+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of BoNT/Wo-NTNH complex | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | The complex of botulinum neurotoxin-like protein from Weissella oryzae and its non-toxic non-hemagglutinin partner. / TOXIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationbontoxilysin / metalloendopeptidase activity / proteolysis / zinc ion binding / metal ion binding Similarity search - Function | |||||||||
| Biological species |  Weissella oryzae (bacteria) Weissella oryzae (bacteria) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 2.98 Å | |||||||||
 Authors Authors | Kosenina S / Skerlova J / Stenmark P | |||||||||
| Funding support |  Sweden, 2 items Sweden, 2 items
| |||||||||
 Citation Citation |  Journal: FEBS J / Year: 2024 Journal: FEBS J / Year: 2024Title: The cryo-EM structure of the BoNT/Wo-NTNH complex reveals two immunoglobulin-like domains. Authors: Sara Košenina / Jana Škerlová / Sicai Zhang / Min Dong / Pål Stenmark /    Abstract: The botulinum neurotoxin-like toxin from Weissella oryzae (BoNT/Wo) is one of the BoNT-like toxins recently identified outside of the Clostridium genus. We show that, like the canonical BoNTs, ...The botulinum neurotoxin-like toxin from Weissella oryzae (BoNT/Wo) is one of the BoNT-like toxins recently identified outside of the Clostridium genus. We show that, like the canonical BoNTs, BoNT/Wo forms a complex with its non-toxic non-hemagglutinin (NTNH) partner, which in traditional BoNT serotypes protects the toxin from proteases and the acidic environment of the hosts' guts. We here report the cryo-EM structure of the 300 kDa BoNT/Wo-NTNH/Wo complex together with pH stability studies of the complex. The structure reveals molecular details of the toxin's interactions with its protective partner. The overall structural arrangement is similar to other reported BoNT-NTNH complexes, but NTNH/Wo uniquely contains two extra bacterial immunoglobulin-like (Big) domains on the C-terminus. Although the function of these Big domains is unknown, they are structurally most similar to bacterial proteins involved in adhesion to host cells. In addition, the BoNT/Wo protease domain contains an internal disulfide bond not seen in other BoNTs. Mass photometry analysis revealed that the BoNT/Wo-NTNH/Wo complex is stable under acidic conditions and may dissociate at neutral to basic pH. These findings established that BoNT/Wo-NTNH/Wo shares the general fold of canonical BoNT-NTNH complexes. The presence of unique structural features suggests that it may have an alternative mode of activation, translocation and recognition of host cells, raising interesting questions about the activity and the mechanism of action of BoNT/Wo as well as about its target environment, receptors and substrates. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_16475.map.gz emd_16475.map.gz | 118 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-16475-v30.xml emd-16475-v30.xml emd-16475.xml emd-16475.xml | 21 KB 21 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_16475_fsc.xml emd_16475_fsc.xml | 14.7 KB | Display |  FSC data file FSC data file |
| Images |  emd_16475.png emd_16475.png | 90.4 KB | ||
| Filedesc metadata |  emd-16475.cif.gz emd-16475.cif.gz | 7.4 KB | ||
| Others |  emd_16475_half_map_1.map.gz emd_16475_half_map_1.map.gz emd_16475_half_map_2.map.gz emd_16475_half_map_2.map.gz | 116.1 MB 116.1 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-16475 http://ftp.pdbj.org/pub/emdb/structures/EMD-16475 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-16475 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-16475 | HTTPS FTP |
-Related structure data
| Related structure data |  8c8gMC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_16475.map.gz / Format: CCP4 / Size: 125 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_16475.map.gz / Format: CCP4 / Size: 125 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.82 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #1
| File | emd_16475_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
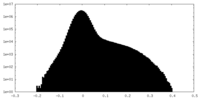
| Density Histograms |
-Half map: #2
| File | emd_16475_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Botulinum neurotoxin-like protein from Weissella oryzae in comple...
| Entire | Name: Botulinum neurotoxin-like protein from Weissella oryzae in complex with its non-toxic non-hemagglutinin partner protein |
|---|---|
| Components |
|
-Supramolecule #1: Botulinum neurotoxin-like protein from Weissella oryzae in comple...
