+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | C. elegans L1 80S ribosome | |||||||||
 Map data Map data | Post-processed map | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Caenorhabditis / elegans / ribosome / 80S / L1 / larva / C. elegans | |||||||||
| Biological species |  | |||||||||
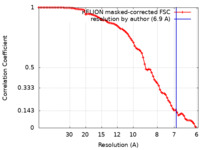
| Method | subtomogram averaging / cryo EM / Resolution: 6.9 Å | |||||||||
 Authors Authors | Schioetz OH / Kaiser CJO / Klumpe S / Beck F / Plitzko JM | |||||||||
| Funding support |  Germany, 1 items Germany, 1 items
| |||||||||
 Citation Citation |  Journal: Nat Methods / Year: 2024 Journal: Nat Methods / Year: 2024Title: Serial Lift-Out: sampling the molecular anatomy of whole organisms. Authors: Oda Helene Schiøtz / Christoph J O Kaiser / Sven Klumpe / Dustin R Morado / Matthias Poege / Jonathan Schneider / Florian Beck / David P Klebl / Christopher Thompson / Jürgen M Plitzko /    Abstract: Cryo-focused ion beam milling of frozen-hydrated cells and subsequent cryo-electron tomography (cryo-ET) has enabled the structural elucidation of macromolecular complexes directly inside cells. ...Cryo-focused ion beam milling of frozen-hydrated cells and subsequent cryo-electron tomography (cryo-ET) has enabled the structural elucidation of macromolecular complexes directly inside cells. Application of the technique to multicellular organisms and tissues, however, is still limited by sample preparation. While high-pressure freezing enables the vitrification of thicker samples, it prolongs subsequent preparation due to increased thinning times and the need for extraction procedures. Additionally, thinning removes large portions of the specimen, restricting the imageable volume to the thickness of the final lamella, typically <300 nm. Here we introduce Serial Lift-Out, an enhanced lift-out technique that increases throughput and obtainable contextual information by preparing multiple sections from single transfers. We apply Serial Lift-Out to Caenorhabditis elegans L1 larvae, yielding a cryo-ET dataset sampling the worm's anterior-posterior axis, and resolve its ribosome structure to 7 Å and a subregion of the 11-protofilament microtubule to 13 Å, illustrating how Serial Lift-Out enables the study of multicellular molecular anatomy. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_17241.map.gz emd_17241.map.gz | 42.5 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-17241-v30.xml emd-17241-v30.xml emd-17241.xml emd-17241.xml | 17.4 KB 17.4 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_17241_fsc.xml emd_17241_fsc.xml | 8.1 KB | Display |  FSC data file FSC data file |
| Images |  emd_17241.png emd_17241.png | 103.1 KB | ||
| Masks |  emd_17241_msk_1.map emd_17241_msk_1.map | 45.2 MB |  Mask map Mask map | |
| Filedesc metadata |  emd-17241.cif.gz emd-17241.cif.gz | 4.5 KB | ||
| Others |  emd_17241_additional_1.map.gz emd_17241_additional_1.map.gz emd_17241_half_map_1.map.gz emd_17241_half_map_1.map.gz emd_17241_half_map_2.map.gz emd_17241_half_map_2.map.gz | 3.6 MB 23.2 MB 23.2 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-17241 http://ftp.pdbj.org/pub/emdb/structures/EMD-17241 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-17241 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-17241 | HTTPS FTP |
-Related structure data
| Related structure data | C: citing same article ( |
|---|
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_17241.map.gz / Format: CCP4 / Size: 45.2 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_17241.map.gz / Format: CCP4 / Size: 45.2 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Post-processed map | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 2.98 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Mask #1
| File |  emd_17241_msk_1.map emd_17241_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Additional map: Masked post-processed map
| File | emd_17241_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Masked post-processed map | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: Half map 1
| File | emd_17241_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half map 1 | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : C. elegans L1 larva, CGC strain NK2476
| Entire | Name: C. elegans L1 larva, CGC strain NK2476 |
|---|---|
| Components |
|
-Supramolecule #1: C. elegans L1 larva, CGC strain NK2476
| Supramolecule | Name: C. elegans L1 larva, CGC strain NK2476 / type: complex / ID: 1 / Parent: 0 |
|---|---|
| Source (natural) | Organism:  |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | subtomogram averaging |
| Aggregation state | tissue |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 / Details: M9 buffer + 20% Ficoll 400 |
|---|---|
| Grid | Model: Homemade / Material: COPPER / Mesh: 75 / Support film - Material: FORMVAR / Support film - topology: CONTINUOUS / Support film - Film thickness: 100 |
| Vitrification | Cryogen name: NITROGEN / Details: High pressure freezing, Leica EM ICE. |
| Details | Developmentally arrested L1 larvae |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Specialist optics | Energy filter - Name: TFS Selectris X / Energy filter - Slit width: 10 eV |
| Image recording | Film or detector model: FEI FALCON IV (4k x 4k) / Digitization - Dimensions - Width: 4096 pixel / Digitization - Dimensions - Height: 4096 pixel / Number real images: 1 / Average exposure time: 0.68 sec. / Average electron dose: 3.2 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 50.0 µm / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 4.5 µm / Nominal defocus min: 1.5 µm / Nominal magnification: 64000 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller













 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)