+ データを開く
データを開く
- 基本情報
基本情報
| 登録情報 |  | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| タイトル | TIR-SAVED effector bound to cA3 | ||||||||||||
 マップデータ マップデータ | sharpened map | ||||||||||||
 試料 試料 |
| ||||||||||||
 キーワード キーワード | Microbacterium ketosireducens TIR SAVED complex bound to cA3 / SIGNALING PROTEIN | ||||||||||||
| 機能・相同性 | SMODS-associated and fused to various effectors / SMODS-associated and fused to various effectors sensor domain / TIR domain / Toll/interleukin-1 receptor homology (TIR) domain / Toll/interleukin-1 receptor homology (TIR) domain superfamily / signal transduction / TIR domain-containing protein 機能・相同性情報 機能・相同性情報 | ||||||||||||
| 生物種 |  Microbacterium ketosireducens (バクテリア) Microbacterium ketosireducens (バクテリア) | ||||||||||||
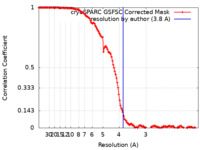
| 手法 | 単粒子再構成法 / クライオ電子顕微鏡法 / 解像度: 3.8 Å | ||||||||||||
 データ登録者 データ登録者 | Spagnolo L / White MF / Hogrel G | ||||||||||||
| 資金援助 |  英国, 3件 英国, 3件
| ||||||||||||
 引用 引用 |  ジャーナル: Nature / 年: 2022 ジャーナル: Nature / 年: 2022タイトル: Cyclic nucleotide-induced helical structure activates a TIR immune effector. 著者: Gaëlle Hogrel / Abbie Guild / Shirley Graham / Hannah Rickman / Sabine Grüschow / Quentin Bertrand / Laura Spagnolo / Malcolm F White /    要旨: Cyclic nucleotide signalling is a key component of antiviral defence in all domains of life. Viral detection activates a nucleotide cyclase to generate a second messenger, resulting in activation of ...Cyclic nucleotide signalling is a key component of antiviral defence in all domains of life. Viral detection activates a nucleotide cyclase to generate a second messenger, resulting in activation of effector proteins. This is exemplified by the metazoan cGAS-STING innate immunity pathway, which originated in bacteria. These defence systems require a sensor domain to bind the cyclic nucleotide and are often coupled with an effector domain that, when activated, causes cell death by destroying essential biomolecules. One example is the Toll/interleukin-1 receptor (TIR) domain, which degrades the essential cofactor NAD when activated in response to infection in plants and bacteria or during programmed nerve cell death. Here we show that a bacterial antiviral defence system generates a cyclic tri-adenylate that binds to a TIR-SAVED effector, acting as the 'glue' to allow assembly of an extended superhelical solenoid structure. Adjacent TIR subunits interact to organize and complete a composite active site, allowing NAD degradation. Activation requires extended filament formation, both in vitro and in vivo. Our study highlights an example of large-scale molecular assembly controlled by cyclic nucleotides and reveals key details of the mechanism of TIR enzyme activation. | ||||||||||||
| 履歴 |
|
- 構造の表示