| Supramolecule | Name: Botulinum neurotoxin-like protein from Weissella oryzae in complex with its non-toxic non-hemagglutinin partner protein type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  Weissella oryzae (bacteria) Weissella oryzae (bacteria) |
-Macromolecule #1: Putative botulinum-like toxin Wo
| Macromolecule | Name: Putative botulinum-like toxin Wo / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO / EC number: bontoxilysin |
|---|---|
| Source (natural) | Organism:  Weissella oryzae (bacteria) Weissella oryzae (bacteria) |
| Molecular weight | Theoretical: 156.964484 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MDVLEMFDVN YESPILESFD STTQSLNDVH VFMSRIQMSA YDADGEGRIE YRNLKLYEIS SGIFISTDRL DTGASGVEDD HEMVDYYSS ARLTREFLGE SLDSQKSDYF EGIKKVFSFY KNKCNESRYI KEFFEEIQFR NICGFPKQAG TSSTDIFDQF N SVDVLLQD ...String: MDVLEMFDVN YESPILESFD STTQSLNDVH VFMSRIQMSA YDADGEGRIE YRNLKLYEIS SGIFISTDRL DTGASGVEDD HEMVDYYSS ARLTREFLGE SLDSQKSDYF EGIKKVFSFY KNKCNESRYI KEFFEEIQFR NICGFPKQAG TSSTDIFDQF N SVDVLLQD PVTSVWNKKV GSKKANIVII PPATNLPITE ACATAGFQPE GFPKLGSGSF FTVQFDPFFS TRFKAHETDD VA LLDPTLT LLHEMTHGLH FQKGIANPVN RSGETPAWAT TWGRVTGDND AFKETPMEEL LTFNKHTIDD DIEISDHLKS TYI GFLYNG RNEDDPTESV DGVYQNVSSF LNQYRGFEIS SDFQHFIESC YGVKYNQESK KFIVNPRNIK RYVQDGFFID EAKF ARILN IKTRSYYTLM PDNLGVWSYR VDILNRLRET FDEDRGLLSQ ELDFHTALTP VVSENPALEL EVAGMQRMVS LPKIK ASYL PSDIKIKNFT GQKISHDTIL DTNISGIIIS KIKYKSDFVV DESMPRSSLN TTNYNLSPIK GTKFETDIRD KTSVKV TVS EITAPMINHV MKLDNSKVLT ERPSLNEDLE ETFKNTKDVY IPKTTAMMKL KEGADQTLGA VGFAVWSGQI LEDLYNL AQ KKEVSIDQIK DDLMSILPFY CAYKNLSAEK YEQAFANATL DAFLIFATDG GGFAGLGITV GAIAINSMYA KAETMEAY D SMFGKYVDQY QNDIKNFTLN AYVQWENNIL SRLWNESRLA ITGFRNMLKT VKTVMEFDAT NQAYSEEDRK IIKAKCEEI FSEFPMLMQT FAKNSMTANL ENASKIFNDI VWQKIKEELD QYVIDSKKYF LDSLEEAYNN GSISAESYYK YQTEAREKFV SPREVIDLY IAAHDTVVKR KRYIRRYSRK YDLATDFKGN TVHLNGLGEG TQDIQDLYGN YSVYADKKTV STQEGHFDQT I KIAKDTNT INKVVLAVSS NNGKEYALNK DEQYTISFWL RMPVPSSSEE RRIFSYSAVS GVNKEVEELI LQVKNNEFVL AT ANLLRNS EFVIEPRIAL NRWVKITIVN ENTRIKVYQN DNLLGLIKDS SRKKPIAQRG TFKFYNYNVD YQLDDISYYN GTI SQRDIK YTFKEDHGQF VYDHWGERLQ YNKAYYLLSD DNKSAFETVY ETKRLKLKSV PGVDIKYLGM NDRVYGYYGG LQFK LVPLD SKNMNNYVRW GDKFTMQSIE TTNLSLAIIQ DNAYFAPTQL KLISNEGKSE EEIFTFDRNI KLQNAAILVG TGNSK QGPI SAYKRGYSGD LWINGARLDG YVTVVNKSNY SNDEIQEKFK WIFVPKDANW VEGSYPYDVP DYALESGKET AAAKFE RQH MDSSTSAA UniProtKB: Putative botulinum-like toxin Wo |
-Macromolecule #2: Structural protein
| Macromolecule | Name: Structural protein / type: protein_or_peptide / ID: 2 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Weissella oryzae (bacteria) Weissella oryzae (bacteria) |
