+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 8bn3 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Yeast 80S, ES7s delta, eIF5A, Stm1 containing | |||||||||
 Components Components |
| |||||||||
 Keywords Keywords | RIBOSOME / eIF5A / Stm1 / Rpl41A / delta ES7s | |||||||||
| Function / homology |  Function and homology information Function and homology informationpositive regulation of translational termination / ribosomal subunit / positive regulation of translational elongation / regulation of amino acid metabolic process / negative regulation of glucose mediated signaling pathway / translational readthrough / mTORC1-mediated signalling / Protein hydroxylation / positive regulation of protein kinase activity / ribosome-associated ubiquitin-dependent protein catabolic process ...positive regulation of translational termination / ribosomal subunit / positive regulation of translational elongation / regulation of amino acid metabolic process / negative regulation of glucose mediated signaling pathway / translational readthrough / mTORC1-mediated signalling / Protein hydroxylation / positive regulation of protein kinase activity / ribosome-associated ubiquitin-dependent protein catabolic process / pre-mRNA 5'-splice site binding / GDP-dissociation inhibitor activity / cytosolic large ribosomal subunit assembly / nonfunctional rRNA decay / Formation of the ternary complex, and subsequently, the 43S complex / Translation initiation complex formation / response to cycloheximide / Ribosomal scanning and start codon recognition / cleavage in ITS2 between 5.8S rRNA and LSU-rRNA of tricistronic rRNA transcript (SSU-rRNA, 5.8S rRNA, LSU-rRNA) / Major pathway of rRNA processing in the nucleolus and cytosol / mRNA destabilization / SRP-dependent cotranslational protein targeting to membrane / GTP hydrolysis and joining of the 60S ribosomal subunit / negative regulation of mRNA splicing, via spliceosome / preribosome, large subunit precursor / Formation of a pool of free 40S subunits / Nonsense Mediated Decay (NMD) independent of the Exon Junction Complex (EJC) / Nonsense Mediated Decay (NMD) enhanced by the Exon Junction Complex (EJC) / L13a-mediated translational silencing of Ceruloplasmin expression / negative regulation of translational frameshifting / G-protein alpha-subunit binding / ribosomal large subunit export from nucleus / 90S preribosome / endonucleolytic cleavage to generate mature 3'-end of SSU-rRNA from (SSU-rRNA, 5.8S rRNA, LSU-rRNA) / translation elongation factor activity / ribosomal subunit export from nucleus / regulation of translational fidelity / protein-RNA complex assembly / maturation of LSU-rRNA / endonucleolytic cleavage in ITS1 to separate SSU-rRNA from 5.8S rRNA and LSU-rRNA from tricistronic rRNA transcript (SSU-rRNA, 5.8S rRNA, LSU-rRNA) / translation regulator activity / rescue of stalled cytosolic ribosome / cellular response to amino acid starvation / protein kinase C binding / ribosomal large subunit biogenesis / maturation of LSU-rRNA from tricistronic rRNA transcript (SSU-rRNA, 5.8S rRNA, LSU-rRNA) / positive regulation of apoptotic signaling pathway / maturation of SSU-rRNA from tricistronic rRNA transcript (SSU-rRNA, 5.8S rRNA, LSU-rRNA) / macroautophagy / maturation of SSU-rRNA / small-subunit processome / translational initiation / maintenance of translational fidelity / modification-dependent protein catabolic process / protein tag activity / cytoplasmic stress granule / rRNA processing / ribosomal small subunit assembly / ribosome biogenesis / ribosome binding / ribosomal small subunit biogenesis / 5S rRNA binding / small ribosomal subunit / ribosomal large subunit assembly / small ribosomal subunit rRNA binding / cytosolic small ribosomal subunit / large ribosomal subunit rRNA binding / cytosolic large ribosomal subunit / nucleic acid binding / cytoplasmic translation / negative regulation of translation / rRNA binding / structural constituent of ribosome / protein ubiquitination / ribosome / translation / G protein-coupled receptor signaling pathway / ribonucleoprotein complex / negative regulation of gene expression / response to antibiotic / mRNA binding / ubiquitin protein ligase binding / nucleolus / mitochondrion / RNA binding / zinc ion binding / nucleoplasm / metal ion binding / nucleus / cytoplasm / cytosol Similarity search - Function | |||||||||
