[English] 日本語
 Yorodumi
Yorodumi- PDB-8b8r: Complex of Echovirus 11 with its attaching receptor decay-acceler... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 8b8r | ||||||
|---|---|---|---|---|---|---|---|
| Title | Complex of Echovirus 11 with its attaching receptor decay-accelerating factor (CD55) | ||||||
 Components Components |
| ||||||
 Keywords Keywords | VIRUS / Echovirus / receptor / DAF / CD55 | ||||||
| Function / homology | SPHINGOSINE Function and homology information Function and homology information | ||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human)  Echovirus E11 Echovirus E11 | ||||||
| Method | ELECTRON MICROSCOPY / single particle reconstruction / cryo EM / Resolution: 3.1 Å | ||||||
 Authors Authors | Stuart, D.I. / Ren, J. / Zhou, D. / Qin, L. | ||||||
| Funding support |  United Kingdom, 1items United Kingdom, 1items
| ||||||
 Citation Citation |  Journal: Viruses / Year: 2022 Journal: Viruses / Year: 2022Title: Switching of Receptor Binding Poses between Closely Related Enteroviruses. Authors: Daming Zhou / Ling Qin / Helen M E Duyvesteyn / Yuguang Zhao / Tzou-Yien Lin / Elizabeth E Fry / Jingshan Ren / Kuan-Ying A Huang / David I Stuart /   Abstract: Echoviruses, for which there are currently no approved vaccines or drugs, are responsible for a range of human diseases, for example echovirus 11 (E11) is a major cause of serious neonatal morbidity ...Echoviruses, for which there are currently no approved vaccines or drugs, are responsible for a range of human diseases, for example echovirus 11 (E11) is a major cause of serious neonatal morbidity and mortality. Decay-accelerating factor (DAF, also known as CD55) is an attachment receptor for E11. Here, we report the structure of the complex of E11 and the full-length ectodomain of DAF (short consensus repeats, SCRs, 1-4) at 3.1 Å determined by cryo-electron microscopy (cryo-EM). SCRs 3 and 4 of DAF interact with E11 at the southern rim of the canyon via the VP2 EF and VP3 BC loops. We also observe an unexpected interaction between the N-linked glycan (residue 95 of DAF) and the VP2 BC loop of E11. DAF is a receptor for at least 20 enteroviruses and we classify its binding patterns from reported DAF/virus complexes into two distinct positions and orientations, named as E6 and E11 poses. Whilst 60 DAF molecules can attach to the virion in the E6 pose, no more than 30 can attach to E11 due to steric restrictions. Analysis of the distinct modes of interaction and structure and sequence-based phylogenies suggests that the two modes evolved independently, with the E6 mode likely found earlier. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  8b8r.cif.gz 8b8r.cif.gz | 213.4 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb8b8r.ent.gz pdb8b8r.ent.gz | 170.3 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  8b8r.json.gz 8b8r.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/b8/8b8r https://data.pdbj.org/pub/pdb/validation_reports/b8/8b8r ftp://data.pdbj.org/pub/pdb/validation_reports/b8/8b8r ftp://data.pdbj.org/pub/pdb/validation_reports/b8/8b8r | HTTPS FTP |
|---|
-Related structure data
| Related structure data | 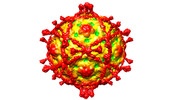 15920MC  8b9fC M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 | x 60
|
- Components
Components
-Protein , 5 types, 5 molecules ABCDE
| #1: Protein | Mass: 32838.828 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)   Echovirus E11 Echovirus E11 |
|---|---|
| #2: Protein | Mass: 28886.428 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)   Echovirus E11 Echovirus E11 |
| #3: Protein | Mass: 26029.674 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)   Echovirus E11 Echovirus E11 |
| #4: Protein | Mass: 7498.273 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)   Echovirus E11 Echovirus E11 |
| #5: Protein | Mass: 29771.059 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Cell line (production host): HEK293T / Production host: Homo sapiens (human) / Cell line (production host): HEK293T / Production host:  Homo sapiens (human) Homo sapiens (human) |
-Non-polymers , 1 types, 1 molecules 
| #6: Chemical | ChemComp-SPH / |
|---|
-Details
| Has ligand of interest | Y |
|---|
-Experimental details
-Experiment
| Experiment | Method: ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method: single particle reconstruction |
- Sample preparation
Sample preparation
| Component | Name: Complex of Echovirus 11 with its attaching receptor DECAY ACCELERATING FACTOR (CD55) Type: COMPLEX / Entity ID: #1-#5 / Source: NATURAL |
|---|---|
| Molecular weight | Experimental value: NO |
| Source (natural) | Organism:   Echovirus E11 Echovirus E11 |
| Buffer solution | pH: 7.4 |
| Specimen | Conc.: 1 mg/ml / Embedding applied: NO / Shadowing applied: NO / Staining applied: NO / Vitrification applied: YES |
| Vitrification | Cryogen name: ETHANE-PROPANE |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TITAN KRIOS |
| Electron gun | Electron source:  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: SPOT SCAN FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: SPOT SCAN |
| Electron lens | Mode: DARK FIELD / Nominal defocus max: 1000 nm / Nominal defocus min: 3000 nm |
| Image recording | Electron dose: 41 e/Å2 / Film or detector model: FEI FALCON IV (4k x 4k) |
- Processing
Processing
| CTF correction | Type: PHASE FLIPPING AND AMPLITUDE CORRECTION |
|---|---|
| 3D reconstruction | Resolution: 3.1 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 6394 / Symmetry type: POINT |
 Movie
Movie Controller
Controller



 PDBj
PDBj

