[English] 日本語
 Yorodumi
Yorodumi- EMDB-14801: Cryo-EM structure of holo-PdxR from Bacillus clausii bound to its... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of holo-PdxR from Bacillus clausii bound to its target DNA in the closed conformation, C2 symmetry | |||||||||
 Map data Map data | Full cryo-EM map of holo-PdxR in complex with its target dsDNA in the closed conformation (C2 symmetry) | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | transcription factor / PLP-binding protein / domain-swap homodimer / DNA binding protein | |||||||||
| Function / homology |  Function and homology information Function and homology informationtransaminase activity / pyridoxal phosphate binding / DNA-binding transcription factor activity / DNA binding Similarity search - Function | |||||||||
| Biological species |  Alkalihalobacillus clausii (bacteria) Alkalihalobacillus clausii (bacteria) | |||||||||
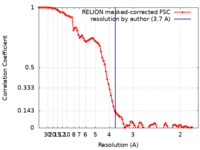
| Method | single particle reconstruction / cryo EM / Resolution: 3.7 Å | |||||||||
 Authors Authors | Freda I / Montemiglio LC / Tramonti A / Contestabile R / Vallone B / Exertier C / Savino C / Chaves Sanjuan A / Bolognesi M | |||||||||
| Funding support | 1 items
| |||||||||
 Citation Citation |  Journal: Nucleic Acids Res / Year: 2023 Journal: Nucleic Acids Res / Year: 2023Title: Structural insights into the DNA recognition mechanism by the bacterial transcription factor PdxR. Authors: Ida Freda / Cécile Exertier / Anna Barile / Antonio Chaves-Sanjuan / Mirella Vivoli Vega / Michail N Isupov / Nicholas J Harmer / Elena Gugole / Paolo Swuec / Martino Bolognesi / Anita ...Authors: Ida Freda / Cécile Exertier / Anna Barile / Antonio Chaves-Sanjuan / Mirella Vivoli Vega / Michail N Isupov / Nicholas J Harmer / Elena Gugole / Paolo Swuec / Martino Bolognesi / Anita Scipioni / Carmelinda Savino / Martino Luigi Di Salvo / Roberto Contestabile / Beatrice Vallone / Angela Tramonti / Linda Celeste Montemiglio /   Abstract: Specificity in protein-DNA recognition arises from the synergy of several factors that stem from the structural and chemical signatures encoded within the targeted DNA molecule. Here, we deciphered ...Specificity in protein-DNA recognition arises from the synergy of several factors that stem from the structural and chemical signatures encoded within the targeted DNA molecule. Here, we deciphered the nature of the interactions driving DNA recognition and binding by the bacterial transcription factor PdxR, a member of the MocR family responsible for the regulation of pyridoxal 5'-phosphate (PLP) biosynthesis. Single particle cryo-EM performed on the PLP-PdxR bound to its target DNA enabled the isolation of three conformers of the complex, which may be considered as snapshots of the binding process. Moreover, the resolution of an apo-PdxR crystallographic structure provided a detailed description of the transition of the effector domain to the holo-PdxR form triggered by the binding of the PLP effector molecule. Binding analyses of mutated DNA sequences using both wild type and PdxR variants revealed a central role of electrostatic interactions and of the intrinsic asymmetric bending of the DNA in allosterically guiding the holo-PdxR-DNA recognition process, from the first encounter through the fully bound state. Our results detail the structure and dynamics of the PdxR-DNA complex, clarifying the mechanism governing the DNA-binding mode of the holo-PdxR and the regulation features of the MocR family of transcription factors. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_14801.map.gz emd_14801.map.gz | 58.9 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-14801-v30.xml emd-14801-v30.xml emd-14801.xml emd-14801.xml | 21.2 KB 21.2 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_14801_fsc.xml emd_14801_fsc.xml | 9.1 KB | Display |  FSC data file FSC data file |
| Images |  emd_14801.png emd_14801.png | 45.8 KB | ||
| Filedesc metadata |  emd-14801.cif.gz emd-14801.cif.gz | 6.9 KB | ||
| Others |  emd_14801_half_map_1.map.gz emd_14801_half_map_1.map.gz emd_14801_half_map_2.map.gz emd_14801_half_map_2.map.gz | 48.5 MB 48.5 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-14801 http://ftp.pdbj.org/pub/emdb/structures/EMD-14801 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-14801 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-14801 | HTTPS FTP |
-Related structure data
| Related structure data |  7zn5MC  7pq9C  7zlaC  7zpaC  7zthC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_14801.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_14801.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Full cryo-EM map of holo-PdxR in complex with its target dsDNA in the closed conformation (C2 symmetry) | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.889 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: Half cryo-EM map 2 of holo-PdxR in complex...
| File | emd_14801_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half cryo-EM map 2 of holo-PdxR in complex with its target dsDNA in the closed conformation (C2 symmetry) | ||||||||||||
| Projections & Slices |
| ||||||||||||
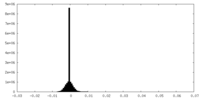
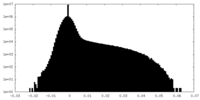
| Density Histograms |
-Half map: Half cryo-EM map 1 of holo-PdxR in complex...
| File | emd_14801_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half cryo-EM map 1 of holo-PdxR in complex with its target dsDNA in the closed conformation (C2 symmetry) | ||||||||||||
| Projections & Slices |
| ||||||||||||
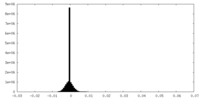
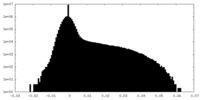
| Density Histograms |
- Sample components
Sample components
-Entire : Binary complex between the PLP-bound homodimeric PdxR and a 48bp ...
