[English] 日本語
 Yorodumi
Yorodumi- EMDB-13387: Low resolution Cryo-EM structure of the full-length insulin recep... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-13387 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Low resolution Cryo-EM structure of the full-length insulin receptor bound to 3 insulin, conf 2 | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Insulin / Receptor / Complex / MEMBRANE PROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationregulation of female gonad development / positive regulation of meiotic cell cycle / insulin-like growth factor II binding / positive regulation of developmental growth / male sex determination / insulin receptor complex / insulin-like growth factor I binding / insulin receptor activity / positive regulation of protein-containing complex disassembly / exocrine pancreas development ...regulation of female gonad development / positive regulation of meiotic cell cycle / insulin-like growth factor II binding / positive regulation of developmental growth / male sex determination / insulin receptor complex / insulin-like growth factor I binding / insulin receptor activity / positive regulation of protein-containing complex disassembly / exocrine pancreas development / dendritic spine maintenance / insulin binding / adrenal gland development / cargo receptor activity / negative regulation of glycogen catabolic process / : / PTB domain binding / negative regulation of fatty acid metabolic process / Signaling by Insulin receptor / negative regulation of feeding behavior / IRS activation / Insulin processing / regulation of protein secretion / positive regulation of peptide hormone secretion / neuronal cell body membrane / positive regulation of respiratory burst / negative regulation of acute inflammatory response / Regulation of gene expression in beta cells / alpha-beta T cell activation / amyloid-beta clearance / regulation of embryonic development / positive regulation of receptor internalization / insulin receptor substrate binding / Synthesis, secretion, and deacylation of Ghrelin / epidermis development / negative regulation of protein secretion / positive regulation of dendritic spine maintenance / negative regulation of gluconeogenesis / fatty acid homeostasis / positive regulation of glycogen biosynthetic process / positive regulation of insulin receptor signaling pathway / Signal attenuation / FOXO-mediated transcription of oxidative stress, metabolic and neuronal genes / protein kinase activator activity / negative regulation of respiratory burst involved in inflammatory response / negative regulation of lipid catabolic process / positive regulation of lipid biosynthetic process / negative regulation of oxidative stress-induced intrinsic apoptotic signaling pathway / nitric oxide-cGMP-mediated signaling / transport across blood-brain barrier / phosphatidylinositol 3-kinase binding / heart morphogenesis / regulation of protein localization to plasma membrane / positive regulation of nitric-oxide synthase activity / transport vesicle / Insulin receptor recycling / COPI-mediated anterograde transport / negative regulation of reactive oxygen species biosynthetic process / positive regulation of brown fat cell differentiation / insulin-like growth factor receptor binding / NPAS4 regulates expression of target genes / neuron projection maintenance / endoplasmic reticulum-Golgi intermediate compartment membrane / positive regulation of mitotic nuclear division / Insulin receptor signalling cascade / receptor-mediated endocytosis / dendrite membrane / positive regulation of glycolytic process / positive regulation of cytokine production / endosome lumen / acute-phase response / positive regulation of D-glucose import across plasma membrane / insulin receptor binding / positive regulation of long-term synaptic potentiation / learning / positive regulation of protein secretion / positive regulation of cell differentiation / wound healing / Regulation of insulin secretion / hormone activity / receptor protein-tyrosine kinase / positive regulation of neuron projection development / negative regulation of protein catabolic process / receptor internalization / regulation of synaptic plasticity / caveola / positive regulation of protein localization to nucleus / Golgi lumen / male gonad development / cellular response to growth factor stimulus / cognition / vasodilation / glucose metabolic process / cellular response to insulin stimulus / memory / positive regulation of nitric oxide biosynthetic process / insulin receptor signaling pathway / protein autophosphorylation / late endosome / cell-cell signaling Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
