+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 8hpo | ||||||
|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of a SIN3/HDAC complex from budding yeast | ||||||
 Components Components |
| ||||||
 Keywords Keywords | DNA BINDING PROTEIN / SIN3 / HDAC / deacetylase / Rpd3 | ||||||
| Function / homology |  Function and homology information Function and homology information: / regulation of invasive growth in response to glucose limitation / PI5P Regulates TP53 Acetylation / conjugation with cellular fusion / : / Snt2C complex / negative regulation of reciprocal meiotic recombination / negative regulation of silent mating-type cassette heterochromatin formation / positive regulation of invasive growth in response to glucose limitation / invasive growth in response to glucose limitation ...: / regulation of invasive growth in response to glucose limitation / PI5P Regulates TP53 Acetylation / conjugation with cellular fusion / : / Snt2C complex / negative regulation of reciprocal meiotic recombination / negative regulation of silent mating-type cassette heterochromatin formation / positive regulation of invasive growth in response to glucose limitation / invasive growth in response to glucose limitation / protein localization to nucleolar rDNA repeats / negative regulation of rDNA heterochromatin formation / Rpd3S complex / Rpd3L complex / Rpd3L-Expanded complex / regulation of meiotic nuclear division / SUMOylation of transcription cofactors / rDNA chromatin condensation / nucleophagy / HDACs deacetylate histones / NuRD complex / cell adhesion involved in single-species biofilm formation / histone deacetylase / SUMOylation of chromatin organization proteins / regulation of DNA-templated DNA replication initiation / negative regulation of transcription by RNA polymerase I / histone deacetylase activity / Sin3-type complex / NuA4 histone acetyltransferase complex / Estrogen-dependent gene expression / histone deacetylase complex / positive regulation of macroautophagy / Ub-specific processing proteases / heterochromatin formation / histone acetyltransferase activity / methylated histone binding / nuclear periphery / meiotic cell cycle / transcription elongation by RNA polymerase II / transcription coregulator activity / histone deacetylase binding / double-strand break repair via nonhomologous end joining / transcription corepressor activity / nucleosome assembly / chromatin organization / histone binding / response to oxidative stress / transcription coactivator activity / chromatin remodeling / cell cycle / cell division / regulation of DNA-templated transcription / chromatin / regulation of transcription by RNA polymerase II / negative regulation of transcription by RNA polymerase II / positive regulation of transcription by RNA polymerase II / identical protein binding / nucleus / metal ion binding / cytoplasm / cytosol Similarity search - Function | ||||||
| Biological species |  | ||||||
| Method | ELECTRON MICROSCOPY / single particle reconstruction / cryo EM / Resolution: 2.6 Å | ||||||
 Authors Authors | Guo, Z. / Zhan, X. / Wang, C. | ||||||
| Funding support |  China, 1items China, 1items
| ||||||
 Citation Citation |  Journal: Nat Struct Mol Biol / Year: 2023 Journal: Nat Struct Mol Biol / Year: 2023Title: Structure of a SIN3-HDAC complex from budding yeast. Authors: Zhouyan Guo / Chen Chu / Yichen Lu / Xiaofeng Zhang / Yihang Xiao / Mingxuan Wu / Shuaixin Gao / Catherine C L Wong / Xiechao Zhan / Chengcheng Wang /   Abstract: SIN3-HDAC (histone deacetylases) complexes have important roles in facilitating local histone deacetylation to regulate chromatin accessibility and gene expression. Here, we present the cryo-EM ...SIN3-HDAC (histone deacetylases) complexes have important roles in facilitating local histone deacetylation to regulate chromatin accessibility and gene expression. Here, we present the cryo-EM structure of the budding yeast SIN3-HDAC complex Rpd3L at an average resolution of 2.6 Å. The structure reveals that two distinct arms (ARM1 and ARM2) hang on a T-shaped scaffold formed by two coiled-coil domains. In each arm, Sin3 interacts with different subunits to create a different environment for the histone deacetylase Rpd3. ARM1 is in the inhibited state with the active site of Rpd3 blocked, whereas ARM2 is in an open conformation with the active site of Rpd3 exposed to the exterior space. The observed asymmetric architecture of Rpd3L is different from those of available structures of other class I HDAC complexes. Our study reveals the organization mechanism of the SIN3-HDAC complex and provides insights into the interaction pattern by which it targets histone deacetylase to chromatin. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  8hpo.cif.gz 8hpo.cif.gz | 680.8 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb8hpo.ent.gz pdb8hpo.ent.gz | 521.1 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  8hpo.json.gz 8hpo.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Summary document |  8hpo_validation.pdf.gz 8hpo_validation.pdf.gz | 513.1 KB | Display |  wwPDB validaton report wwPDB validaton report |
