+ データを開く
データを開く
- 基本情報
基本情報
| 登録情報 | データベース: PDB / ID: 7un5 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| タイトル | Structure of Type II Prion filaments from Gerstmann-Straussler-Scheinker disease | |||||||||
 要素 要素 | Major prion protein | |||||||||
 キーワード キーワード | PROTEIN FIBRIL / Prion / PrP / GSS / filament / fibril / human brain derived / neurodegenerative | |||||||||
| 機能・相同性 |  機能・相同性情報 機能・相同性情報negative regulation of amyloid precursor protein catabolic process / regulation of glutamate receptor signaling pathway / lamin binding / aspartic-type endopeptidase inhibitor activity / regulation of calcium ion import across plasma membrane / glycosaminoglycan binding / NCAM1 interactions / type 5 metabotropic glutamate receptor binding / positive regulation of glutamate receptor signaling pathway / negative regulation of interleukin-17 production ...negative regulation of amyloid precursor protein catabolic process / regulation of glutamate receptor signaling pathway / lamin binding / aspartic-type endopeptidase inhibitor activity / regulation of calcium ion import across plasma membrane / glycosaminoglycan binding / NCAM1 interactions / type 5 metabotropic glutamate receptor binding / positive regulation of glutamate receptor signaling pathway / negative regulation of interleukin-17 production / cupric ion binding / ATP-dependent protein binding / regulation of potassium ion transmembrane transport / negative regulation of protein processing / negative regulation of dendritic spine maintenance / dendritic spine maintenance / extrinsic component of membrane / negative regulation of calcineurin-NFAT signaling cascade / negative regulation of interleukin-2 production / Insertion of tail-anchored proteins into the endoplasmic reticulum membrane / negative regulation of activated T cell proliferation / negative regulation of amyloid-beta formation / response to amyloid-beta / negative regulation of type II interferon production / cuprous ion binding / negative regulation of long-term synaptic potentiation / intracellular copper ion homeostasis / negative regulation of T cell receptor signaling pathway / positive regulation of protein targeting to membrane / response to cadmium ion / long-term memory / neuron projection maintenance / inclusion body / positive regulation of calcium-mediated signaling / molecular function activator activity / cellular response to copper ion / positive regulation of protein localization to plasma membrane / molecular condensate scaffold activity / tubulin binding / protein homooligomerization / protein destabilization / cellular response to xenobiotic stimulus / cellular response to amyloid-beta / terminal bouton / positive regulation of neuron apoptotic process / amyloid-beta binding / protein-folding chaperone binding / signaling receptor activity / protease binding / response to oxidative stress / nuclear membrane / microtubule binding / molecular adaptor activity / transmembrane transporter binding / learning or memory / regulation of cell cycle / postsynaptic density / intracellular signal transduction / postsynapse / membrane raft / copper ion binding / external side of plasma membrane / dendrite / negative regulation of apoptotic process / protein-containing complex binding / cell surface / negative regulation of transcription by RNA polymerase II / endoplasmic reticulum / Golgi apparatus / extracellular exosome / identical protein binding / plasma membrane / cytoplasm / cytosol 類似検索 - 分子機能 | |||||||||
