[English] 日本語
 Yorodumi
Yorodumi- EMDB-41456: Escherichia coli RNA polymerase closed complex intermediate at th... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Escherichia coli RNA polymerase closed complex intermediate at the lambda PR promoter | |||||||||
 Map data Map data | EcRNAP-LPR promoter closed complex sharpened map | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | DNA-dependent RNA polymerase / transcription / intermediate / DNA promoter / DNA unwinding / transcription-DNA complex | |||||||||
| Function / homology |  Function and homology information Function and homology informationRNA polymerase complex / submerged biofilm formation / cellular response to cell envelope stress / regulation of DNA-templated transcription initiation / sigma factor activity / bacterial-type flagellum assembly / bacterial-type RNA polymerase core enzyme binding / cytosolic DNA-directed RNA polymerase complex / bacterial-type flagellum-dependent cell motility / nitrate assimilation ...RNA polymerase complex / submerged biofilm formation / cellular response to cell envelope stress / regulation of DNA-templated transcription initiation / sigma factor activity / bacterial-type flagellum assembly / bacterial-type RNA polymerase core enzyme binding / cytosolic DNA-directed RNA polymerase complex / bacterial-type flagellum-dependent cell motility / nitrate assimilation / regulation of DNA-templated transcription elongation / transcription elongation factor complex / DNA-directed RNA polymerase complex / transcription antitermination / DNA-templated transcription initiation / cell motility / ribonucleoside binding / DNA-directed RNA polymerase / DNA-directed RNA polymerase activity / response to heat / protein-containing complex assembly / intracellular iron ion homeostasis / protein dimerization activity / response to antibiotic / DNA-templated transcription / magnesium ion binding / DNA binding / zinc ion binding / membrane / metal ion binding / cytoplasm / cytosol Similarity search - Function | |||||||||
| Biological species |    Escherichia phage Lambda (virus) Escherichia phage Lambda (virus) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.1 Å | |||||||||
 Authors Authors | Darst SA / Saecker RM / Mueller AU | |||||||||
| Funding support |  United States, 1 items United States, 1 items
| |||||||||
 Citation Citation |  Journal: Nat Struct Mol Biol / Year: 2024 Journal: Nat Struct Mol Biol / Year: 2024Title: Early intermediates in bacterial RNA polymerase promoter melting visualized by time-resolved cryo-electron microscopy. Authors: Ruth M Saecker / Andreas U Mueller / Brandon Malone / James Chen / William C Budell / Venkata P Dandey / Kashyap Maruthi / Joshua H Mendez / Nina Molina / Edward T Eng / Laura Y Yen / ...Authors: Ruth M Saecker / Andreas U Mueller / Brandon Malone / James Chen / William C Budell / Venkata P Dandey / Kashyap Maruthi / Joshua H Mendez / Nina Molina / Edward T Eng / Laura Y Yen / Clinton S Potter / Bridget Carragher / Seth A Darst /  Abstract: During formation of the transcription-competent open complex (RPo) by bacterial RNA polymerases (RNAPs), transient intermediates pile up before overcoming a rate-limiting step. Structural ...During formation of the transcription-competent open complex (RPo) by bacterial RNA polymerases (RNAPs), transient intermediates pile up before overcoming a rate-limiting step. Structural descriptions of these interconversions in real time are unavailable. To address this gap, here we use time-resolved cryogenic electron microscopy (cryo-EM) to capture four intermediates populated 120 ms or 500 ms after mixing Escherichia coli σ-RNAP and the λP promoter. Cryo-EM snapshots revealed that the upstream edge of the transcription bubble unpairs rapidly, followed by stepwise insertion of two conserved nontemplate strand (nt-strand) bases into RNAP pockets. As the nt-strand 'read-out' extends, the RNAP clamp closes, expelling an inhibitory σ domain from the active-site cleft. The template strand is fully unpaired by 120 ms but remains dynamic, indicating that yet unknown conformational changes complete RPo formation in subsequent steps. Given that these events likely describe DNA opening at many bacterial promoters, this study provides insights into how DNA sequence regulates steps of RPo formation. #1:  Journal: Nat.Struct.Mol.Biol. / Year: 2024 Journal: Nat.Struct.Mol.Biol. / Year: 2024Title: Early intermediates in bacterial RNA polymerase promoter melting visualized by time-resolved cryo-electron microscopy Authors: Darst SA / Saecker RM / Mueller AU / Malone B / Chen J / Budell WC / Dandey VP / Maruthi K / Mendez JH / Molina N / Eng ET / Yen LY / Potter CS / Carragher B | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_41456.map.gz emd_41456.map.gz | 4.6 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-41456-v30.xml emd-41456-v30.xml emd-41456.xml emd-41456.xml | 39.2 KB 39.2 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_41456.png emd_41456.png | 99.4 KB | ||