|---|---|---|---|---|
| Full document |  8hpo_full_validation.pdf.gz 8hpo_full_validation.pdf.gz | 600.1 KB | Display | |
| Data in XML |  8hpo_validation.xml.gz 8hpo_validation.xml.gz | 75.5 KB | Display | |
| Data in CIF |  8hpo_validation.cif.gz 8hpo_validation.cif.gz | 113 KB | Display | |
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/hp/8hpo https://data.pdbj.org/pub/pdb/validation_reports/hp/8hpo ftp://data.pdbj.org/pub/pdb/validation_reports/hp/8hpo ftp://data.pdbj.org/pub/pdb/validation_reports/hp/8hpo | HTTPS FTP |
-Related structure data
| Related structure data |  34935MC M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
-Transcriptional regulatory protein ... , 8 types, 9 molecules KDIABHCEJ
| #1: Protein | Mass: 51075.602 Da / Num. of mol.: 1 / Source method: isolated from a natural source Source: (natural)  References: UniProt: Q03010 | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| #2: Protein | Mass: 37765.277 Da / Num. of mol.: 1 / Source method: isolated from a natural source Source: (natural)  References: UniProt: P40505 | ||||||||
| #5: Protein | Mass: 23144.193 Da / Num. of mol.: 1 / Source method: isolated from a natural source Source: (natural)  References: UniProt: P38429 | ||||||||
| #6: Protein | Mass: 175047.266 Da / Num. of mol.: 2 / Source method: isolated from a natural source Source: (natural)  References: UniProt: P22579 #7: Protein | | Mass: 47035.723 Da / Num. of mol.: 1 / Source method: isolated from a natural source Source: (natural)  References: UniProt: P31385 #8: Protein | | Mass: 37081.430 Da / Num. of mol.: 1 / Source method: isolated from a natural source Source: (natural)  References: UniProt: P50947 #9: Protein | | Mass: 48844.043 Da / Num. of mol.: 1 / Source method: isolated from a natural source Source: (natural)  References: UniProt: P38255 #10: Protein | | Mass: 33851.535 Da / Num. of mol.: 1 / Source method: isolated from a natural source Source: (natural)  References: UniProt: Q07458 |
-Histone deacetylase ... , 2 types, 2 molecules FG
| #3: Protein | Mass: 49361.848 Da / Num. of mol.: 1 / Source method: isolated from a natural source Source: (natural)  References: UniProt: P32561, histone deacetylase |
|---|---|
| #4: Protein | Mass: 48961.957 Da / Num. of mol.: 1 / Source method: isolated from a natural source Source: (natural)  References: UniProt: P32561, histone deacetylase |
-Non-polymers , 3 types, 7 molecules 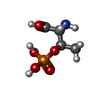




| #11: Chemical | ChemComp-TPO / | ||
|---|---|---|---|
| #12: Chemical | | #13: Chemical | ChemComp-K / |
-Details
| Has ligand of interest | N |
|---|
-Experimental details
-Experiment
| Experiment | Method: ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method: single particle reconstruction |
- Sample preparation
Sample preparation
| Component | Name: The Rpd3L complex / Type: COMPLEX / Entity ID: #1-#10 / Source: MULTIPLE SOURCES |
|---|---|
| Source (natural) | Organism:  |
| Buffer solution | pH: 7.4 |
| Specimen | Embedding applied: NO / Shadowing applied: NO / Staining applied: NO / Vitrification applied: YES |
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TITAN KRIOS |
| Electron gun | Electron source:  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM |
| Electron lens | Mode: BRIGHT FIELD / Nominal defocus max: 2300 nm / Nominal defocus min: 1800 nm |
| Image recording | Electron dose: 50 e/Å2 / Film or detector model: GATAN K3 (6k x 4k) |
- Processing
Processing
| Software | Name: PHENIX / Version: 1.17.1_3660: / Classification: refinement | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CTF correction | Type: NONE | ||||||||||||||||||||||||
| 3D reconstruction | Resolution: 2.6 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 665105 / Symmetry type: POINT | ||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller





 PDBj
PDBj








