[English] 日本語
 Yorodumi
Yorodumi- EMDB-36721: Structure of TbAQP2 in complex with anti-trypanosomatid drug mela... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Structure of TbAQP2 in complex with anti-trypanosomatid drug melarsoprol | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Aquaporin / MEMBRANE PROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationglycerol channel activity / urea transmembrane transport / glycerol transmembrane transport / urea transmembrane transporter activity / water channel activity / water transport / membrane / plasma membrane Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) / Homo sapiens (human) /  | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 2.5 Å | |||||||||
 Authors Authors | Chen W / Wang C | |||||||||
| Funding support |  China, 1 items China, 1 items
| |||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2024 Journal: Nat Commun / Year: 2024Title: Structural insights into drug transport by an aquaglyceroporin. Authors: Wanbiao Chen / Rongfeng Zou / Yi Mei / Jiawei Li / Yumi Xuan / Bing Cui / Junjie Zou / Juncheng Wang / Shaoquan Lin / Zhe Zhang / Chongyuan Wang /  Abstract: Pentamidine and melarsoprol are primary drugs used to treat the lethal human sleeping sickness caused by the parasite Trypanosoma brucei. Cross-resistance to these two drugs has recently been linked ...Pentamidine and melarsoprol are primary drugs used to treat the lethal human sleeping sickness caused by the parasite Trypanosoma brucei. Cross-resistance to these two drugs has recently been linked to aquaglyceroporin 2 of the trypanosome (TbAQP2). TbAQP2 is the first member of the aquaporin family described as capable of drug transport; however, the underlying mechanism remains unclear. Here, we present cryo-electron microscopy structures of TbAQP2 bound to pentamidine or melarsoprol. Our structural studies, together with the molecular dynamic simulations, reveal the mechanisms shaping substrate specificity and drug permeation. Multiple amino acids in TbAQP2, near the extracellular entrance and inside the pore, create an expanded conducting tunnel, sterically and energetically allowing the permeation of pentamidine and melarsoprol. Our study elucidates the mechanism of drug transport by TbAQP2, providing valuable insights to inform the design of drugs against trypanosomiasis. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_36721.map.gz emd_36721.map.gz | 59.8 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-36721-v30.xml emd-36721-v30.xml emd-36721.xml emd-36721.xml | 17.9 KB 17.9 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_36721.png emd_36721.png | 131.7 KB | ||
| Filedesc metadata |  emd-36721.cif.gz emd-36721.cif.gz | 6.3 KB | ||
| Others |  emd_36721_half_map_1.map.gz emd_36721_half_map_1.map.gz emd_36721_half_map_2.map.gz emd_36721_half_map_2.map.gz | 58.3 MB 58.4 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-36721 http://ftp.pdbj.org/pub/emdb/structures/EMD-36721 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-36721 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-36721 | HTTPS FTP |
-Related structure data
| Related structure data |  8jy6MC  8jy7C  8jy8C M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_36721.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_36721.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.064 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: Author stated: The maps were generate from non-uniform...
| File | emd_36721_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Author stated: The maps were generate from non-uniform refinement in cryosparc, an algorithm based on cross-validation optimization, which automatically regularizes 3D density maps during refinement to account for spatial variability. Unlike common shift-invariant regularizers, non-uniform refinement systematically removes noise from disordered regions, while retaining signal useful for aligning particle images, yielding dramatically improved resolution and 3D map quality in many cases (Nature methods 17.12 (2020): 1214-1221; https://www.nature.com/articles/s41592-020-00990-8). That is why the raw maps look like filtered. | ||||||||||||
| Projections & Slices |
| ||||||||||||
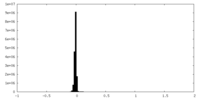
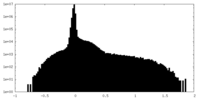
| Density Histograms |
-Half map: #1
| File | emd_36721_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
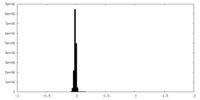
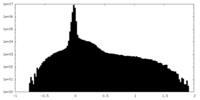
| Density Histograms |
- Sample components
Sample components
-Entire : TbAQP2 complex with melarsorpol
| Entire | Name: TbAQP2 complex with melarsorpol |
|---|---|
| Components |
|
-Supramolecule #1: TbAQP2 complex with melarsorpol
| Supramolecule | Name: TbAQP2 complex with melarsorpol / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: Aquaglyceroporin 2
| Macromolecule | Name: Aquaglyceroporin 2 / type: protein_or_peptide / ID: 1 Details: Author stated: Residues from 313 to 343 are twin-strep tags for affinity purification. Number of copies: 4 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 36.775914 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: MQSQPDNVAY PMELQAVNKD GTVEVRVQGN VDNSSNERWD ADVQKHEVAE AQEKPVGGIN FWAPRELRLN YRDYVAEFLG NFVLIYIAK GAVITSLLVP DFGLLGLTIG IGVAVTMALY VSLGISGGHL NSAVTVGNAV FGDFPWRKVP GYIAAQMLGT F LGAACAYG ...String: MQSQPDNVAY PMELQAVNKD GTVEVRVQGN VDNSSNERWD ADVQKHEVAE AQEKPVGGIN FWAPRELRLN YRDYVAEFLG NFVLIYIAK GAVITSLLVP DFGLLGLTIG IGVAVTMALY VSLGISGGHL NSAVTVGNAV FGDFPWRKVP GYIAAQMLGT F LGAACAYG VFADLLKAHG GGELIAFGEK GIAWVFAMYP AEGNGIFYPI FAELISTAVL LLCVCGIFDP NNSPAKGYET VA IGALVFV MVNNFGLASP LAMNPSLDFG PRVFGAILLG GEVFSHANYY FWVPLVVPFF GAILGLFLYK YFLPHSNSWS HPQ FEKGGG SGGGSGGSAW SHPQFEK UniProtKB: Aquaglyceroporin-2 |
-Macromolecule #2: [(4~{R})-2-[4-[[4,6-bis(azanyl)-1,3,5-triazin-2-yl]amino]phenyl]-...
| Macromolecule | Name: [(4~{R})-2-[4-[[4,6-bis(azanyl)-1,3,5-triazin-2-yl]amino]phenyl]-1,3,2-dithiarsolan-4-yl]methanol type: ligand / ID: 2 / Number of copies: 4 / Formula: L1U |
|---|---|
| Molecular weight | Theoretical: 398.339 Da |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 |
|---|---|
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Average electron dose: 71.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 3.0 µm / Nominal defocus min: 0.8 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller





 X (Sec.)
X (Sec.) Y (Row.)
Y (Row.) Z (Col.)
Z (Col.)