| Molecular weight | Theoretical: 166.969734 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MGSDYKDDDD KSGMDNKLKT ENIRYFRTAA GSEDVLEVKA YEVYPNVWAI PSRYMMEPLQ DLDEVTNPEQ FSIYDKKYLA DIQEQDEFL KSIQAAIEDI KKRTFGLELL TAVSGAVPLP KDTGATNTTL QCIDENGKHT HDVVANVVLW GPGNNLNSNR L ISKSDDDS ...String: MGSDYKDDDD KSGMDNKLKT ENIRYFRTAA GSEDVLEVKA YEVYPNVWAI PSRYMMEPLQ DLDEVTNPEQ FSIYDKKYLA DIQEQDEFL KSIQAAIEDI KKRTFGLELL TAVSGAVPLP KDTGATNTTL QCIDENGKHT HDVVANVVLW GPGNNLNSNR L ISKSDDDS NGIGSMVELI WNPQILIKNI GTNRIKPATD ELVGLLTKAL FRLYGLGLNK IRYPFYQLDD KKYYSLTAED LI SYGGFSA NVVNLQPYYF LEDQFTKVKE KYESAKKRID DIKVNDEYSQ MLTLKYQFDL YSLFHISTSY IVSTVIPAND KYG GLVSYY TGPNALIDSK TDEKLTSMVK IPLKKIKYSK NQSREYDEYD LTNGEDSTQY FENFTFPKSK HVFVETQPTP ENVF VNLPS EEITKIILPV IPAESDLIKI PFQPATPKSI TTELITTDVP TLGLIFPAVK SKQNLSDIKM TSKLSDALDS DKQTF AFDN TLVDKLSELT SVSDAELFGI IRLIKNELLS VIDNFTTFGD NWSCPRWIDY CFQQVFGSDL KNLIVQGDFE KVFNIS DTL ILPKQLPEDI LQLKPYLFYQ WYAKRYTRIL RLESLFYQIL NEHITLIRSL VSSNNKGQYL QGFMNDLDKI AYNAQYM LS DWTIQLGYYD FKNQVTQVIK TSSMTSEFNI DDLLYDYDTF KLTISQFGAD SINNFTPSQD LKLALNDNNS PILLLGND E IKSNGSITQT DDSLDDETSL LLSKNTSFEG NFSAKYLLSS VGVNFTFKSI ENLNFSVDFM NINIAFSNNF FEITQTGQE TKKYSIAKLF GWNSLVYLIK HSSVEIWDIH SNILLVSHDL TAPQNNIVKA PIKLTNLDNE LILKSFEVFE QDEEANYNDI EQGFKNGII YTAKKMPIIV GEKYALKSSI LDDMGILTSD ENKKYPVFST DVEVESSLNI ILESTTGDKI SVDAGVNIRT I NSNGEENY LGIEDNHLIF VPKEEAELFY LKKAVVEDTI DIFYVVKTLG NMFINVERIS DNIYRLNFKA GILYSTMESD ML VLPAEEA NTAFYIQPIG LASLEVKDSV LGEGNPWLKE DNFLDATDDY GNQIDLSDNR ISVTGSVDTD KVGTYSVVYS YTG IDKTNT EKATITVKLD KSSIKTQDST LQNGKEWVRA DNLVEVIDED GNKVDYSDDR IIQEGDVDIN KAGVYDITFR YRGK FKIIS SSFKVTVIND IWYDSIKNAC KTYLIDYGER INDVKGITFQ NILEATRGKL YGYRVVYDNP HDVINQNPPK DFHFD LIKP FDVKNPSRVH LADYSGYLRL FIISTGKINT DIKVKIYAVL ENKDEIEIFD NHQNDKRHEE IAEIYKSNFD DNNYSA DGK YFISVLFKND VQAVVKDEIY GYEIFYSYFT KFRKDTAFQT DGSKRIFFHD YFNFEVPLDY KDSTFINVIL KNGEKIR IY KFAGYYYYSG HHHHHH UniProtKB: Ig-like domain-containing protein |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation #1
Sample preparation #1
| Preparation ID | 1 | ||||||
|---|---|---|---|---|---|---|---|
| Concentration | 0.7 mg/mL | ||||||
| Buffer | pH: 5.5 / Component:
| ||||||
| Grid | Model: Quantifoil R2/2 / Material: COPPER / Mesh: 300 / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 60 sec. | ||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 277 K / Instrument: FEI VITROBOT MARK IV |
- Sample preparation #2
Sample preparation #2
| Preparation ID | 2 | ||||||
|---|---|---|---|---|---|---|---|
| Concentration | 0.2 mg/mL | ||||||
| Buffer | pH: 5.5 / Component:
| ||||||
| Grid | Model: Quantifoil R2/2 / Material: COPPER / Mesh: 300 / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 60 sec. | ||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 277 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: COUNTING / Number grids imaged: 2 / Number real images: 5946 / Average electron dose: 40.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: OTHER / Imaging mode: BRIGHT FIELD / Nominal defocus max: 3.2 µm / Nominal defocus min: 1.6 µm / Nominal magnification: 165000 |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Refinement | Space: REAL / Protocol: AB INITIO MODEL |
|---|---|
| Output model |  PDB-8c8g: |
 Movie
Movie Controller
Controller




 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)