| Entire | Name: Binary complex between the PLP-bound homodimeric PdxR and a 48bp double strand DNA fragment |
|---|---|
| Components |
|
-Supramolecule #1: Binary complex between the PLP-bound homodimeric PdxR and a 48bp ...
| Supramolecule | Name: Binary complex between the PLP-bound homodimeric PdxR and a 48bp double strand DNA fragment type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  Alkalihalobacillus clausii (bacteria) Alkalihalobacillus clausii (bacteria) |
| Molecular weight | Theoretical: 136 KDa |
-Macromolecule #1: PLP-dependent aminotransferase family protein
| Macromolecule | Name: PLP-dependent aminotransferase family protein / type: protein_or_peptide / ID: 1 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Alkalihalobacillus clausii (bacteria) Alkalihalobacillus clausii (bacteria) |
| Molecular weight | Theoretical: 55.499121 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MELLWCELNR DLPTPLYEQL YAHIKTEITE GRIGYGTKLP SKRKLADSLK LSQNTVEAAY EQLVAEGYVE VIPRKGFYVQ AYEDLEYIR APQAPGDALA TKQDTIRYNF HPTHIDTTSF PFEQWRKYFK QTMCKENHRL LLNGDHQGEA SFRREIAYYL H HSRGVNCT ...String: MELLWCELNR DLPTPLYEQL YAHIKTEITE GRIGYGTKLP SKRKLADSLK LSQNTVEAAY EQLVAEGYVE VIPRKGFYVQ AYEDLEYIR APQAPGDALA TKQDTIRYNF HPTHIDTTSF PFEQWRKYFK QTMCKENHRL LLNGDHQGEA SFRREIAYYL H HSRGVNCT PEQVVVGAGV ETLLQQLFLL LGESKVYGIE DPGYQLMRKL LSHYPNDYVP FQVDEEGIDV DSIVRTAVDV VY TTPSRHF PYGSVLSINR RKQLLHWAEA HENRYIIEDD YDSEFRYTGK TIPSLQSMDV HNKVIYLGAF S(LLP)SLIPSVR ISYMVLPAPL AHLYKNKFSY YHSTVSRIDQ QVLTAFMKQG DFEKHLNRMR KIYRRKLEKV LSLLKRYEDK LLIIGERSGL HIVLVVKNG MDEQTLVEKA LAAKAKVYPL SAYSLERAIH PPQIVLGFGS IPEDELEEAI ATVLNAWGFL VPRGSLEHHH H HH UniProtKB: PLP-dependent aminotransferase family protein |
-Macromolecule #2: DNA (48-MER)
| Macromolecule | Name: DNA (48-MER) / type: dna / ID: 2 / Number of copies: 1 / Classification: DNA |
|---|---|
| Source (natural) | Organism:  Alkalihalobacillus clausii (bacteria) Alkalihalobacillus clausii (bacteria) |
| Molecular weight | Theoretical: 14.667447 KDa |
| Sequence | String: (DC)(DT)(DG)(DA)(DC)(DC)(DT)(DC)(DA)(DT) (DC)(DA)(DT)(DT)(DT)(DT)(DC)(DT)(DT)(DA) (DA)(DA)(DA)(DA)(DC)(DT)(DG)(DA)(DC) (DA)(DC)(DT)(DT)(DA)(DC)(DA)(DA)(DT)(DG) (DT) (DG)(DG)(DT)(DC)(DA)(DG)(DT)(DT) |
-Macromolecule #3: DNA (48-MER)
| Macromolecule | Name: DNA (48-MER) / type: dna / ID: 3 / Number of copies: 1 / Classification: DNA |
|---|---|
| Source (natural) | Organism:  Alkalihalobacillus clausii (bacteria) Alkalihalobacillus clausii (bacteria) |
| Molecular weight | Theoretical: 14.894607 KDa |
| Sequence | String: (DA)(DA)(DC)(DT)(DG)(DA)(DC)(DC)(DA)(DC) (DA)(DT)(DT)(DG)(DT)(DA)(DA)(DG)(DT)(DG) (DT)(DC)(DA)(DG)(DT)(DT)(DT)(DT)(DT) (DA)(DA)(DG)(DA)(DA)(DA)(DA)(DT)(DG)(DA) (DT) (DG)(DA)(DG)(DG)(DT)(DC)(DA)(DG) |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.5 mg/mL | ||||||
|---|---|---|---|---|---|---|---|
| Buffer | pH: 7.5 / Component:
| ||||||
| Grid | Model: Quantifoil R1.2/1.3 / Material: COPPER / Mesh: 300 / Support film - Material: CARBON / Support film - topology: HOLEY / Support film - Film thickness: 12 / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 30 sec. / Details: GloQube system (Quorum Technologies) | ||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 277.15 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TALOS ARCTICA |
|---|---|
| Image recording | Film or detector model: FEI FALCON III (4k x 4k) / Detector mode: COUNTING / Number grids imaged: 1 / Number real images: 3284 / Average exposure time: 1.0 sec. / Average electron dose: 40.0 e/Å2 |
| Electron beam | Acceleration voltage: 200 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.2 µm / Nominal defocus min: 0.8 µm / Nominal magnification: 120000 |
| Sample stage | Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Talos Arctica / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Initial model |
| ||||||
|---|---|---|---|---|---|---|---|
| Refinement | Space: REAL / Protocol: RIGID BODY FIT / Overall B value: 142 | ||||||
| Output model |  PDB-7zn5: |
 Movie
Movie Controller
Controller






 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)