+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-9947 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Structure of ASC CARD filament | |||||||||
 Map data Map data | None | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | filament / SIGNALING PROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationNLRP6 inflammasome complex / myosin I binding / negative regulation of protein serine/threonine kinase activity / Pyrin domain binding / myeloid dendritic cell activation involved in immune response / positive regulation of antigen processing and presentation of peptide antigen via MHC class II / regulation of intrinsic apoptotic signaling pathway / myeloid dendritic cell activation / IkappaB kinase complex / The AIM2 inflammasome ...NLRP6 inflammasome complex / myosin I binding / negative regulation of protein serine/threonine kinase activity / Pyrin domain binding / myeloid dendritic cell activation involved in immune response / positive regulation of antigen processing and presentation of peptide antigen via MHC class II / regulation of intrinsic apoptotic signaling pathway / myeloid dendritic cell activation / IkappaB kinase complex / The AIM2 inflammasome / AIM2 inflammasome complex / icosanoid biosynthetic process / NLRP1 inflammasome complex / macropinocytosis / canonical inflammasome complex / interleukin-6 receptor binding / BMP receptor binding / NLRP3 inflammasome complex / NLRP3 inflammasome complex assembly / positive regulation of adaptive immune response / cysteine-type endopeptidase activator activity / CLEC7A/inflammasome pathway / negative regulation of interferon-beta production / regulation of tumor necrosis factor-mediated signaling pathway / osmosensory signaling pathway / positive regulation of activated T cell proliferation / : / positive regulation of extrinsic apoptotic signaling pathway / positive regulation of macrophage cytokine production / pattern recognition receptor signaling pathway / pattern recognition receptor activity / tropomyosin binding / positive regulation of release of cytochrome c from mitochondria / positive regulation of actin filament polymerization / intrinsic apoptotic signaling pathway by p53 class mediator / positive regulation of interleukin-10 production / The NLRP3 inflammasome / : / pyroptotic inflammatory response / intrinsic apoptotic signaling pathway in response to DNA damage by p53 class mediator / cellular response to interleukin-1 / positive regulation of T cell migration / Purinergic signaling in leishmaniasis infection / positive regulation of defense response to virus by host / positive regulation of chemokine production / negative regulation of cytokine production involved in inflammatory response / activation of innate immune response / intrinsic apoptotic signaling pathway / positive regulation of phagocytosis / tumor necrosis factor-mediated signaling pathway / : / positive regulation of interleukin-1 beta production / positive regulation of interleukin-8 production / regulation of autophagy / negative regulation of canonical NF-kappaB signal transduction / apoptotic signaling pathway / regulation of protein stability / protein homooligomerization / positive regulation of non-canonical NF-kappaB signal transduction / positive regulation of interleukin-6 production / positive regulation of JNK cascade / cellular response to tumor necrosis factor / positive regulation of type II interferon production / positive regulation of T cell activation / positive regulation of tumor necrosis factor production / positive regulation of inflammatory response / SARS-CoV-1 activates/modulates innate immune responses / azurophil granule lumen / cellular response to lipopolysaccharide / protease binding / regulation of inflammatory response / secretory granule lumen / defense response to virus / defense response to Gram-negative bacterium / microtubule / transmembrane transporter binding / protein-macromolecule adaptor activity / positive regulation of canonical NF-kappaB signal transduction / positive regulation of ERK1 and ERK2 cascade / protein dimerization activity / defense response to Gram-positive bacterium / positive regulation of apoptotic process / inflammatory response / Golgi membrane / innate immune response / neuronal cell body / apoptotic process / Neutrophil degranulation / nucleolus / enzyme binding / endoplasmic reticulum / signal transduction / protein homodimerization activity / protein-containing complex / mitochondrion / extracellular region / nucleoplasm / identical protein binding / nucleus / cytoplasm Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | helical reconstruction / cryo EM / Resolution: 4.1 Å | |||||||||
 Authors Authors | Gong Q / Xu C | |||||||||
| Funding support |  Singapore, 1 items Singapore, 1 items