| Biological species |  | |||||||||
| Method | ELECTRON MICROSCOPY / single particle reconstruction / cryo EM / Resolution: 2.4 Å | |||||||||
 Authors Authors | Dimitrova-Paternoga, L. / Paternoga, H. / Wilson, D.N. | |||||||||
| Funding support |  Germany, Germany,  Switzerland, 2items Switzerland, 2items
| |||||||||
 Citation Citation |  Journal: Nucleic Acids Res / Year: 2024 Journal: Nucleic Acids Res / Year: 2024Title: Evolving precision: rRNA expansion segment 7S modulates translation velocity and accuracy in eukaryal ribosomes. Authors: Robert Rauscher / Cristian Eggers / Lyudmila Dimitrova-Paternoga / Vaishnavi Shankar / Alessia Rosina / Marina Cristodero / Helge Paternoga / Daniel N Wilson / Sebastian A Leidel / Norbert Polacek /   Abstract: Ribosome-enhanced translational miscoding of the genetic code causes protein dysfunction and loss of cellular fitness. During evolution, open reading frame length increased, necessitating mechanisms ...Ribosome-enhanced translational miscoding of the genetic code causes protein dysfunction and loss of cellular fitness. During evolution, open reading frame length increased, necessitating mechanisms for enhanced translation fidelity. Indeed, eukaryal ribosomes are more accurate than bacterial counterparts, despite their virtually identical, conserved active centers. During the evolution of eukaryotic organisms ribosome expansions at the rRNA and protein level occurred, which potentially increases the options for translation regulation and cotranslational events. Here we tested the hypothesis that ribosomal RNA expansions can modulate the core function of the ribosome, faithful protein synthesis. We demonstrate that a short expansion segment present in all eukaryotes' small subunit, ES7S, is crucial for accurate protein synthesis as its presence adjusts codon-specific velocities and guarantees high levels of cognate tRNA selection. Deletion of ES7S in yeast enhances mistranslation and causes protein destabilization and aggregation, dramatically reducing cellular fitness. Removal of ES7S did not alter ribosome architecture but altered the structural dynamics of inter-subunit bridges thus affecting A-tRNA selection. Exchanging the yeast ES7S sequence with the human ES7S increases accuracy whereas shortening causes the opposite effect. Our study demonstrates that ES7S provided eukaryal ribosomes with higher accuracy without perturbing the structurally conserved decoding center. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  8bn3.cif.gz 8bn3.cif.gz | 5.2 MB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb8bn3.ent.gz pdb8bn3.ent.gz | Display |  PDB format PDB format | |
| PDBx/mmJSON format |  8bn3.json.gz 8bn3.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/bn/8bn3 https://data.pdbj.org/pub/pdb/validation_reports/bn/8bn3 ftp://data.pdbj.org/pub/pdb/validation_reports/bn/8bn3 ftp://data.pdbj.org/pub/pdb/validation_reports/bn/8bn3 | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  16127MC M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
+60S ribosomal protein ... , 30 types, 30 molecules BPL2L3L6L7L8M3M4M6M7M8M9N0N1N2N3N5N7N8N9O0O1O3O5O6O9Q0Q2Q3
+Protein , 21 types, 21 molecules S5C5D0D8S2C7D2D6D9L4L5L9M0M1M5N4O2O8SMeIE1
-Small ribosomal subunit protein ... , 9 types, 9 molecules C0S0S1S6S9C1D1SRS4
| #3: Protein | Mass: 11585.125 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
|---|---|
| #15: Protein | Mass: 27920.133 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #16: Protein | Mass: 24312.199 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #19: Protein | Mass: 25895.072 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #22: Protein | Mass: 21210.662 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #23: Protein | Mass: 17668.766 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #28: Protein | Mass: 9758.829 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #36: Protein | Mass: 34710.023 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #80: Protein | Mass: 29338.133 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
-40S ribosomal protein ... , 13 types, 13 molecules C4C8D5S3S7S8C3C6C9D3D4D7E0
| #4: Protein | Mass: 13446.426 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
|---|---|
