+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | PZM21 bound Mu Opioid Receptor-Gi Protein Complex | |||||||||
 Map data Map data | PZM21 bound muOR Gi scFv16 complex | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | GPCR / MEMBRANE PROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationOpioid Signalling / spine apparatus / positive regulation of appetite / sperm ejaculation / G-protein activation / adenylate cyclase-inhibiting opioid receptor signaling pathway / Peptide ligand-binding receptors / beta-endorphin receptor activity / morphine receptor activity / negative regulation of Wnt protein secretion ...Opioid Signalling / spine apparatus / positive regulation of appetite / sperm ejaculation / G-protein activation / adenylate cyclase-inhibiting opioid receptor signaling pathway / Peptide ligand-binding receptors / beta-endorphin receptor activity / morphine receptor activity / negative regulation of Wnt protein secretion / negative regulation of luteinizing hormone secretion / G protein-coupled opioid receptor activity / G protein-coupled opioid receptor signaling pathway / regulation of cellular response to stress / G alpha (i) signalling events / negative regulation of nitric oxide biosynthetic process / negative regulation of adenylate cyclase-activating G protein-coupled receptor signaling pathway / adenylate cyclase-inhibiting G protein-coupled acetylcholine receptor signaling pathway / behavioral response to ethanol / regulation of NMDA receptor activity / neuropeptide binding / positive regulation of neurogenesis / eating behavior / negative regulation of cytosolic calcium ion concentration / transmission of nerve impulse / G-protein alpha-subunit binding / social behavior / voltage-gated calcium channel activity / adenylate cyclase inhibitor activity / positive regulation of protein localization to cell cortex / T cell migration / positive regulation of relaxation of smooth muscle / Adenylate cyclase inhibitory pathway / D2 dopamine receptor binding / adenylate cyclase-inhibiting serotonin receptor signaling pathway / G protein-coupled serotonin receptor binding / T-tubule / sensory perception of pain / positive regulation of gluconeogenesis / dendrite membrane / cellular response to forskolin / presynaptic modulation of chemical synaptic transmission / dendrite cytoplasm / regulation of mitotic spindle organization / chemokine-mediated signaling pathway / sarcoplasmic reticulum / Regulation of insulin secretion / neuropeptide signaling pathway / excitatory postsynaptic potential / response to prostaglandin E / locomotory behavior / electron transport chain / positive regulation of cholesterol biosynthetic process / negative regulation of insulin secretion / G protein-coupled receptor binding / sarcolemma / response to peptide hormone / G protein-coupled receptor activity / GABA-ergic synapse / adenylate cyclase-modulating G protein-coupled receptor signaling pathway / G-protein beta/gamma-subunit complex binding / centriolar satellite / adenylate cyclase-inhibiting G protein-coupled receptor signaling pathway / Olfactory Signaling Pathway / Activation of the phototransduction cascade / G protein-coupled acetylcholine receptor signaling pathway / G beta:gamma signalling through PLC beta / Presynaptic function of Kainate receptors / Thromboxane signalling through TP receptor / Activation of G protein gated Potassium channels / Inhibition of voltage gated Ca2+ channels via Gbeta/gamma subunits / G-protein activation / Glucagon signaling in metabolic regulation / G beta:gamma signalling through CDC42 / Prostacyclin signalling through prostacyclin receptor / G beta:gamma signalling through BTK / Synthesis, secretion, and inactivation of Glucagon-like Peptide-1 (GLP-1) / photoreceptor disc membrane / ADP signalling through P2Y purinoceptor 12 / Sensory perception of sweet, bitter, and umami (glutamate) taste / Glucagon-type ligand receptors / GDP binding / Adrenaline,noradrenaline inhibits insulin secretion / Vasopressin regulates renal water homeostasis via Aquaporins / Glucagon-like Peptide-1 (GLP1) regulates insulin secretion / G alpha (z) signalling events / cellular response to catecholamine stimulus / ADP signalling through P2Y purinoceptor 1 / ADORA2B mediated anti-inflammatory cytokines production / G beta:gamma signalling through PI3Kgamma / adenylate cyclase-activating dopamine receptor signaling pathway / Cooperation of PDCL (PhLP1) and TRiC/CCT in G-protein beta folding / GPER1 signaling / cellular response to prostaglandin E stimulus / heterotrimeric G-protein complex / G-protein beta-subunit binding / G alpha (12/13) signalling events / Inactivation, recovery and regulation of the phototransduction cascade / extracellular vesicle / sensory perception of taste Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) / Homo sapiens (human) /  | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 2.9 Å | |||||||||