| |||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2021 Journal: Nat Commun / Year: 2021Title: Structural basis for distinct inflammasome complex assembly by human NLRP1 and CARD8. Authors: Qin Gong / Kim Robinson / Chenrui Xu / Phuong Thao Huynh / Kelvin Han Chung Chong / Eddie Yong Jun Tan / Jiawen Zhang / Zhao Zhi Boo / Daniel Eng Thiam Teo / Kenneth Lay / Yaming Zhang / ...Authors: Qin Gong / Kim Robinson / Chenrui Xu / Phuong Thao Huynh / Kelvin Han Chung Chong / Eddie Yong Jun Tan / Jiawen Zhang / Zhao Zhi Boo / Daniel Eng Thiam Teo / Kenneth Lay / Yaming Zhang / John Soon Yew Lim / Wah Ing Goh / Graham Wright / Franklin L Zhong / Bruno Reversade / Bin Wu /   Abstract: Nod-like receptor (NLR) proteins activate pyroptotic cell death and IL-1 driven inflammation by assembling and activating the inflammasome complex. Closely related sensor proteins NLRP1 and CARD8 ...Nod-like receptor (NLR) proteins activate pyroptotic cell death and IL-1 driven inflammation by assembling and activating the inflammasome complex. Closely related sensor proteins NLRP1 and CARD8 undergo unique auto-proteolysis-dependent activation and are implicated in auto-inflammatory diseases; however, their mechanisms of activation are not understood. Here we report the structural basis of how the activating domains (FIIND-CARD) of NLRP1 and CARD8 self-oligomerize to assemble distinct inflammasome complexes. Recombinant FIIND-CARD of NLRP1 forms a two-layered filament, with an inner core of oligomerized CARD surrounded by an outer ring of FIIND. Biochemically, self-assembled NLRP1-CARD filaments are sufficient to drive ASC speck formation in cultured human cells-a process that is greatly enhanced by NLRP1-FIIND which forms oligomers in vitro. The cryo-EM structures of NLRP1-CARD and CARD8-CARD filaments, solved here at 3.7 Å, uncover unique structural features that enable NLRP1 and CARD8 to discriminate between ASC and pro-caspase-1. In summary, our findings provide structural insight into the mechanisms of activation for human NLRP1 and CARD8 and reveal how highly specific signaling can be achieved by heterotypic CARD interactions within the inflammasome complexes. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_9947.map.gz emd_9947.map.gz | 2.5 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-9947-v30.xml emd-9947-v30.xml emd-9947.xml emd-9947.xml | 18.3 KB 18.3 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_9947_fsc.xml emd_9947_fsc.xml | 4.9 KB | Display |  FSC data file FSC data file |
| Images |  emd_9947.png emd_9947.png | 84 KB | ||
| Filedesc metadata |  emd-9947.cif.gz emd-9947.cif.gz | 5.7 KB | ||
| Others |  emd_9947_additional.map.gz emd_9947_additional.map.gz emd_9947_half_map_1.map.gz emd_9947_half_map_1.map.gz emd_9947_half_map_2.map.gz emd_9947_half_map_2.map.gz | 562.2 KB 2.5 MB 2.5 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-9947 http://ftp.pdbj.org/pub/emdb/structures/EMD-9947 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-9947 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-9947 | HTTPS FTP |
-Related structure data
| Related structure data |  6k99MC  9943C  9946C  9948C  6k7vC  6k8jC  6k9fC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_9947.map.gz / Format: CCP4 / Size: 4.9 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_9947.map.gz / Format: CCP4 / Size: 4.9 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | None | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. generated in cubic-lattice coordinate | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.1 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
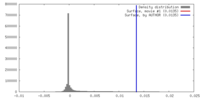
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
-Additional map: cropped 12mer from filament, used for model alignment...
| File | emd_9947_additional.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | cropped 12mer from filament, used for model alignment and refinement, not for publication | ||||||||||||
| Projections & Slices |
| ||||||||||||
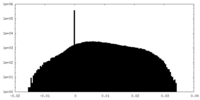
| Density Histograms |
-Half map: #2
| File | emd_9947_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #1
| File | emd_9947_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : ASC CARD filament
| Entire | Name: ASC CARD filament |
|---|---|
| Components |
|
-Supramolecule #1: ASC CARD filament
| Supramolecule | Name: ASC CARD filament / type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: Apoptosis-associated speck-like protein containing a CARD
| Macromolecule | Name: Apoptosis-associated speck-like protein containing a CARD type: protein_or_peptide / ID: 1 / Number of copies: 12 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 9.775143 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: LHFIDQHRAA LIARVTNVEW LLDALYGKVL TDEQYQAVRA EPTNPSKMRK LFSFTPAWNW TCKDLLLQAL RESQSYLVED LER UniProtKB: Apoptosis-associated speck-like protein containing a CARD |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | helical reconstruction |
| Aggregation state | filament |
- Sample preparation
Sample preparation
| Buffer | pH: 8 |
|---|---|
| Grid | Model: Quantifoil R2/2 / Material: COPPER / Mesh: 300 / Support film - Material: CARBON / Support film - topology: CONTINUOUS / Support film - Film thickness: 2 / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 30 sec. / Pretreatment - Atmosphere: AIR |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 277.15 K / Instrument: FEI VITROBOT MARK III |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Number real images: 3183 / Average electron dose: 40.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Calibrated defocus max: 2.5 µm / Calibrated defocus min: 1.5 µm / Illumination mode: FLOOD BEAM / Imaging mode: DARK FIELD / Cs: 2.7 mm / Nominal magnification: 115000 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Refinement | Space: REAL / Protocol: RIGID BODY FIT |
|---|---|
| Output model |  PDB-6k99: |
 Movie
Movie Controller
Controller






















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)