| 生物種 |  Homo sapiens (ヒト) Homo sapiens (ヒト) | |||||||||
| 手法 | 電子顕微鏡法 / らせん対称体再構成法 / クライオ電子顕微鏡法 / 解像度: 3.13 Å | |||||||||
 データ登録者 データ登録者 | Ozcan, K.A. / Hoq, M.R. / Bharath, S.R. / Jiang, W. | |||||||||
| 資金援助 |  米国, 2件 米国, 2件
| |||||||||
 引用 引用 |  ジャーナル: Acta Neuropathol / 年: 2022 ジャーナル: Acta Neuropathol / 年: 2022タイトル: Cryo-EM structures of prion protein filaments from Gerstmann-Sträussler-Scheinker disease. 著者: Grace I Hallinan / Kadir A Ozcan / Md Rejaul Hoq / Laura Cracco / Frank S Vago / Sakshibeedu R Bharath / Daoyi Li / Max Jacobsen / Emma H Doud / Amber L Mosley / Anllely Fernandez / Holly J ...著者: Grace I Hallinan / Kadir A Ozcan / Md Rejaul Hoq / Laura Cracco / Frank S Vago / Sakshibeedu R Bharath / Daoyi Li / Max Jacobsen / Emma H Doud / Amber L Mosley / Anllely Fernandez / Holly J Garringer / Wen Jiang / Bernardino Ghetti / Ruben Vidal /  要旨: Prion protein (PrP) aggregation and formation of PrP amyloid (APrP) are central events in the pathogenesis of prion diseases. In the dominantly inherited prion protein amyloidosis known as Gerstmann- ...Prion protein (PrP) aggregation and formation of PrP amyloid (APrP) are central events in the pathogenesis of prion diseases. In the dominantly inherited prion protein amyloidosis known as Gerstmann-Sträussler-Scheinker (GSS) disease, plaques made of PrP amyloid are present throughout the brain. The c.593t > c mutation in the prion protein gene (PRNP) results in a phenylalanine to serine amino acid substitution at PrP residue 198 (F198S) and causes the most severe amyloidosis among GSS variants. It has been shown that neurodegeneration in this disease is associated with the presence of extracellular APrP plaques and neuronal intracytoplasmic Tau inclusions, that have been shown to contain paired helical filaments identical to those found in Alzheimer disease. Using cryogenic electron microscopy (cryo-EM), we determined for the first time the structures of filaments of human APrP, isolated post-mortem from the brain of two symptomatic PRNP F198S mutation carriers. We report that in GSS (F198S) APrP filaments are composed of dimeric, trimeric and tetrameric left-handed protofilaments with their protomers sharing a common protein fold. The protomers in the cross-β spines consist of 62 amino acids and span from glycine 80 to phenylalanine 141, adopting a previously unseen spiral fold with a thicker outer layer and a thinner inner layer. Each protomer comprises nine short β-strands, with the β1 and β8 strands, as well as the β4 and β9 strands, forming a steric zipper. The data obtained by cryo-EM provide insights into the structural complexity of the PrP filament in a dominantly inherited human PrP amyloidosis. The novel findings highlight the urgency of extending our knowledge of the filaments' structures that may underlie distinct clinical and pathologic phenotypes of human neurodegenerative diseases. | |||||||||