 Authors Authors | Huang W / Qu Q | |||||||||
| Funding support |  United States, 1 items United States, 1 items
| |||||||||
 Citation Citation |  Journal: Angew Chem Int Ed Engl / Year: 2022 Journal: Angew Chem Int Ed Engl / Year: 2022Title: Structure-Based Evolution of G Protein-Biased μ-Opioid Receptor Agonists. Authors: Haoqing Wang / Florian Hetzer / Weijiao Huang / Qianhui Qu / Justin Meyerowitz / Jonas Kaindl / Harald Hübner / Georgios Skiniotis / Brian K Kobilka / Peter Gmeiner /    Abstract: The μ-opioid receptor (μOR) is the major target for opioid analgesics. Activation of μOR initiates signaling through G protein pathways as well as through β-arrestin recruitment. μOR agonists ...The μ-opioid receptor (μOR) is the major target for opioid analgesics. Activation of μOR initiates signaling through G protein pathways as well as through β-arrestin recruitment. μOR agonists that are biased towards G protein signaling pathways demonstrate diminished side effects. PZM21, discovered by computational docking, is a G protein biased μOR agonist. Here we report the cryoEM structure of PZM21 bound μOR in complex with G protein. Structure-based evolution led to multiple PZM21 analogs with more pronounced G protein bias and increased lipophilicity to improve CNS penetration. Among them, FH210 shows extremely low potency and efficacy for arrestin recruitment. We further determined the cryoEM structure of FH210 bound to μOR in complex with G protein and confirmed its expected binding pose. The structural and pharmacological studies reveal a potential mechanism to reduce β-arrestin recruitment by the μOR, and hold promise for developing next-generation analgesics with fewer adverse effects. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_24978.map.gz emd_24978.map.gz | 23.2 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-24978-v30.xml emd-24978-v30.xml emd-24978.xml emd-24978.xml | 18.7 KB 18.7 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_24978.png emd_24978.png | 100.6 KB | ||
| Filedesc metadata |  emd-24978.cif.gz emd-24978.cif.gz | 7.1 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-24978 http://ftp.pdbj.org/pub/emdb/structures/EMD-24978 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-24978 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-24978 | HTTPS FTP |
-Related structure data
| Related structure data |  7sbfMC  7scgC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_24978.map.gz / Format: CCP4 / Size: 40.6 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_24978.map.gz / Format: CCP4 / Size: 40.6 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | PZM21 bound muOR Gi scFv16 complex | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.06 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
- Sample components
Sample components
-Entire : PZM21 bound Mu Opioid Receptor-Gi Protein Complex
| Entire | Name: PZM21 bound Mu Opioid Receptor-Gi Protein Complex |
|---|---|
| Components |
|
-Supramolecule #1: PZM21 bound Mu Opioid Receptor-Gi Protein Complex
| Supramolecule | Name: PZM21 bound Mu Opioid Receptor-Gi Protein Complex / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#5 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: Guanine nucleotide-binding protein G(i) subunit alpha-1
| Macromolecule | Name: Guanine nucleotide-binding protein G(i) subunit alpha-1 type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 40.415031 KDa |
| Recombinant expression | Organism:  Trichoplusia ni (cabbage looper) Trichoplusia ni (cabbage looper) |
| Sequence | String: MGCTLSAEDK AAVERSKMID RNLREDGEKA AREVKLLLLG AGESGKSTIV KQMKIIHEAG YSEEECKQYK AVVYSNTIQS IIAIIRAMG RLKIDFGDSA RADDARQLFV LAGAAEEGFM TAELAGVIKR LWKDSGVQAC FNRSREYQLN DSAAYYLNDL D RIAQPNYI ...String: MGCTLSAEDK AAVERSKMID RNLREDGEKA AREVKLLLLG AGESGKSTIV KQMKIIHEAG YSEEECKQYK AVVYSNTIQS IIAIIRAMG RLKIDFGDSA RADDARQLFV LAGAAEEGFM TAELAGVIKR LWKDSGVQAC FNRSREYQLN DSAAYYLNDL D RIAQPNYI PTQQDVLRTR VKTTGIVETH FTFKDLHFKM FDVGGQRSER KKWIHCFEGV TAIIFCVALS DYDLVLAEDE EM NRMHESM KLFDSICNNK WFTDTSIILF LNKKDLFEEK IKKSPLTICY PEYAGSNTYE EAAAYIQCQF EDLNKRKDTK EIY THFTCA TDTKNVQFVF DAVTDVIIKN NLKDCGLF UniProtKB: Guanine nucleotide-binding protein G(i) subunit alpha-1 |