| Filedesc metadata |  emd-41456.cif.gz emd-41456.cif.gz | 10.6 KB | ||
| Others |  emd_41456_additional_1.map.gz emd_41456_additional_1.map.gz emd_41456_additional_2.map.gz emd_41456_additional_2.map.gz emd_41456_half_map_1.map.gz emd_41456_half_map_1.map.gz emd_41456_half_map_2.map.gz emd_41456_half_map_2.map.gz | 108.7 MB 204.2 MB 200.5 MB 200.5 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-41456 http://ftp.pdbj.org/pub/emdb/structures/EMD-41456 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-41456 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-41456 | HTTPS FTP |
-Related structure data
| Related structure data |  8tomMC  8to1C  8to6C  8to8C  8toeC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_41456.map.gz / Format: CCP4 / Size: 216 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_41456.map.gz / Format: CCP4 / Size: 216 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | EcRNAP-LPR promoter closed complex sharpened map | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.1 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Additional map: EcRNAP-LPR promoter closed complex unsharpened map
| File | emd_41456_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | EcRNAP-LPR promoter closed complex unsharpened map | ||||||||||||
| Projections & Slices |
| ||||||||||||
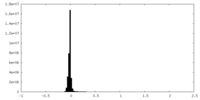
| Density Histograms |
-Additional map: Additional Map 2
| File | emd_41456_additional_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Additional Map 2 | ||||||||||||
| Projections & Slices |
| ||||||||||||
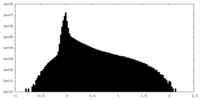
| Density Histograms |
-Half map: EcRNAP-LPR promoter closed complex half map A
| File | emd_41456_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | EcRNAP-LPR promoter closed complex half map A | ||||||||||||
| Projections & Slices |
| ||||||||||||
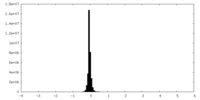
| Density Histograms |
-Half map: EcRNAP-LPR promoter closed complex half map B
| File | emd_41456_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | EcRNAP-LPR promoter closed complex half map B | ||||||||||||
| Projections & Slices |
| ||||||||||||
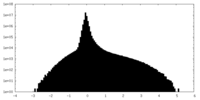
| Density Histograms |
- Sample components
Sample components
+Entire : Escherichia coli RNA polymerase closed complex intermediate at th...
+Supramolecule #1: Escherichia coli RNA polymerase closed complex intermediate at th...
+Macromolecule #1: DNA-directed RNA polymerase subunit alpha
+Macromolecule #2: DNA-directed RNA polymerase subunit beta
+Macromolecule #3: DNA-directed RNA polymerase subunit beta'
+Macromolecule #4: DNA-directed RNA polymerase subunit omega
+Macromolecule #5: RNA polymerase sigma factor RpoD
+Macromolecule #6: Nontemplate strand of lamdba PR promoter DNA
+Macromolecule #7: Template strand of lamdba PR promoter DNA
+Macromolecule #8: CHAPSO
+Macromolecule #9: MAGNESIUM ION
+Macromolecule #10: ZINC ION
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 8 Component:
| |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Grid | Model: C-flat-1.2/1.3 / Material: GOLD / Mesh: 400 / Support film - Material: CARBON / Support film - topology: HOLEY / Pretreatment - Type: PLASMA CLEANING / Pretreatment - Time: 20 sec. / Pretreatment - Atmosphere: AIR | |||||||||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 278.2 K / Instrument: FEI VITROBOT MARK IV Details: CHAPSO was added (from 80 mM stock) to 8 mM final.. | |||||||||||||||
| Details | Es70 RNAP 15 micromolar, LPR DNA 18 micromolar |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Specialist optics | Energy filter - Name: GIF Bioquantum / Energy filter - Slit width: 20 eV |
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: SUPER-RESOLUTION / Average electron dose: 60.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.5 µm / Nominal defocus min: 1.0 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller









 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)