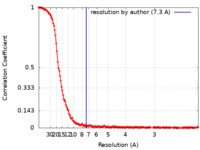
| Method | single particle reconstruction / cryo EM / Resolution: 7.3 Å | |||||||||
 Authors Authors | Nielsen JA / Slaaby R | |||||||||
| Funding support |  Denmark, 1 items Denmark, 1 items
| |||||||||
 Citation Citation |  Journal: J Mol Biol / Year: 2022 Journal: J Mol Biol / Year: 2022Title: Structural Investigations of Full-Length Insulin Receptor Dynamics and Signalling. Authors: Jeppe Nielsen / Jakob Brandt / Thomas Boesen / Tina Hummelshøj / Rita Slaaby / Gerd Schluckebier / Poul Nissen /  Abstract: Insulin regulates glucose homeostasis via binding and activation of the insulin receptor dimer at two distinct pairs of binding sites 1 and 2. Here, we present cryo-EM studies of full-length human ...Insulin regulates glucose homeostasis via binding and activation of the insulin receptor dimer at two distinct pairs of binding sites 1 and 2. Here, we present cryo-EM studies of full-length human insulin receptor (hIR) in an active state obtained at non-saturating, physiologically relevant insulin conditions. Insulin binds asymmetrically to the receptor under these conditions, occupying up to three of the four possible binding sites. Deletion analysis of the receptor together with site specific peptides and insulin analogs used in binding studies show that both sites 1 and 2 are required for high insulin affinity. We identify a homotypic interaction of the fibronectin type III domain (FnIII-3) of IR resulting in tight interaction of membrane proximal domains of the active, asymmetric receptor dimer. Our results show how insulin binding at two distinct types of sites disrupts the autoinhibited apo-IR dimer and stabilizes the active dimer. We propose an insulin binding and activation mechanism, which is sequential, exhibits negative cooperativity, and is based on asymmetry at physiological insulin concentrations with one to three insulin molecules activating IR. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_13387.map.gz emd_13387.map.gz | 143.1 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-13387-v30.xml emd-13387-v30.xml emd-13387.xml emd-13387.xml | 18.6 KB 18.6 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_13387_fsc.xml emd_13387_fsc.xml | 19.4 KB | Display |  FSC data file FSC data file |
| Images |  emd_13387.png emd_13387.png | 64.2 KB | ||
| Masks |  emd_13387_msk_1.map emd_13387_msk_1.map | 282.6 MB |  Mask map Mask map | |
| Filedesc metadata |  emd-13387.cif.gz emd-13387.cif.gz | 6.4 KB | ||
| Others |  emd_13387_half_map_1.map.gz emd_13387_half_map_1.map.gz emd_13387_half_map_2.map.gz emd_13387_half_map_2.map.gz | 261.8 MB 261.8 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-13387 http://ftp.pdbj.org/pub/emdb/structures/EMD-13387 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-13387 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-13387 | HTTPS FTP |
-Related structure data
| Related structure data |  7pg3MC  7pg0C  7pg2C  7pg4C M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_13387.map.gz / Format: CCP4 / Size: 282.6 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_13387.map.gz / Format: CCP4 / Size: 282.6 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.0898 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
-Mask #1
| File |  emd_13387_msk_1.map emd_13387_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #1
| File | emd_13387_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #2
| File | emd_13387_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : DDM solubilised full-length human insulin receptor with three ins...
| Entire | Name: DDM solubilised full-length human insulin receptor with three insulins bound |
|---|---|
| Components |
|
-Supramolecule #1: DDM solubilised full-length human insulin receptor with three ins...
