[English] 日本語
 Yorodumi
Yorodumi- EMDB-12913: Nog1-TAP associated immature ribosomal particles from S. cerevisi... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-12913 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Nog1-TAP associated immature ribosomal particles from S. cerevisiae after rpL34 expression shut down, population B | |||||||||
 Map data Map data | Nog1-TAP associated immature ribosomal particles from S. cerevisiae after rpL34 expression shut down, population B, full map | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | ribosomal assembly state / RIBOSOME | |||||||||
| Function / homology |  Function and homology information Function and homology informationpositive regulation of ATP-dependent activity / maturation of 5.8S rRNA from tricistronic rRNA transcript (SSU-rRNA, 5.8S rRNA, LSU-rRNA) / cytosolic large ribosomal subunit assembly / maturation of 5.8S rRNA / cleavage in ITS2 between 5.8S rRNA and LSU-rRNA of tricistronic rRNA transcript (SSU-rRNA, 5.8S rRNA, LSU-rRNA) / SRP-dependent cotranslational protein targeting to membrane / GTP hydrolysis and joining of the 60S ribosomal subunit / ribosomal large subunit binding / preribosome, large subunit precursor / Formation of a pool of free 40S subunits ...positive regulation of ATP-dependent activity / maturation of 5.8S rRNA from tricistronic rRNA transcript (SSU-rRNA, 5.8S rRNA, LSU-rRNA) / cytosolic large ribosomal subunit assembly / maturation of 5.8S rRNA / cleavage in ITS2 between 5.8S rRNA and LSU-rRNA of tricistronic rRNA transcript (SSU-rRNA, 5.8S rRNA, LSU-rRNA) / SRP-dependent cotranslational protein targeting to membrane / GTP hydrolysis and joining of the 60S ribosomal subunit / ribosomal large subunit binding / preribosome, large subunit precursor / Formation of a pool of free 40S subunits / Nonsense Mediated Decay (NMD) independent of the Exon Junction Complex (EJC) / Nonsense Mediated Decay (NMD) enhanced by the Exon Junction Complex (EJC) / ATPase activator activity / L13a-mediated translational silencing of Ceruloplasmin expression / nuclear-transcribed mRNA catabolic process / translational elongation / ribosomal large subunit export from nucleus / ribosomal subunit export from nucleus / maturation of LSU-rRNA / translation initiation factor activity / ribosomal large subunit biogenesis / cytosolic ribosome assembly / maturation of LSU-rRNA from tricistronic rRNA transcript (SSU-rRNA, 5.8S rRNA, LSU-rRNA) / assembly of large subunit precursor of preribosome / macroautophagy / maturation of SSU-rRNA / small-subunit processome / maintenance of translational fidelity / rRNA processing / ATPase binding / ribosomal large subunit assembly / large ribosomal subunit rRNA binding / cytosolic large ribosomal subunit / cytoplasmic translation / negative regulation of translation / rRNA binding / structural constituent of ribosome / ribosome / translation / mRNA binding / GTPase activity / GTP binding / nucleolus / RNA binding / zinc ion binding / nucleoplasm / nucleus / cytoplasm / cytosol Similarity search - Function | |||||||||
| Biological species |    | |||||||||
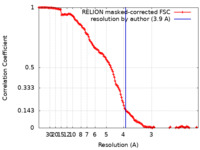
| Method | single particle reconstruction / cryo EM / Resolution: 3.9 Å | |||||||||
 Authors Authors | Milkereit P / Poell G | |||||||||
| Funding support |  Germany, 1 items Germany, 1 items
| |||||||||
 Citation Citation |  Journal: PLoS One / Year: 2021 Journal: PLoS One / Year: 2021Title: Analysis of subunit folding contribution of three yeast large ribosomal subunit proteins required for stabilisation and processing of intermediate nuclear rRNA precursors. Authors: Gisela Pöll / Michael Pilsl / Joachim Griesenbeck / Herbert Tschochner / Philipp Milkereit /  Abstract: In yeast and human cells many of the ribosomal proteins (r-proteins) are required for the stabilisation and productive processing of rRNA precursors. Functional coupling of r-protein assembly with ...In yeast and human cells many of the ribosomal proteins (r-proteins) are required for the stabilisation and productive processing of rRNA precursors. Functional coupling of r-protein assembly with the stabilisation and maturation of subunit precursors potentially promotes the production of ribosomes with defined composition. To further decipher mechanisms of such an intrinsic quality control pathway we analysed here the contribution of three yeast large ribosomal subunit r-proteins rpL2 (uL2), rpL25 (uL23) and rpL34 (eL34) for intermediate nuclear subunit folding steps. Structure models obtained from single particle cryo-electron microscopy analyses provided evidence for specific and hierarchic effects on the stable positioning and remodelling of large ribosomal subunit domains. Based on these structural and previous biochemical data we discuss possible mechanisms of r-protein dependent hierarchic domain arrangement and the resulting impact on the stability of misassembled subunits. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_12913.map.gz emd_12913.map.gz | 193 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-12913-v30.xml emd-12913-v30.xml emd-12913.xml emd-12913.xml | 48.8 KB 48.8 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_12913_fsc.xml emd_12913_fsc.xml | 14.2 KB | Display |  FSC data file FSC data file |
