+ Open data
Open data
- Basic information
Basic information
| Entry |  | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of psXR | ||||||||||||||||||
 Map data Map data | |||||||||||||||||||
 Sample Sample |
| ||||||||||||||||||
 Keywords Keywords | rhodopsin / inward proton pump / MEMBRANE PROTEIN | ||||||||||||||||||
| Biological species |  Gloeobacter violaceus (bacteria) Gloeobacter violaceus (bacteria) | ||||||||||||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 2.64 Å | ||||||||||||||||||
 Authors Authors | Murakoshi S / Marin MC / Tanaka T / Shihoya W / Beja O / Nureki O | ||||||||||||||||||
| Funding support |  Japan, 5 items Japan, 5 items
| ||||||||||||||||||
 Citation Citation |  Journal: Commun Biol / Year: 2025 Journal: Commun Biol / Year: 2025Title: Light-harvesting by antenna-containing xanthorhodopsin from an Antarctic Pseudanabaenaceae cyanobacterium. Authors: María Del Carmen Marín / Shunya Murakoshi / Andrey Rozenberg / Tatsuki Tanaka / Masae Konno / Wataru Shihoya / Osamu Nureki / Oded Béjà / Keiichi Inoue /    Abstract: Microbial rhodopsins are light-sensitive proteins vital to various phototrophic and sensory processes in microorganisms. Xanthorhodopsins, with their dual chromophore system involving retinal and ...Microbial rhodopsins are light-sensitive proteins vital to various phototrophic and sensory processes in microorganisms. Xanthorhodopsins, with their dual chromophore system involving retinal and carotenoids, have been predominantly studied in the extreme halophilic bacterium Salinibacter ruber and in the early-branching thylakoid-less cyanobacterium Gloeobacter violaceus, where they facilitate light-driven outward proton pumping. However, their distribution, binding specificity, and ecological significance in cyanobacteria remain poorly understood. Here we report the incidence of xanthorhodopsin genes in cyanobacterial genomes and characterize psXR, a xanthorhodopsin from an uncultured Antarctic cyanobacterium from the filamentous family of Pseudanabaenaceae that binds a hydroxylated carotenoid antenna. Through bioinformatic, spectroscopic, functional and structural analyses, we determine the properties of psXR and potential physiological roles of cyanobacterial xanthorhodopsins. Our findings suggest xanthorhodopsins' role in modulating light-harvesting efficiency in cyanobacteria, particularly in extreme environments. The antenna binding and associated structural changes likely provide selective advantages for adapting to polar light conditions such as prolonged low light intensities and spectral shifts, contributing to cyanobacterial survival in harsh habitats. | ||||||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_65924.map.gz emd_65924.map.gz | 36.3 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-65924-v30.xml emd-65924-v30.xml emd-65924.xml emd-65924.xml | 18.9 KB 18.9 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_65924_fsc.xml emd_65924_fsc.xml | 8 KB | Display |  FSC data file FSC data file |
| Images |  emd_65924.png emd_65924.png | 161.2 KB | ||
| Masks |  emd_65924_msk_1.map emd_65924_msk_1.map | 38.4 MB |  Mask map Mask map | |
| Filedesc metadata |  emd-65924.cif.gz emd-65924.cif.gz | 6.2 KB | ||
| Others |  emd_65924_half_map_1.map.gz emd_65924_half_map_1.map.gz emd_65924_half_map_2.map.gz emd_65924_half_map_2.map.gz | 35.6 MB 35.6 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-65924 http://ftp.pdbj.org/pub/emdb/structures/EMD-65924 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-65924 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-65924 | HTTPS FTP |
-Related structure data
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_65924.map.gz / Format: CCP4 / Size: 38.4 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_65924.map.gz / Format: CCP4 / Size: 38.4 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.11 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Mask #1
| File |  emd_65924_msk_1.map emd_65924_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #2
| File | emd_65924_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #1
| File | emd_65924_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Cyanobacterial xanthorhodopsin reconstituted in MSP2N2 nanodiscs ...
| Entire | Name: Cyanobacterial xanthorhodopsin reconstituted in MSP2N2 nanodiscs with soyPC |
|---|---|
| Components |
|
-Supramolecule #1: Cyanobacterial xanthorhodopsin reconstituted in MSP2N2 nanodiscs ...
| Supramolecule | Name: Cyanobacterial xanthorhodopsin reconstituted in MSP2N2 nanodiscs with soyPC type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1 |
|---|---|
| Source (natural) | Organism:  Gloeobacter violaceus (bacteria) Gloeobacter violaceus (bacteria) |
| Molecular weight | Theoretical: 320 KDa |
-Macromolecule #1: psXR
| Macromolecule | Name: psXR / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Gloeobacter violaceus (bacteria) Gloeobacter violaceus (bacteria) |
| Molecular weight | Theoretical: 32.057432 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MSISLITDFA LLAQSNLPET VSKVDFLSSG QYNLVYNAFS FAIATMLAAA LFFFNVRGQV GQKYQLALIV SALVVSIAGY HYYRIFGSW EAAYSLQNGN YVLTGAPFND AYRYVDWLLT VPLLLVETVA VLALPGMEAR PLLIKLVVAA ILMIATGYPG E ISTDLNTR ...String: MSISLITDFA LLAQSNLPET VSKVDFLSSG QYNLVYNAFS FAIATMLAAA LFFFNVRGQV GQKYQLALIV SALVVSIAGY HYYRIFGSW EAAYSLQNGN YVLTGAPFND AYRYVDWLLT VPLLLVETVA VLALPGMEAR PLLIKLVVAA ILMIATGYPG E ISTDLNTR IVWGAVSTVP FIYILYILWI ELSKSLLRQP EGVQTLVKNM RLLLLFSWGV YPIAYLLPML GISGATADVG VQ IGYTIAD VLAKPIFGLL VFAIALEKTR VDAGEQPTKE SPMTMISSHH HHHH |
-Macromolecule #2: RETINAL
| Macromolecule | Name: RETINAL / type: ligand / ID: 2 / Number of copies: 1 / Formula: RET |
|---|---|
| Molecular weight | Theoretical: 284.436 Da |
| Chemical component information |  ChemComp-RET: |
-Macromolecule #3: (3R,3'R,6S)-4,5-DIDEHYDRO-5,6-DIHYDRO-BETA,BETA-CAROTENE-3,3'-DIOL
| Macromolecule | Name: (3R,3'R,6S)-4,5-DIDEHYDRO-5,6-DIHYDRO-BETA,BETA-CAROTENE-3,3'-DIOL type: ligand / ID: 3 / Number of copies: 1 / Formula: LUT |
|---|---|
| Molecular weight | Theoretical: 568.871 Da |
| Chemical component information | 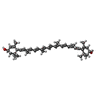 ChemComp-LUT: |
-Macromolecule #4: water
| Macromolecule | Name: water / type: ligand / ID: 4 / Number of copies: 25 / Formula: HOH |
|---|---|
| Molecular weight | Theoretical: 18.015 Da |
| Chemical component information |  ChemComp-HOH: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 8 |
|---|---|
| Grid | Model: Quantifoil R1.2/1.3 / Material: GOLD / Mesh: 300 |
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | TFS KRIOS |
|---|---|
| Specialist optics | Energy filter - Name: GIF Bioquantum / Energy filter - Slit width: 15 eV |
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Number grids imaged: 1 / Number real images: 5949 / Average electron dose: 48.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 1.6 µm / Nominal defocus min: 0.8 µm / Nominal magnification: 105000 |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller





 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)













