| #6: Protein | Mass: 16940.443 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #8: Protein | Mass: 8001.377 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #18: Protein | Mass: 24702.791 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #20: Protein | Mass: 21000.492 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #21: Protein | Mass: 21147.180 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #24: Protein | Mass: 16928.748 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #25: Protein | Mass: 15659.216 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #27: Protein | Mass: 15810.930 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #30: Protein | Mass: 15942.699 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #31: Protein | Mass: 15231.650 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #33: Protein | Mass: 8762.195 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #35: Protein | Mass: 6779.086 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
-RNA chain , 4 types, 4 molecules 3412
| #11: RNA chain | Mass: 38951.105 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
|---|---|
| #12: RNA chain | Mass: 50682.922 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #13: RNA chain | Mass: 1022006.562 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #14: RNA chain | Mass: 559592.188 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
-Large ribosomal subunit protein ... , 4 types, 4 molecules N6O4O7Q1
| #60: Protein | Mass: 14134.588 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
|---|---|
| #68: Protein | Mass: 12479.651 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #71: Protein | Mass: 9675.122 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #75: Protein/peptide | Mass: 3354.243 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
-Non-polymers , 6 types, 2113 molecules 

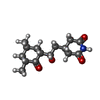








| #82: Chemical | ChemComp-MG / #83: Chemical | ChemComp-K / #84: Chemical | ChemComp-3HE / | #85: Chemical | #86: Chemical | ChemComp-ZN / #87: Water | ChemComp-HOH / | |
|---|
-Details
| Has ligand of interest | Y |
|---|---|
| Has protein modification | Y |
-Experimental details
-Experiment
| Experiment | Method: ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method: single particle reconstruction |
- Sample preparation
Sample preparation
| Component | Name: Dormant yeast 80S ribosome / Type: RIBOSOME Entity ID: #11-#12, #1, #10, #15-#18, #80, #2, #19-#22, #3, #23-#24, #4-#5, #25-#26, #6, #27, #7, #28-#31, #8, #32-#33, #9, #34-#35, #81, #36, #38, #40-#44, #46-#48, #50-#62, #64-#67, #69-#70, #72-#79 Source: NATURAL |
|---|---|
| Molecular weight | Value: 3.3 MDa / Experimental value: NO |
| Source (natural) | Organism:  |
| Buffer solution | pH: 7.5 |
| Specimen | Embedding applied: NO / Shadowing applied: NO / Staining applied: NO / Vitrification applied: YES / Details: 80S peak from Sucrose gradient |
| Specimen support | Grid material: COPPER / Grid mesh size: 300 divisions/in. / Grid type: Quantifoil |
| Vitrification | Cryogen name: ETHANE-PROPANE |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Talos Arctica / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TALOS ARCTICA |
| Electron gun | Electron source:  FIELD EMISSION GUN / Accelerating voltage: 200 kV / Illumination mode: FLOOD BEAM FIELD EMISSION GUN / Accelerating voltage: 200 kV / Illumination mode: FLOOD BEAM |
| Electron lens | Mode: BRIGHT FIELD / Nominal defocus max: 1800 nm / Nominal defocus min: 600 nm |
| Image recording | Electron dose: 32 e/Å2 / Detector mode: COUNTING / Film or detector model: GATAN K2 QUANTUM (4k x 4k) |
| Image scans | Movie frames/image: 40 |
- Processing
Processing
| EM software |
| ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CTF correction | Type: PHASE FLIPPING AND AMPLITUDE CORRECTION | ||||||||||||||||||||||||||||
| Particle selection | Num. of particles selected: 254202 | ||||||||||||||||||||||||||||
| 3D reconstruction | Resolution: 2.4 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 127945 / Algorithm: FOURIER SPACE / Num. of class averages: 2 / Symmetry type: POINT | ||||||||||||||||||||||||||||
| Atomic model building | Protocol: RIGID BODY FIT / Target criteria: Correlation coefficient |
 Movie
Movie Controller
Controller



 PDBj
PDBj



