| 履歴 |
|
- 構造の表示
構造の表示
| 構造ビューア | 分子:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- ダウンロードとリンク
ダウンロードとリンク
- ダウンロード
ダウンロード
| PDBx/mmCIF形式 |  7un5.cif.gz 7un5.cif.gz | 102.9 KB | 表示 |  PDBx/mmCIF形式 PDBx/mmCIF形式 |
|---|---|---|---|---|
| PDB形式 |  pdb7un5.ent.gz pdb7un5.ent.gz | 80.8 KB | 表示 |  PDB形式 PDB形式 |
| PDBx/mmJSON形式 |  7un5.json.gz 7un5.json.gz | ツリー表示 |  PDBx/mmJSON形式 PDBx/mmJSON形式 | |
| その他 |  その他のダウンロード その他のダウンロード |
-検証レポート
| アーカイブディレクトリ |  https://data.pdbj.org/pub/pdb/validation_reports/un/7un5 https://data.pdbj.org/pub/pdb/validation_reports/un/7un5 ftp://data.pdbj.org/pub/pdb/validation_reports/un/7un5 ftp://data.pdbj.org/pub/pdb/validation_reports/un/7un5 | HTTPS FTP |
|---|
-関連構造データ
| 関連構造データ |  26613MC  7umqC M: このデータのモデリングに利用したマップデータ C: 同じ文献を引用 ( |
|---|---|
| 類似構造データ | 類似検索 - 機能・相同性  F&H 検索 F&H 検索 |
- リンク
リンク
- 集合体
集合体
| 登録構造単位 | 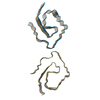
|
|---|---|
| 1 |
|
- 要素
要素
| #1: タンパク質 | 分子量: 6219.043 Da / 分子数: 10 / 由来タイプ: 天然 / 由来: (天然)  Homo sapiens (ヒト) / 器官: Brain / 組織: Cerebellum / 参照: UniProt: P04156 Homo sapiens (ヒト) / 器官: Brain / 組織: Cerebellum / 参照: UniProt: P04156 |
|---|
-実験情報
-実験
| 実験 | 手法: 電子顕微鏡法 |
|---|---|
| EM実験 | 試料の集合状態: FILAMENT / 3次元再構成法: らせん対称体再構成法 |
- 試料調製
試料調製
| 構成要素 | 名称: Type II Prion protein filament from Gerstmann-Straussler-Scheinker disease タイプ: TISSUE / Entity ID: all / 由来: NATURAL | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 由来(天然) | 生物種:  Homo sapiens (ヒト) / 器官: Brain / 組織: Cerebellum Homo sapiens (ヒト) / 器官: Brain / 組織: Cerebellum | ||||||||||||||||
| 緩衝液 | pH: 8 | ||||||||||||||||
| 緩衝液成分 |
| ||||||||||||||||
| 試料 | 包埋: NO / シャドウイング: NO / 染色: NO / 凍結: YES | ||||||||||||||||
| 試料支持 | 詳細: Glow discharged using PELCO easiGlow at 15 mA for 10 seconds. グリッドの材料: COPPER / グリッドのサイズ: 300 divisions/in. / グリッドのタイプ: Quantifoil R1.2/1.3 | ||||||||||||||||
| 急速凍結 | 装置: GATAN CRYOPLUNGE 3 / 凍結剤: ETHANE / 湿度: 95 % / 凍結前の試料温度: 293 K / 詳細: Plunge frozen in a BSL-2 hood |
- 電子顕微鏡撮影
電子顕微鏡撮影
| 実験機器 |  モデル: Titan Krios / 画像提供: FEI Company |
|---|---|
| 顕微鏡 | モデル: TFS KRIOS |
| 電子銃 | 電子線源:  FIELD EMISSION GUN / 加速電圧: 300 kV / 照射モード: FLOOD BEAM FIELD EMISSION GUN / 加速電圧: 300 kV / 照射モード: FLOOD BEAM |
| 電子レンズ | モード: BRIGHT FIELD / 倍率(公称値): 81000 X / 最大 デフォーカス(公称値): 2000 nm / 最小 デフォーカス(公称値): 400 nm / Cs: 2.7 mm |
| 試料ホルダ | 凍結剤: NITROGEN 試料ホルダーモデル: FEI TITAN KRIOS AUTOGRID HOLDER |
| 撮影 | 電子線照射量: 56.5 e/Å2 フィルム・検出器のモデル: GATAN K3 BIOQUANTUM (6k x 4k) 撮影したグリッド数: 1 / 実像数: 8600 |
| 電子光学装置 | エネルギーフィルター名称: GIF Bioquantum / エネルギーフィルタースリット幅: 20 eV |
- 解析
解析
| EMソフトウェア |
| ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CTF補正 | タイプ: PHASE FLIPPING AND AMPLITUDE CORRECTION | ||||||||||||||||||||||||
| らせん対称 | 回転角度/サブユニット: -0.94 ° / 軸方向距離/サブユニット: 4.83 Å / らせん対称軸の対称性: C1 | ||||||||||||||||||||||||
| 3次元再構成 | 解像度: 3.13 Å / 解像度の算出法: FSC 0.143 CUT-OFF / 粒子像の数: 19341 / 対称性のタイプ: HELICAL | ||||||||||||||||||||||||
| 原子モデル構築 | プロトコル: AB INITIO MODEL / 空間: REAL | ||||||||||||||||||||||||
| 精密化 | 交差検証法: NONE 立体化学のターゲット値: GeoStd + Monomer Library + CDL v1.2 | ||||||||||||||||||||||||
| 原子変位パラメータ | Biso mean: 99.08 Å2 | ||||||||||||||||||||||||
| 拘束条件 |
|
 ムービー
ムービー コントローラー
コントローラー




 PDBj
PDBj