-Macromolecule #2: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1
| Macromolecule | Name: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1 type: protein_or_peptide / ID: 2 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 37.671102 KDa |
| Recombinant expression | Organism:  Trichoplusia ni (cabbage looper) Trichoplusia ni (cabbage looper) |
| Sequence | String: PGSSGSELDQ LRQEAEQLKN QIRDARKACA DATLSQITNN IDPVGRIQMR TRRTLRGHLA KIYAMHWGTD SRLLVSASQD GKLIIWDSY TTNKVHAIPL RSSWVMTCAY APSGNYVACG GLDNICSIYN LKTREGNVRV SRELAGHTGY LSCCRFLDDN Q IVTSSGDT ...String: PGSSGSELDQ LRQEAEQLKN QIRDARKACA DATLSQITNN IDPVGRIQMR TRRTLRGHLA KIYAMHWGTD SRLLVSASQD GKLIIWDSY TTNKVHAIPL RSSWVMTCAY APSGNYVACG GLDNICSIYN LKTREGNVRV SRELAGHTGY LSCCRFLDDN Q IVTSSGDT TCALWDIETG QQTTTFTGHT GDVMSLSLAP DTRLFVSGAC DASAKLWDVR EGMCRQTFTG HESDINAICF FP NGNAFAT GSDDATCRLF DLRADQELMT YSHDNIICGI TSVSFSKSGR LLLAGYDDFN CNVWDALKAD RAGVLAGHDN RVS CLGVTD DGMAVATGSW DSFLKIWN UniProtKB: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1 |
-Macromolecule #3: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2
| Macromolecule | Name: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2 type: protein_or_peptide / ID: 3 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 7.861143 KDa |
| Recombinant expression | Organism:  Trichoplusia ni (cabbage looper) Trichoplusia ni (cabbage looper) |
| Sequence | String: MASNNTASIA QARKLVEQLK MEANIDRIKV SKAAADLMAY CEAHAKEDPL LTPVPASENP FREKKFFCAI L UniProtKB: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2 |
-Macromolecule #4: scFv16
| Macromolecule | Name: scFv16 / type: protein_or_peptide / ID: 4 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 27.784896 KDa |
| Recombinant expression | Organism:  Trichoplusia ni (cabbage looper) Trichoplusia ni (cabbage looper) |
| Sequence | String: DVQLVESGGG LVQPGGSRKL SCSASGFAFS SFGMHWVRQA PEKGLEWVAY ISSGSGTIYY ADTVKGRFTI SRDDPKNTLF LQMTSLRSE DTAMYYCVRS IYYYGSSPFD FWGQGTTLTV SSGGGGSGGG GSGGGGSDIV MTQATSSVPV TPGESVSISC R SSKSLLHS ...String: DVQLVESGGG LVQPGGSRKL SCSASGFAFS SFGMHWVRQA PEKGLEWVAY ISSGSGTIYY ADTVKGRFTI SRDDPKNTLF LQMTSLRSE DTAMYYCVRS IYYYGSSPFD FWGQGTTLTV SSGGGGSGGG GSGGGGSDIV MTQATSSVPV TPGESVSISC R SSKSLLHS NGNTYLYWFL QRPGQSPQLL IYRMSNLASG VPDRFSGSGS GTAFTLTISR LEAEDVGVYY CMQHLEYPLT FG AGTKLEL KAAAHHHHHH HH |
-Macromolecule #5: Soluble cytochrome b562, Mu-type opioid receptor chimera,Mu-type ...
| Macromolecule | Name: Soluble cytochrome b562, Mu-type opioid receptor chimera,Mu-type opioid receptor type: protein_or_peptide / ID: 5 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 55.123824 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: CLVFADYKDD DDALEVLFQG PADLEDNWET LNDNLKVIEK ADNAAQVKDA LTKMRAAALD AQKATPPKLE DKSPDSPEMK DFRHGFDIL VGQIDDALKL ANEGKVKEAQ AAAEQLKTTR NAYIQKYLAG SPGARSASSL ALAIAITALY SIVCVVGLFG N FLVMYVIV ...String: CLVFADYKDD DDALEVLFQG PADLEDNWET LNDNLKVIEK ADNAAQVKDA LTKMRAAALD AQKATPPKLE DKSPDSPEMK DFRHGFDIL VGQIDDALKL ANEGKVKEAQ AAAEQLKTTR NAYIQKYLAG SPGARSASSL ALAIAITALY SIVCVVGLFG N FLVMYVIV RYTKMKTATN IYIFNLALAD ALATSTLPFQ SVNYLMGTWP FGNILCKIVI SIDYYNMFTS IFTLCTMSVD RY IAVCHPV KALDFRTPRN AKIVNVCNWI LSSAIGLPVM FMATTKYRQG SIDCTLTFSH PTWYWENLLK ICVFIFAFIM PVL IITVCY GLMILRLKSV RMLSGSKEKD RNLRRITRMV LVVVAVFIVC WTPIHIYVII KALITIPETT FQTVSWHFCI ALGY TNSCL NPVLYAFLDE NFKRCFREFC IPTSSTILEV LFQGPEQQNS ARIRQNTREH PSTANTVDRT NHQLENLEAE TAPLP DIHH HHHH UniProtKB: Soluble cytochrome b562, Mu-type opioid receptor |
-Macromolecule #6: N-[(2S)-2-(dimethylamino)-3-(4-hydroxyphenyl)propyl]-N'-[(2S)-1-(...
| Macromolecule | Name: N-[(2S)-2-(dimethylamino)-3-(4-hydroxyphenyl)propyl]-N'-[(2S)-1-(thiophen-3-yl)propan-2-yl]urea type: ligand / ID: 6 / Number of copies: 1 / Formula: 8QY |
|---|---|
| Molecular weight | Theoretical: 361.502 Da |
| Chemical component information | 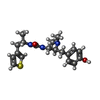 ChemComp-8QY: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.4 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | TFS KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Average electron dose: 66.75 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.5 µm / Nominal defocus min: 0.7000000000000001 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller




































 X (Sec.)
X (Sec.) Y (Row.)
Y (Row.) Z (Col.)
Z (Col.)





