| Supramolecule | Name: DDM solubilised full-length human insulin receptor with three insulins bound type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 460 KDa |
-Macromolecule #1: Isoform Short of Insulin receptor
| Macromolecule | Name: Isoform Short of Insulin receptor / type: protein_or_peptide / ID: 1 / Number of copies: 2 / Enantiomer: LEVO / EC number: receptor protein-tyrosine kinase |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 156.697578 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MATGGRRGAA AAPLLVAVAA LLLGAAGHLY PGEVCPGMDI RNNLTRLHEL ENCSVIEGHL QILLMFKTRP EDFRDLSFPK LIMITDYLL LFRVYGLESL KDLFPNLTVI RGSRLFFNYA LVIFEMVHLK ELGLYNLMNI TRGSVRIEKN NELCYLATID W SRILDSVE ...String: MATGGRRGAA AAPLLVAVAA LLLGAAGHLY PGEVCPGMDI RNNLTRLHEL ENCSVIEGHL QILLMFKTRP EDFRDLSFPK LIMITDYLL LFRVYGLESL KDLFPNLTVI RGSRLFFNYA LVIFEMVHLK ELGLYNLMNI TRGSVRIEKN NELCYLATID W SRILDSVE DNYIVLNKDD NEECGDICPG TAKGKTNCPA TVINGQFVER CWTHSHCQKV CPTICKSHGC TAEGLCCHSE CL GNCSQPD DPTKCVACRN FYLDGRCVET CPPPYYHFQD WRCVNFSFCQ DLHHKCKNSR RQGCHQYVIH NNKCIPECPS GYT MNSSNL LCTPCLGPCP KVCHLLEGEK TIDSVTSAQE LRGCTVINGS LIINIRGGNN LAAELEANLG LIEEISGYLK IRRS YALVS LSFFRKLRLI RGETLEIGNY SFYALDNQNL RQLWDWSKHN LTITQGKLFF HYNPKLCLSE IHKMEEVSGT KGRQE RNDI ALKTNGDQAS CENELLKFSY IRTSFDKILL RWEPYWPPDF RDLLGFMLFY KEAPYQNVTE FDGQDACGSN SWTVVD IDP PLRSNDPKSQ NHPGWLMRGL KPWTQYAIFV KTLVTFSDER RTYGAKSDII YVQTDATNPS VPLDPISVSN SSSQIIL KW KPPSDPNGNI THYLVFWERQ AEDSELFELD YCLKGLKLPS RTWSPPFESE DSQKHNQSEY EDSAGECCSC PKTDSQIL K ELEESSFRKT FEDYLHNVVF VPRPSRKRRS LGDVGNVTVA VPTVAAFPNT SSTSVPTSPE EHRPFEKVVN KESLVISGL RHFTGYRIEL QACNQDTPEE RCSVAAYVSA RTMPEAKADD IVGPVTHEIF ENNVVHLMWQ EPKEPNGLIV LYEVSYRRYG DEELHLCVS RKHFALERGC RLRGLSPGNY SVRIRATSLA GNGSWTEPTY FYVTDYLDVP SNIAKIIIGP LIFVFLFSVV I GSIYLFLR KRQPDGPLGP LYASSNPEYL SASDVFPCSV YVPDEWEVSR EKITLLRELG QGSFGMVYEG NARDIIKGEA ET RVAVKTV NESASLRERI EFLNEASVMK GFTCHHVVRL LGVVSKGQPT LVVMELMAHG DLKSYLRSLR PEAENNPGRP PPT LQEMIQ MAAEIADGMA YLNAKKFVHR DLAARNCMVA HDFTVKIGDF GMTRDIYETD YYRKGGKGLL PVRWMAPESL KDGV FTTSS DMWSFGVVLW EITSLAEQPY QGLSNEQVLK FVMDGGYLDQ PDNCPERVTD LMRMCWQFNP KMRPTFLEIV NLLKD DLHP SFPEVSFFHS EENKAPESEE LEMEFEDMEN VPLDRSSHCQ REEAGGRDGG SSLGFKRSYE EHIPYTHMNG GKKNGR ILT LPRSNPSEDQ VDPRLIDGK UniProtKB: Insulin receptor |
-Macromolecule #2: Insulin
| Macromolecule | Name: Insulin / type: protein_or_peptide / ID: 2 / Number of copies: 3 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 2.383698 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: GIVEQCCTSI CSLYQLENYC N UniProtKB: Insulin |
-Macromolecule #3: Insulin
| Macromolecule | Name: Insulin / type: protein_or_peptide / ID: 3 / Number of copies: 3 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 3.433953 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: FVNQHLCGSH LVEALYLVCG ERGFFYTPKT UniProtKB: Insulin |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 4.0 mg/mL | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 7.8 Component:
| ||||||||||||
| Grid | Model: C-flat-2/2 / Material: COPPER / Support film - Material: CARBON / Support film - topology: HOLEY ARRAY / Pretreatment - Type: GLOW DISCHARGE | ||||||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 297 K / Instrument: LEICA EM GP / Details: Blotted for 3s prior to plunging. |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Average exposure time: 15.0 sec. / Average electron dose: 60.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.0 µm / Nominal defocus min: 0.5 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Refinement | Protocol: FLEXIBLE FIT |
|---|---|
| Output model |  PDB-7pg3: |
 Movie
Movie Controller
Controller


































 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)