[English] 日本語
 Yorodumi
Yorodumi- PDB-8a6l: Human 4F2hc-LAT2 heterodimeric amino acid transporter in complex ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 8a6l | ||||||
|---|---|---|---|---|---|---|---|
| Title | Human 4F2hc-LAT2 heterodimeric amino acid transporter in complex with anticalin D11vs | ||||||
 Components Components |
| ||||||
 Keywords Keywords | MEMBRANE PROTEIN / complex / 4F2hc / LAT2 / anticalin | ||||||
| Function / homology |  Function and homology information Function and homology informationproline transmembrane transport / glycine transmembrane transporter activity / organic cation transmembrane transporter activity / glycine transport / thyroid hormone transmembrane transporter activity / amino acid import across plasma membrane / L-leucine import across plasma membrane / L-alanine transmembrane transporter activity / L-alanine import across plasma membrane / L-amino acid transmembrane transporter activity ...proline transmembrane transport / glycine transmembrane transporter activity / organic cation transmembrane transporter activity / glycine transport / thyroid hormone transmembrane transporter activity / amino acid import across plasma membrane / L-leucine import across plasma membrane / L-alanine transmembrane transporter activity / L-alanine import across plasma membrane / L-amino acid transmembrane transporter activity / valine transport / amino acid transmembrane transport / L-leucine transmembrane transporter activity / L-leucine transport / thyroid hormone transport / toxin transmembrane transporter activity / neutral amino acid transport / amino acid transmembrane transporter activity / Amino acid transport across the plasma membrane / neutral L-amino acid transmembrane transporter activity / antiporter activity / Basigin interactions / microvillus membrane / amino acid transport / tryptophan transport / transport across blood-brain barrier / basal plasma membrane / peptide antigen binding / basolateral plasma membrane / apical plasma membrane / protein heterodimerization activity / plasma membrane Similarity search - Function | ||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||
| Method | ELECTRON MICROSCOPY / single particle reconstruction / cryo EM / Resolution: 3.18 Å | ||||||
 Authors Authors | Jeckelmann, J.M. / Fotiadis, D. | ||||||
| Funding support |  Switzerland, 1items Switzerland, 1items
| ||||||
 Citation Citation |  Journal: Sci Rep / Year: 2022 Journal: Sci Rep / Year: 2022Title: Structure of the human heterodimeric transporter 4F2hc-LAT2 in complex with Anticalin, an alternative binding protein for applications in single-particle cryo-EM. Authors: Jean-Marc Jeckelmann / Thomas Lemmin / Martin Schlapschy / Arne Skerra / Dimitrios Fotiadis /   Abstract: Cryo-EM structure determination of relatively small and flexible membrane proteins at high resolution is challenging. Increasing the size and structural features by binding of high affinity proteins ...Cryo-EM structure determination of relatively small and flexible membrane proteins at high resolution is challenging. Increasing the size and structural features by binding of high affinity proteins to the biomolecular target allows for better particle alignment and may result in structural models of higher resolution and quality. Anticalins are alternative binding proteins to antibodies, which are based on the lipocalin scaffold and show potential for theranostic applications. The human heterodimeric amino acid transporter 4F2hc-LAT2 is a membrane protein complex that mediates transport of certain amino acids and derivatives thereof across the plasma membrane. Here, we present and discuss the cryo-EM structure of human 4F2hc-LAT2 in complex with the anticalin D11vs at 3.2 Å resolution. Relative high local map resolution (2.8-3.0 Å) in the LAT2 substrate binding site together with molecular dynamics simulations indicated the presence of fixed water molecules potentially involved in shaping and stabilizing this region. Finally, the presented work expands the application portfolio of anticalins and widens the toolset of binding proteins to promote high-resolution structure solution by single-particle cryo-EM. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  8a6l.cif.gz 8a6l.cif.gz | 362 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb8a6l.ent.gz pdb8a6l.ent.gz | 296.6 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  8a6l.json.gz 8a6l.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/a6/8a6l https://data.pdbj.org/pub/pdb/validation_reports/a6/8a6l ftp://data.pdbj.org/pub/pdb/validation_reports/a6/8a6l ftp://data.pdbj.org/pub/pdb/validation_reports/a6/8a6l | HTTPS FTP |
|---|
-Related structure data
| Related structure data | 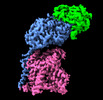 15210MC M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
| #1: Protein | Mass: 60063.934 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Production host: Homo sapiens (human) / Production host:  Komagataella pastoris (fungus) Komagataella pastoris (fungus) | ||||
|---|---|---|---|---|---|
| #2: Protein | Mass: 60121.078 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: SLC7A8, LAT2 / Production host: Homo sapiens (human) / Gene: SLC7A8, LAT2 / Production host:  Komagataella pastoris (fungus) / References: UniProt: Q9UHI5 Komagataella pastoris (fungus) / References: UniProt: Q9UHI5 | ||||
| #3: Protein | Mass: 21327.084 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Production host: Homo sapiens (human) / Production host:  | ||||
| #4: Sugar | ChemComp-NAG / #5: Water | ChemComp-HOH / | Has ligand of interest | Y | |
-Experimental details
-Experiment
| Experiment | Method: ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method: single particle reconstruction |
- Sample preparation
Sample preparation
| Component | Name: Heterodimeric amino acid transporter 4F2hc-LAT2 in complex with anticalin D11vs Type: COMPLEX / Entity ID: #1-#3 / Source: RECOMBINANT | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Molecular weight | Value: 0.15 MDa / Experimental value: NO | ||||||||||||||||||||
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) | ||||||||||||||||||||
| Source (recombinant) | Organism:  Komagataella pastoris (fungus) / Strain: KM71H Komagataella pastoris (fungus) / Strain: KM71H | ||||||||||||||||||||
| Buffer solution | pH: 8 | ||||||||||||||||||||
| Buffer component |
| ||||||||||||||||||||
| Specimen | Conc.: 3 mg/ml / Embedding applied: NO / Shadowing applied: NO / Staining applied: NO / Vitrification applied: YES | ||||||||||||||||||||
| Specimen support | Grid material: COPPER / Grid mesh size: 200 divisions/in. / Grid type: Quantifoil R2/1 | ||||||||||||||||||||
| Vitrification | Instrument: FEI VITROBOT MARK IV / Cryogen name: ETHANE / Humidity: 100 % / Chamber temperature: 277 K |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TITAN KRIOS |
| Electron gun | Electron source:  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: OTHER FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: OTHER |
| Electron lens | Mode: BRIGHT FIELD / Nominal defocus max: 1800 nm / Nominal defocus min: 800 nm |
| Specimen holder | Cryogen: NITROGEN / Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER |
| Image recording | Electron dose: 50 e/Å2 / Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) |
- Processing
Processing
| Software | Name: PHENIX / Version: 1.20_4459: / Classification: refinement | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| EM software |
| ||||||||||||||||||||||||||||||||||||
| CTF correction | Type: PHASE FLIPPING AND AMPLITUDE CORRECTION | ||||||||||||||||||||||||||||||||||||
| Symmetry | Point symmetry: C1 (asymmetric) | ||||||||||||||||||||||||||||||||||||
| 3D reconstruction | Resolution: 3.18 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 552926 / Symmetry type: POINT | ||||||||||||||||||||||||||||||||||||
| Atomic model building | Protocol: FLEXIBLE FIT / Space: REAL | ||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller


 PDBj
PDBj




