834.cif.gz | 6.3 KB | ||
| Others |  emd_25834_half_map_1.map.gz emd_25834_half_map_1.map.gz emd_25834_half_map_2.map.gz emd_25834_half_map_2.map.gz | 59.4 MB 59.4 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-25834 http://ftp.pdbj.org/pub/emdb/structures/EMD-25834 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-25834 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-25834 | HTTPS FTP |
-Related structure data
| Related structure data |  7tdmMC  7tdnC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_25834.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_25834.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | main map | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.12 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
-Half map: half map A
| File | emd_25834_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | half map A | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: half map B
| File | emd_25834_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | half map B | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Human claudin-4/cCpE/COP-2 Complex
| Entire | Name: Human claudin-4/cCpE/COP-2 Complex |
|---|---|
| Components |
|
-Supramolecule #1: Human claudin-4/cCpE/COP-2 Complex
| Supramolecule | Name: Human claudin-4/cCpE/COP-2 Complex / type: complex / ID: 1 / Parent: 0 / Macromolecule list: all Details: COP-2 sFab fragment bound to Claudin-4/cCpE complex |
|---|---|
| Molecular weight | Theoretical: 85 kDa/nm |
-Macromolecule #1: Claudin-4
| Macromolecule | Name: Claudin-4 / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 22.090201 KDa |
| Recombinant expression | Organism:  Trichoplusia ni (cabbage looper) Trichoplusia ni (cabbage looper) |
| Sequence | String: MASMGLQVMG IALAVLGWLA VMLCCALPMW RVTAFIGSNI VTSQTIWEGL WMNCVVQSTG QMQCKVYDSL LALPQDLQAA RALVIISII VAALGVLLSV VGGKCTNCLE DESAKAKTMI VAGVVFLLAG LMVIVPVSWT AHNIIQDFYN PLVASGQKRE M GASLYVGW ...String: MASMGLQVMG IALAVLGWLA VMLCCALPMW RVTAFIGSNI VTSQTIWEGL WMNCVVQSTG QMQCKVYDSL LALPQDLQAA RALVIISII VAALGVLLSV VGGKCTNCLE DESAKAKTMI VAGVVFLLAG LMVIVPVSWT AHNIIQDFYN PLVASGQKRE M GASLYVGW AASGLLLLGG GLLCCNCPPR TDKPYSAKYS AARSAAASNY V UniProtKB: Claudin-4 |
-Macromolecule #2: Heat-labile enterotoxin B chain
| Macromolecule | Name: Heat-labile enterotoxin B chain / type: protein_or_peptide / ID: 2 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 15.114945 KDa |
| Recombinant expression | Organism:  Trichoplusia ni (cabbage looper) Trichoplusia ni (cabbage looper) |
| Sequence | String: MSTDIEKEIL DLAAATERLN LTDALNSNPA GNLYDWRSSN SYPWTQKLNL HLTITATGQK YRILASKIVD FNIYSNNFNN LVKLEQSLG DGVKDHYVDI SLDAGQYVLV MKANSSYSGN YPYSILFQKF GLVPR UniProtKB: Heat-labile enterotoxin B chain |
-Macromolecule #3: COP-2 Fab Heavy chain
| Macromolecule | Name: COP-2 Fab Heavy chain / type: protein_or_peptide / ID: 3 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 25.26301 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: EISEVQLVES GGGLVQPGGS LRLSCAASGF NFSSSSIHWV RQAPGKGLEW VASISSYSGY TSYADSVKGR FTISADTSKN TAYLQMNSL RAEDTAVYYC ARYWSWYNSS HYIYSALDYW GQGTLVTVSS ASTKGPSVFP LAPSSKSTSG GTAALGCLVK D YFPEPVTV ...String: EISEVQLVES GGGLVQPGGS LRLSCAASGF NFSSSSIHWV RQAPGKGLEW VASISSYSGY TSYADSVKGR FTISADTSKN TAYLQMNSL RAEDTAVYYC ARYWSWYNSS HYIYSALDYW GQGTLVTVSS ASTKGPSVFP LAPSSKSTSG GTAALGCLVK D YFPEPVTV SWNSGALTSG VHTFPAVLQS SGLYSLSSVV TVPSSSLGTQ TYICNVNHKP SNTKVDKKVE PKSCDKTHT |
-Macromolecule #4: COP-2 Fab Light chain
| Macromolecule | Name: COP-2 Fab Light chain / type: protein_or_peptide / ID: 4 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 23.517057 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: SDIQMTQSPS SLSASVGDRV TITCRASQSV SSAVAWYQQK PGKAPKLLIY SASSLYSGVP SRFSGSRSGT DFTLTISSLQ PEDFATYYC QQSYEWAPVT FGQGTKVEIK RTVAAPSVFI FPPSDSQLKS GTASVVCLLN NFYPREAKVQ WKVDNALQSG N SQESVTEQ ...String: SDIQMTQSPS SLSASVGDRV TITCRASQSV SSAVAWYQQK PGKAPKLLIY SASSLYSGVP SRFSGSRSGT DFTLTISSLQ PEDFATYYC QQSYEWAPVT FGQGTKVEIK RTVAAPSVFI FPPSDSQLKS GTASVVCLLN NFYPREAKVQ WKVDNALQSG N SQESVTEQ DSKDSTYSLS STLTLSKADY EKHKVYACEV THQGLSSPVT KSFNRGEC |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 8.0 mg/mL |
|---|---|
| Buffer | pH: 7.4 |
| Grid | Model: Quantifoil R1.2/1.3 / Mesh: 200 / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 45 sec. / Pretreatment - Pressure: 0.001 kPa |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 277 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TALOS ARCTICA |
|---|---|
| Image recording | Film or detector model: FEI FALCON III (4k x 4k) / Detector mode: COUNTING / Number grids imaged: 1 / Number real images: 1946901 / Average electron dose: 32.0 e/Å2 |
| Electron beam | Acceleration voltage: 200 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.6 µm / Nominal defocus min: 0.8 µm / Nominal magnification: 92000 |
| Experimental equipment |  Model: Talos Arctica / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Initial model |
| ||||||
|---|---|---|---|---|---|---|---|
| Refinement | Space: REAL / Protocol: FLEXIBLE FIT / Overall B value: 164.03 | ||||||
| Output model |  PDB-7tdm: |
 Movie
Movie Controller
Controller






 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)