+ Open data
Open data
- Basic information
Basic information
| Entry |  | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Release state - ES27up (pre-60S) | ||||||||||||
 Map data Map data | Composite map | ||||||||||||
 Sample Sample |
| ||||||||||||
| Biological species |  | ||||||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.0 Å | ||||||||||||
 Authors Authors | Thoms M / Mitterer V / Buschauer R / Berninghausen O / Hurt E / Beckmann R | ||||||||||||
| Funding support |  Germany, European Union, 3 items Germany, European Union, 3 items
| ||||||||||||
 Citation Citation |  Journal: Elife / Year: 2023 Journal: Elife / Year: 2023Title: Concurrent remodelling of nucleolar 60S subunit precursors by the Rea1 ATPase and Spb4 RNA helicase. Authors: Valentin Mitterer / Matthias Thoms / Robert Buschauer / Otto Berninghausen / Ed Hurt / Roland Beckmann /  Abstract: Biogenesis intermediates of nucleolar ribosomal 60S precursor particles undergo a number of structural maturation steps before they transit to the nucleoplasm and are finally exported into the ...Biogenesis intermediates of nucleolar ribosomal 60S precursor particles undergo a number of structural maturation steps before they transit to the nucleoplasm and are finally exported into the cytoplasm. The AAA-ATPase Rea1 participates in the nucleolar exit by releasing the Ytm1-Erb1 heterodimer from the evolving pre-60S particle. Here, we show that the DEAD-box RNA helicase Spb4 with its interacting partner Rrp17 is further integrated into this maturation event. Spb4 binds to a specific class of late nucleolar pre-60S intermediates, whose cryo-EM structure revealed how its helicase activity facilitates melting and restructuring of 25S rRNA helices H62 and H63/H63a prior to Ytm1-Erb1 release. In vitro maturation of such Spb4-enriched pre-60S particles, incubated with purified Rea1 and its associated pentameric Rix1-complex in the presence of ATP, combined with cryo-EM analysis depicted the details of the Rea1-dependent large-scale pre-ribosomal remodeling. Our structural insights unveil how the Rea1 ATPase and Spb4 helicase remodel late nucleolar pre-60S particles by rRNA restructuring and dismantling of a network of several ribosomal assembly factors. | ||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_16276.map.gz emd_16276.map.gz | 19.7 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-16276-v30.xml emd-16276-v30.xml emd-16276.xml emd-16276.xml | 34.7 KB 34.7 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_16276.png emd_16276.png | 87.4 KB | ||
| Others |  emd_16276_additional_1.map.gz emd_16276_additional_1.map.gz emd_16276_additional_2.map.gz emd_16276_additional_2.map.gz emd_16276_additional_3.map.gz emd_16276_additional_3.map.gz emd_16276_additional_4.map.gz emd_16276_additional_4.map.gz emd_16276_additional_5.map.gz emd_16276_additional_5.map.gz emd_16276_additional_6.map.gz emd_16276_additional_6.map.gz emd_16276_additional_7.map.gz emd_16276_additional_7.map.gz emd_16276_half_map_1.map.gz emd_16276_half_map_1.map.gz emd_16276_half_map_2.map.gz emd_16276_half_map_2.map.gz | 6.6 MB 8.4 MB 7.1 MB 2.5 MB 21.7 MB 239.6 MB 450.2 MB 442 MB 442 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-16276 http://ftp.pdbj.org/pub/emdb/structures/EMD-16276 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-16276 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-16276 | HTTPS FTP |
-Validation report
| Summary document |  emd_16276_validation.pdf.gz emd_16276_validation.pdf.gz | 809.5 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_16276_full_validation.pdf.gz emd_16276_full_validation.pdf.gz | 809 KB | Display | |
| Data in XML |  emd_16276_validation.xml.gz emd_16276_validation.xml.gz | 18.6 KB | Display | |
| Data in CIF |  emd_16276_validation.cif.gz emd_16276_validation.cif.gz | 22.1 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-16276 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-16276 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-16276 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-16276 | HTTPS FTP |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_16276.map.gz / Format: CCP4 / Size: 476.8 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_16276.map.gz / Format: CCP4 / Size: 476.8 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Composite map | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.059 Å | ||||||||||||||||||||||||||||||||||||
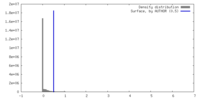
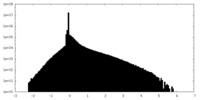
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Additional map: Foot - Local Refinement - Local filtered map
| File | emd_16276_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Foot - Local Refinement - Local filtered map | ||||||||||||
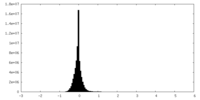
| Projections & Slices |
| ||||||||||||
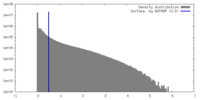
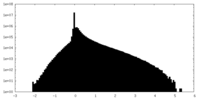
| Density Histograms |
-Additional map: ES27 - Local Refinement - Local filtered map
| File | emd_16276_additional_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | ES27 - Local Refinement - Local filtered map | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Additional map: Nog1 - Local Refinement - Local filtered map
| File | emd_16276_additional_3.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Nog1 - Local Refinement - Local filtered map | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Additional map: Spb1-MTD - Local Refinement - Local filtered map
| File | emd_16276_additional_4.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Spb1-MTD - Local Refinement - Local filtered map | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Additional map: Consensus cryo-EM map - Local filtered map
| File | emd_16276_additional_5.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Consensus cryo-EM map - Local filtered map | ||||||||||||
| Projections & Slices |
| ||||||||||||
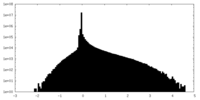
| Density Histograms |
-Additional map: Consensus cryo-EM map - Homogeneous Refinement
| File | emd_16276_additional_6.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Consensus cryo-EM map - Homogeneous Refinement | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Additional map: Consensus cryo-EM map - Homogeneous Refinement - sharpened map
| File | emd_16276_additional_7.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Consensus cryo-EM map - Homogeneous Refinement - sharpened map | ||||||||||||
| Projections & Slices |
| ||||||||||||
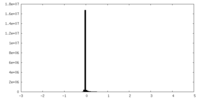
| Density Histograms |
-Half map: Consensus cryo-EM map - half map A
| File | emd_16276_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Consensus cryo-EM map - half map A | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: Consensus cryo-EM map - half map B
| File | emd_16276_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Consensus cryo-EM map - half map B | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Release state - ES27up (pre-60S)
| Entire | Name: Release state - ES27up (pre-60S) |
|---|---|
| Components |
|
-Supramolecule #1: Release state - ES27up (pre-60S)
| Supramolecule | Name: Release state - ES27up (pre-60S) / type: complex / ID: 1 / Chimera: Yes / Parent: 0 / Macromolecule list: #1-#60 |
|---|---|
| Source (natural) | Organism:  |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Average electron dose: 46.8 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: SPOT SCAN / Imaging mode: BRIGHT FIELD / Nominal defocus max: 3.5 µm / Nominal defocus min: 0.5 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: EMDB MAP EMDB ID: |
|---|---|
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 3.0 Å / Resolution method: FSC 0.143 CUT-OFF / Number images used: 29118 |
| Initial angle assignment | Type: MAXIMUM LIKELIHOOD |
| Final angle assignment | Type: MAXIMUM LIKELIHOOD |
 Movie
Movie Controller
Controller




 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)