[English] 日本語
 Yorodumi
Yorodumi- PDB-9l1b: Structure of taurine-bound human Taurine Transporter in the inwar... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 9l1b | |||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Structure of taurine-bound human Taurine Transporter in the inward-occluded conformation | |||||||||||||||||||||||||||
 Components Components | Sodium- and chloride-dependent taurine transporter | |||||||||||||||||||||||||||
 Keywords Keywords | TRANSPORT PROTEIN / cancer / taurine | |||||||||||||||||||||||||||
| Function / homology |  Function and homology information Function and homology informationtaurine transmembrane transporter activity / alanine transmembrane transporter activity / gamma-aminobutyric acid transmembrane transporter activity / taurine:sodium symporter activity / import across plasma membrane / gamma-aminobutyric acid import / alanine transport / gamma-aminobutyric acid:sodium:chloride symporter activity / amino acid:sodium symporter activity / taurine transmembrane transport ...taurine transmembrane transporter activity / alanine transmembrane transporter activity / gamma-aminobutyric acid transmembrane transporter activity / taurine:sodium symporter activity / import across plasma membrane / gamma-aminobutyric acid import / alanine transport / gamma-aminobutyric acid:sodium:chloride symporter activity / amino acid:sodium symporter activity / taurine transmembrane transport / amino acid import across plasma membrane / Amino acid transport across the plasma membrane / amino acid transmembrane transporter activity / SLC-mediated transport of neurotransmitters / microvillus membrane / neurotransmitter transport / amino acid transport / transport across blood-brain barrier / cell projection / sodium ion transmembrane transport / positive regulation of cell differentiation / modulation of chemical synaptic transmission / GABA-ergic synapse / basolateral plasma membrane / postsynaptic membrane / apical plasma membrane / neuronal cell body / dendrite / membrane / plasma membrane Similarity search - Function | |||||||||||||||||||||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||||||||||||||||||||
| Method | ELECTRON MICROSCOPY / single particle reconstruction / cryo EM / Resolution: 2.9 Å | |||||||||||||||||||||||||||
 Authors Authors | Wu, J.X. / Qi, Y. | |||||||||||||||||||||||||||
| Funding support |  China, 1items China, 1items
| |||||||||||||||||||||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2026 Journal: Nat Commun / Year: 2026Title: Structural mechanism of substrate binding and inhibition of human taurine transporter. Authors: Yuhan Qi / Ying Zhang / Duanning Wang / Jiameng Liu / Yue Zhou / Wenming Ji / Xinjing Chen / Luping Liu / Rui Wang / Jing-Xiang Wu /  Abstract: Taurine is a sulfur-containing amino acid that plays several crucial roles in the body. Its uptake is mediated by the taurine transporter (TauT). Genetic mutations and dysregulation of TauT have been ...Taurine is a sulfur-containing amino acid that plays several crucial roles in the body. Its uptake is mediated by the taurine transporter (TauT). Genetic mutations and dysregulation of TauT have been linked to various neurological disorders, cardiomyopathy, childhood progressive retinal degeneration, and cancer, making TauT a promising target for therapeutic intervention in these diseases. However, the structure and mechanism of TauT remain poorly understood. In this study, we present the structures of the human taurine transporter (hTauT) under four conditions: the substrate-free state, the taurine-bound state, the β-alanine-bound state, and the cyclic inhibitor piperidine-4-sulfonate (P4S)-bound state. These structures reveal that taurine binds at the central substrate-binding site of hTauT. Notably, β-alanine and the cyclic P4S inhibitors also mimic taurine, occupying the same substrate-binding site. In the substrate-free and P4S-bound forms, hTauT also adopt an inward-open conformation, where transmembrane helix TM1a bends toward the membrane, facilitating the opening of the intracellular gate for ion and substrate release. These structural insights enhance our understanding of the mechanisms underlying substrate and ion recognition and transport in hTauT, paving the way for the future development of taurine transporter substrate analogues or selective inhibitors. | |||||||||||||||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  9l1b.cif.gz 9l1b.cif.gz | 126.6 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb9l1b.ent.gz pdb9l1b.ent.gz | 96.6 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  9l1b.json.gz 9l1b.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/l1/9l1b https://data.pdbj.org/pub/pdb/validation_reports/l1/9l1b ftp://data.pdbj.org/pub/pdb/validation_reports/l1/9l1b ftp://data.pdbj.org/pub/pdb/validation_reports/l1/9l1b | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  62740MC  9l19C  9l1aC  9l1cC  9l1dC  9l1eC M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
-Protein , 1 types, 1 molecules A
| #1: Protein | Mass: 69881.633 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: SLC6A6 / Cell line (production host): HEK293 / Production host: Homo sapiens (human) / Gene: SLC6A6 / Cell line (production host): HEK293 / Production host:  Homo sapiens (human) / References: UniProt: P31641 Homo sapiens (human) / References: UniProt: P31641 |
|---|
-Non-polymers , 6 types, 8 molecules 

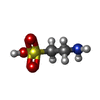








| #2: Chemical | | #3: Chemical | ChemComp-CL / | #4: Chemical | ChemComp-TAU / | #5: Chemical | ChemComp-CLR / | #6: Chemical | #7: Water | ChemComp-HOH / | |
|---|
-Details
| Has ligand of interest | Y |
|---|---|
| Has protein modification | Y |
-Experimental details
-Experiment
| Experiment | Method: ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method: single particle reconstruction |
- Sample preparation
Sample preparation
| Component | Name: TAUT / Type: COMPLEX / Entity ID: #1 / Source: RECOMBINANT |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Source (recombinant) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Buffer solution | pH: 8 |
| Specimen | Embedding applied: NO / Shadowing applied: NO / Staining applied: NO / Vitrification applied: YES |
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Tecnai Polara / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI POLARA 300 |
| Electron gun | Electron source:  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM |
| Electron lens | Mode: BRIGHT FIELD / Calibrated magnification: 64000 X / Nominal defocus max: 2000 nm / Nominal defocus min: 1000 nm |
| Image recording | Electron dose: 50 e/Å2 / Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) |
- Processing
Processing
| EM software |
| ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CTF correction | Type: NONE | ||||||||||||||||
| 3D reconstruction | Resolution: 2.9 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 176066 / Symmetry type: POINT |
 Movie
Movie Controller
Controller







 PDBj
PDBj












