+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | CryoEM structure of Myxococcus xanthus type IV pilus | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | filament / helical reconstruction / CryoEM / CELL ADHESION | |||||||||
| Function / homology |  Function and homology information Function and homology information | |||||||||
| Biological species |  Myxococcus xanthus DK 1622 (bacteria) Myxococcus xanthus DK 1622 (bacteria) | |||||||||
| Method | helical reconstruction / cryo EM / Resolution: 3.0 Å | |||||||||
 Authors Authors | Zheng W / Egelman EH | |||||||||
| Funding support |  United States, 1 items United States, 1 items
| |||||||||
 Citation Citation | Journal: bioRxiv / Year: 2023 Title: Large pilin subunits provide distinct structural and mechanical properties for the type IV pilus. Authors: Anke Treuner-Lange / Weili Zheng / Albertus Viljoen / Steffi Lindow / Marco Herfurth / Yves F Dufrêne / Lotte Søgaard-Andersen / Edward H Egelman Abstract: Type IV pili (T4P) are ubiquitous bacterial cell surface filaments important for surface motility, adhesion to biotic and abiotic surfaces, DNA uptake, biofilm formation, and virulence. T4P are built ...Type IV pili (T4P) are ubiquitous bacterial cell surface filaments important for surface motility, adhesion to biotic and abiotic surfaces, DNA uptake, biofilm formation, and virulence. T4P are built from thousands of copies of the major pilin subunit and tipped by a complex composed of minor pilins and in some systems also the PilY1 adhesin. While the major pilins of structurally characterized T4P have lengths of up to 161 residues, the major pilin PilA of is unusually large with 208 residues. All major pilins have a highly conserved N-terminal domain and a highly variable C-terminal domain, and the additional residues in the PilA are due to a larger C-terminal domain. We solved the structure of the T4P (T4P ) at a resolution of 3.0 Å using cryo-electron microscopy (cryo-EM). The T4P follows the structural blueprint observed in other T4P with the pilus core comprised of the extensively interacting N-terminal α1-helices while the globular domains decorate the T4P surface. The atomic model of PilA built into this map shows that the large C-terminal domain has much more extensive intersubunit contacts than major pilins in other T4P. As expected from these greater contacts, the bending and axial stiffness of the T4P is significantly higher than that of other T4P and supports T4P-dependent motility on surfaces of different stiffnesses. Notably, T4P variants with interrupted intersubunit interfaces had decreased bending stiffness and strongly reduced motility on all surfaces. These observations support an evolutionary scenario whereby the large major pilin enables the formation of a rigid T4P that expands the environmental conditions in which the T4P system functions. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_41298.map.gz emd_41298.map.gz | 6.5 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-41298-v30.xml emd-41298-v30.xml emd-41298.xml emd-41298.xml | 15.1 KB 15.1 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_41298.png emd_41298.png | 104.2 KB | ||
| Filedesc metadata |  emd-41298.cif.gz emd-41298.cif.gz | 5.3 KB | ||
| Others |  emd_41298_half_map_1.map.gz emd_41298_half_map_1.map.gz emd_41298_half_map_2.map.gz emd_41298_half_map_2.map.gz | 59.3 MB 59.3 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-41298 http://ftp.pdbj.org/pub/emdb/structures/EMD-41298 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-41298 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-41298 | HTTPS FTP |
-Related structure data
| Related structure data |  8tj2MC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_41298.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_41298.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.08 Å | ||||||||||||||||||||||||||||||||||||
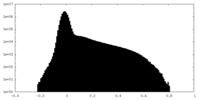
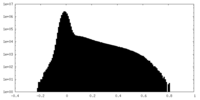
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #1
| File | emd_41298_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
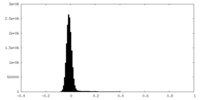
| Density Histograms |
-Half map: #2
| File | emd_41298_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
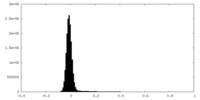
| Density Histograms |
- Sample components
Sample components
-Entire : Myxococcus xanthus type IV pilus
| Entire | Name: Myxococcus xanthus type IV pilus |
|---|---|
| Components |
|
-Supramolecule #1: Myxococcus xanthus type IV pilus
| Supramolecule | Name: Myxococcus xanthus type IV pilus / type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  Myxococcus xanthus DK 1622 (bacteria) Myxococcus xanthus DK 1622 (bacteria) |
-Macromolecule #1: Type IV major pilin protein PilA
| Macromolecule | Name: Type IV major pilin protein PilA / type: protein_or_peptide / ID: 1 / Number of copies: 18 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Myxococcus xanthus DK 1622 (bacteria) Myxococcus xanthus DK 1622 (bacteria) |
| Molecular weight | Theoretical: 21.932318 KDa |
| Sequence | String: FTLIELMIVV AIIGILAAIA IPNFIKFQAR SKQSEAKTNL KALYTAQKSF FSEKDRYSDF ANEIGFAPER GNRYGYRVSA AAGDCEVRN AADLPVPAAG VPCISNDSFR FGANSAIDDP TPVVARFVPQ GAAGWNTTLG VQPTIADCPN CNFFAGARGN A DNEATFDD ...String: FTLIELMIVV AIIGILAAIA IPNFIKFQAR SKQSEAKTNL KALYTAQKSF FSEKDRYSDF ANEIGFAPER GNRYGYRVSA AAGDCEVRN AADLPVPAAG VPCISNDSFR FGANSAIDDP TPVVARFVPQ GAAGWNTTLG VQPTIADCPN CNFFAGARGN A DNEATFDD WVIAGFEGSG QVGPCSEAGN VASGTPYNTR NDVACDGAAQ UniProtKB: Type IV major pilin protein PilA |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | helical reconstruction |
| Aggregation state | filament |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Average electron dose: 55.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.5 µm / Nominal defocus min: 1.0 µm |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Final reconstruction | Applied symmetry - Helical parameters - Δz: 10.0 Å Applied symmetry - Helical parameters - Δ&Phi: 100.7 ° Applied symmetry - Helical parameters - Axial symmetry: C1 (asymmetric) Resolution.type: BY AUTHOR / Resolution: 3.0 Å / Resolution method: FSC 0.143 CUT-OFF / Software - Name: cryoSPARC / Number images used: 1300000 |
|---|---|
| Startup model | Type of model: NONE |
| Final angle assignment | Type: NOT APPLICABLE / Software - Name: cryoSPARC |
 Movie
Movie Controller
Controller





 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)