+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-7347 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | CRISPR RNA-guided surveillance complex, pre-nicking | |||||||||
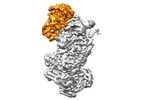
 Map data Map data | CRISPR RNA-guided surveillance complex with Cas3 bound in its pre-nicking state | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | CRISPR-Cas / Cascade / Cas3 / DNA BINDING PROTEIN / DNA BINDING PROTEIN-DNA-RNA complex | |||||||||
| Function / homology |  Function and homology information Function and homology informationnuclease activity / maintenance of CRISPR repeat elements / defense response to virus / RNA helicase activity / RNA binding / ATP binding / identical protein binding / metal ion binding / cytosol Similarity search - Function | |||||||||
| Biological species |   Thermobifida fusca (strain YX) (bacteria) / Thermobifida fusca (strain YX) (bacteria) /   Thermobifida fusca (bacteria) Thermobifida fusca (bacteria) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.66 Å | |||||||||
 Authors Authors | Xiao Y / Luo M | |||||||||
| Funding support |  United States, 2 items United States, 2 items
| |||||||||
 Citation Citation |  Journal: Science / Year: 2018 Journal: Science / Year: 2018Title: Structure basis for RNA-guided DNA degradation by Cascade and Cas3. Authors: Yibei Xiao / Min Luo / Adam E Dolan / Maofu Liao / Ailong Ke /  Abstract: Type I CRISPR-Cas system features a sequential target-searching and degradation process on double-stranded DNA by the RNA-guided Cascade (CRISPR associated complex for antiviral defense) complex and ...Type I CRISPR-Cas system features a sequential target-searching and degradation process on double-stranded DNA by the RNA-guided Cascade (CRISPR associated complex for antiviral defense) complex and the nuclease-helicase fusion enzyme Cas3, respectively. Here, we present a 3.7-angstrom-resolution cryo-electron microscopy (cryo-EM) structure of the Type I-E Cascade/R-loop/Cas3 complex, poised to initiate DNA degradation. Cas3 distinguishes Cascade conformations and only captures the R-loop-forming Cascade, to avoid cleaving partially complementary targets. Its nuclease domain recruits the nontarget strand (NTS) DNA at a bulged region for the nicking of single-stranded DNA. An additional 4.7-angstrom-resolution cryo-EM structure captures the postnicking state, in which the severed NTS retracts to the helicase entrance, to be threaded for adenosine 5'-triphosphate-dependent processive degradation. These snapshots form the basis for understanding RNA-guided DNA degradation in Type I-E CRISPR-Cas systems. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_7347.map.gz emd_7347.map.gz | 60 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-7347-v30.xml emd-7347-v30.xml emd-7347.xml emd-7347.xml | 23.2 KB 23.2 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_7347.png emd_7347.png | 109.4 KB | ||
| Filedesc metadata |  emd-7347.cif.gz emd-7347.cif.gz | 7.8 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-7347 http://ftp.pdbj.org/pub/emdb/structures/EMD-7347 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-7347 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-7347 | HTTPS FTP |
-Validation report
| Summary document |  emd_7347_validation.pdf.gz emd_7347_validation.pdf.gz | 635.8 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_7347_full_validation.pdf.gz emd_7347_full_validation.pdf.gz | 635.4 KB | Display | |
| Data in XML |  emd_7347_validation.xml.gz emd_7347_validation.xml.gz | 6.1 KB | Display | |
| Data in CIF |  emd_7347_validation.cif.gz emd_7347_validation.cif.gz | 7 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-7347 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-7347 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-7347 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-7347 | HTTPS FTP |
-Related structure data
| Related structure data |  6c66MC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_7347.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_7347.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | CRISPR RNA-guided surveillance complex with Cas3 bound in its pre-nicking state | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.24 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
+Entire : CRISPR RNA-guided surveillance complex with Cas3 bound in its pre...
+Supramolecule #1: CRISPR RNA-guided surveillance complex with Cas3 bound in its pre...
+Macromolecule #1: CRISPR-associated helicase, Cas3 family
+Macromolecule #2: CRISPR-associated protein, Cse1 family
+Macromolecule #3: CRISPR-associated protein, Cse4 family
+Macromolecule #4: Uncharacterized protein
+Macromolecule #7: CRISPR-associated protein, Cas5e family
+Macromolecule #9: CRISPR-associated protein, Cse3 family
+Macromolecule #5: crRNA
+Macromolecule #6: Target strand
+Macromolecule #8: Nontarget strand
+Macromolecule #10: FE (III) ION
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 1 mg/mL |
|---|---|
| Buffer | pH: 7.5 / Details: 10 mM HEPES pH 7.5, 150 mM NaCl, 5 mM DTT |
| Grid | Model: Quantifoil R1.2/1.3 / Material: COPPER / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 30 sec. |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 85 % / Instrument: GATAN CRYOPLUNGE 3 |
- Electron microscopy
Electron microscopy
| Microscope | FEI POLARA 300 |
|---|---|
| Temperature | Min: 80.0 K / Max: 105.0 K |
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: COUNTING / Number real images: 1428 / Average electron dose: 8.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Calibrated defocus max: 2.7 µm / Calibrated defocus min: 1.0 µm / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.0 mm / Nominal magnification: 31000 |
| Sample stage | Specimen holder model: GATAN LIQUID NITROGEN / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Tecnai Polara / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Refinement | Protocol: OTHER |
|---|---|
| Output model |  PDB-6c66: |
 Movie
Movie Controller
Controller