構造の表示
| 添付画像 |
|---|
- ダウンロードとリンク
ダウンロードとリンク
-EMDBアーカイブ
| マップデータ |  emd_14122.map.gz emd_14122.map.gz | 230 MB |  EMDBマップデータ形式 EMDBマップデータ形式 | |
|---|---|---|---|---|
| ヘッダ (付随情報) |  emd-14122-v30.xml emd-14122-v30.xml emd-14122.xml emd-14122.xml | 23.6 KB 23.6 KB | 表示 表示 |  EMDBヘッダ EMDBヘッダ |
| FSC (解像度算出) |  emd_14122_fsc.xml emd_14122_fsc.xml | 13.2 KB | 表示 |  FSCデータファイル FSCデータファイル |
| 画像 |  emd_14122.png emd_14122.png | 70.8 KB | ||
| マスクデータ |  emd_14122_msk_1.map emd_14122_msk_1.map | 244.1 MB |  マスクマップ マスクマップ | |
| Filedesc metadata |  emd-14122.cif.gz emd-14122.cif.gz | 6.6 KB | ||
| その他 |  emd_14122_additional_1.map.gz emd_14122_additional_1.map.gz emd_14122_additional_2.map.gz emd_14122_additional_2.map.gz emd_14122_half_map_1.map.gz emd_14122_half_map_1.map.gz emd_14122_half_map_2.map.gz emd_14122_half_map_2.map.gz | 4.8 MB 120.4 MB 226.3 MB 226.3 MB | ||
| アーカイブディレクトリ |  http://ftp.pdbj.org/pub/emdb/structures/EMD-14122 http://ftp.pdbj.org/pub/emdb/structures/EMD-14122 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-14122 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-14122 | HTTPS FTP |
-検証レポート
| 文書・要旨 |  emd_14122_validation.pdf.gz emd_14122_validation.pdf.gz | 1.2 MB | 表示 |  EMDB検証レポート EMDB検証レポート |
|---|---|---|---|---|
| 文書・詳細版 |  emd_14122_full_validation.pdf.gz emd_14122_full_validation.pdf.gz | 1.2 MB | 表示 | |
| XML形式データ |  emd_14122_validation.xml.gz emd_14122_validation.xml.gz | 21.5 KB | 表示 | |
| CIF形式データ |  emd_14122_validation.cif.gz emd_14122_validation.cif.gz | 27.3 KB | 表示 | |
| アーカイブディレクトリ |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-14122 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-14122 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-14122 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-14122 | HTTPS FTP |
-関連構造データ
| 関連構造データ |  7qqkMC M: このマップから作成された原子モデル C: 同じ文献を引用 ( |
|---|---|
| 類似構造データ | 類似検索 - 機能・相同性  F&H 検索 F&H 検索 |
- リンク
リンク
| EMDBのページ |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| 「今月の分子」の関連する項目 |
- マップ
マップ
| ファイル |  ダウンロード / ファイル: emd_14122.map.gz / 形式: CCP4 / 大きさ: 244.1 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) ダウンロード / ファイル: emd_14122.map.gz / 形式: CCP4 / 大きさ: 244.1 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | sharpened map | ||||||||||||||||||||||||||||||||||||
| 投影像・断面図 | 画像のコントロール
画像は Spider により作成 | ||||||||||||||||||||||||||||||||||||
| ボクセルのサイズ | X=Y=Z: 0.997 Å | ||||||||||||||||||||||||||||||||||||
| 密度 |
| ||||||||||||||||||||||||||||||||||||
| 対称性 | 空間群: 1 | ||||||||||||||||||||||||||||||||||||
| 詳細 | EMDB XML:
|
-添付データ
-マスク #1
| ファイル |  emd_14122_msk_1.map emd_14122_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 投影像・断面図 |
| ||||||||||||
| 密度ヒストグラム |
-追加マップ: local filter map