| Images |  emd_12913.png emd_12913.png | 91.9 KB | ||
| Filedesc metadata |  emd-12913.cif.gz emd-12913.cif.gz | 11.7 KB | ||
| Others |  emd_12913_half_map_1.map.gz emd_12913_half_map_1.map.gz emd_12913_half_map_2.map.gz emd_12913_half_map_2.map.gz | 193.7 MB 193.7 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-12913 http://ftp.pdbj.org/pub/emdb/structures/EMD-12913 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-12913 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-12913 | HTTPS FTP |
-Related structure data
| Related structure data |  7ohyMC  7of1C  7oh3C  7ohpC  7ohqC  7ohrC  7ohsC  7ohtC  7ohuC  7ohvC  7ohwC  7ohxC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | |
| EM raw data |  EMPIAR-10775 (Title: Nog1-TAP associated immature ribosomal particles from S. cerevisiae depleted of rpL34 EMPIAR-10775 (Title: Nog1-TAP associated immature ribosomal particles from S. cerevisiae depleted of rpL34Data size: 6.2 TB Data #1: Unaligned multi-frame migrographs of Nog1-TAP associated immature ribosomes depleted of rpL34 [micrographs - multiframe]) |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_12913.map.gz / Format: CCP4 / Size: 244.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_12913.map.gz / Format: CCP4 / Size: 244.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Nog1-TAP associated immature ribosomal particles from S. cerevisiae after rpL34 expression shut down, population B, full map | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.0635 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
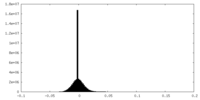
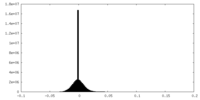
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
-Half map: Nog1-TAP associated immature ribosomal particles from S. cerevisiae...
| File | emd_12913_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Nog1-TAP associated immature ribosomal particles from S. cerevisiae after rpL34 expression shut down, population B, half map 1 | ||||||||||||
| Projections & Slices |
| ||||||||||||
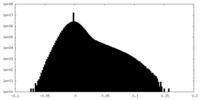
| Density Histograms |
-Half map: Nog1-TAP associated immature ribosomal particles from S. cerevisiae...
| File | emd_12913_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Nog1-TAP associated immature ribosomal particles from S. cerevisiae after rpL34 expression shut down, population B, half map 2 | ||||||||||||
| Projections & Slices |
| ||||||||||||
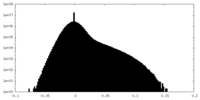
| Density Histograms |
- Sample components
Sample components
+Entire : Nog1-TAP associated immature ribosomal particles from cells deple...
+Supramolecule #1: Nog1-TAP associated immature ribosomal particles from cells deple...
+Macromolecule #1: 25S rRNA
+Macromolecule #2: 5.8S rRNA
+Macromolecule #3: 60S ribosomal protein L3
+Macromolecule #4: 60S ribosomal protein L4-A
+Macromolecule #5: 60S ribosomal protein L6-A
+Macromolecule #6: 60S ribosomal protein L7-A
+Macromolecule #7: 60S ribosomal protein L8-A
+Macromolecule #8: 60S ribosomal protein L9-A
+Macromolecule #9: 60S ribosomal protein L13-A
+Macromolecule #10: 60S ribosomal protein L14-A
+Macromolecule #11: 60S ribosomal protein L15-A
+Macromolecule #12: 60S ribosomal protein L16-A
+Macromolecule #13: 60S ribosomal protein L17-A
+Macromolecule #14: 60S ribosomal protein L18-A
+Macromolecule #15: 60S ribosomal protein L20-A
+Macromolecule #16: 60S ribosomal protein L23-A
+Macromolecule #17: Ribosome assembly factor MRT4
+Macromolecule #18: 60S ribosomal protein L26-A
+Macromolecule #19: Nucleolar GTP-binding protein 1
+Macromolecule #20: 60S ribosomal protein L32
+Macromolecule #21: 60S ribosomal protein L33-A
+Macromolecule #22: 60S ribosomal protein L35-A
+Macromolecule #23: 60S ribosomal protein L37-A
+Macromolecule #24: Ribosome biogenesis protein NSA2
+Macromolecule #25: Ribosome biogenesis protein RLP24
+Macromolecule #26: Eukaryotic translation initiation factor 6
+Macromolecule #27: ZINC ION
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 8 Component:
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Grid | Model: Quantifoil R1.2/1.3 / Material: COPPER / Mesh: 300 / Support film - Material: CARBON / Support film - topology: HOLEY / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 200 sec. / Pretreatment - Atmosphere: AIR / Pretreatment - Pressure: 0.4 kPa | ||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 277 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: FEI FALCON III (4k x 4k) / Detector mode: INTEGRATING / Average exposure time: 5.16 sec. / Average electron dose: 88.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 70.0 µm / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller






























 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)