| ファイル | emd_14122_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | local filter map | ||||||||||||
| 投影像・断面図 |
| ||||||||||||
| 密度ヒストグラム |
-追加マップ: unsharpened map
| ファイル | emd_14122_additional_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | unsharpened map | ||||||||||||
| 投影像・断面図 |
| ||||||||||||
| 密度ヒストグラム |
-ハーフマップ: half mapB
| ファイル | emd_14122_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | half mapB | ||||||||||||
| 投影像・断面図 |
| ||||||||||||
| 密度ヒストグラム |
-ハーフマップ: half map A
| ファイル | emd_14122_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | half map A | ||||||||||||
| 投影像・断面図 |
| ||||||||||||
| 密度ヒストグラム |
- 試料の構成要素
試料の構成要素
-全体 : one tier of TIR_SAVED bound to cA3
| 全体 | 名称: one tier of TIR_SAVED bound to cA3 |
|---|---|
| 要素 |
|
-超分子 #1: one tier of TIR_SAVED bound to cA3
| 超分子 | 名称: one tier of TIR_SAVED bound to cA3 / タイプ: complex / ID: 1 / 親要素: 0 / 含まれる分子: all |
|---|---|
| 由来(天然) | 生物種:  Microbacterium ketosireducens (バクテリア) Microbacterium ketosireducens (バクテリア) |
-分子 #1: TIR_SAVED fusion protein
| 分子 | 名称: TIR_SAVED fusion protein / タイプ: protein_or_peptide / ID: 1 / コピー数: 4 / 光学異性体: LEVO |
|---|---|
| 由来(天然) | 生物種:  Microbacterium ketosireducens (バクテリア) Microbacterium ketosireducens (バクテリア) |
| 分子量 | 理論値: 46.815875 KDa |
| 組換発現 | 生物種:  |
| 配列 | 文字列: MPDTAINPRD PVFVSYRHSD GIALAAELTW LLRAAGIPVW RDVDDLPPGD TDARLQQAID EGISGAVIII TPQIADSRVV REVEAPRLL RLHRSSPQFA LGIVNAIQTS TGVVDYDAPD RVLGMERPEL RSVDQKSASR LGLVTMARQM LWHRIAAIRP L LSASGGEL ...文字列: MPDTAINPRD PVFVSYRHSD GIALAAELTW LLRAAGIPVW RDVDDLPPGD TDARLQQAID EGISGAVIII TPQIADSRVV REVEAPRLL RLHRSSPQFA LGIVNAIQTS TGVVDYDAPD RVLGMERPEL RSVDQKSASR LGLVTMARQM LWHRIAAIRP L LSASGGEL RLSLQTRNTP QVYDRTDADL DIRIRPSAHE KLPSAHGLED FAETAQFLPD AVTRAGANGV RIEGGAHLSV SI AIGAAIP STRVGPMTVV DGRGVHWVSS TEPQLPDEPR LRIVRESTIP STAPAPGRPD VAAYIDLQHP RSDAAFDNYL TEH AAELVA WQHLAPTRTG LLDAADGGTI AAEAVAHIRE LSMTNGNAVV HLMVRGPFGL AVLIGRLTNT LRVVAYEWTD SDAP DGTFM PPRYEPIVQL RASTPAGVIE RVIVADAE UniProtKB: TIR domain-containing protein |
-分子 #2: RNA (5'-R(P*AP*AP*A)-3')
| 分子 | 名称: RNA (5'-R(P*AP*AP*A)-3') / タイプ: rna / ID: 2 / コピー数: 4 |
|---|---|
| 由来(天然) | 生物種:  Microbacterium ketosireducens (バクテリア) Microbacterium ketosireducens (バクテリア) |
| 分子量 | 理論値: 942.66 Da |
| 配列 | 文字列: AAA |
-実験情報
-構造解析
| 手法 | クライオ電子顕微鏡法 |
|---|---|
 解析 解析 | 単粒子再構成法 |
| 試料の集合状態 | particle |
- 試料調製
試料調製
| 緩衝液 | pH: 7.5 |
|---|---|
| 凍結 | 凍結剤: ETHANE |
- 電子顕微鏡法
電子顕微鏡法
| 顕微鏡 | JEOL CRYO ARM 300 |
|---|---|
| 撮影 | フィルム・検出器のモデル: DIRECT ELECTRON DE-64 (8k x 8k) 平均電子線量: 46.0 e/Å2 |
| 電子線 | 加速電圧: 300 kV / 電子線源:  FIELD EMISSION GUN FIELD EMISSION GUN |
| 電子光学系 | 照射モード: FLOOD BEAM / 撮影モード: BRIGHT FIELD / 最大 デフォーカス(公称値): 5.0 µm / 最小 デフォーカス(公称値): 1.5 µm |
 ムービー
ムービー コントローラー
コントローラー





 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)