+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of human HCAR2-Gi complex with acipimox | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | complex / MEMBRANE PROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationneutrophil apoptotic process / nicotinic acid receptor activity / Hydroxycarboxylic acid-binding receptors / positive regulation of neutrophil apoptotic process / Class A/1 (Rhodopsin-like receptors) / positive regulation of adiponectin secretion / negative regulation of lipid catabolic process / adenylate cyclase inhibitor activity / positive regulation of protein localization to cell cortex / T cell migration ...neutrophil apoptotic process / nicotinic acid receptor activity / Hydroxycarboxylic acid-binding receptors / positive regulation of neutrophil apoptotic process / Class A/1 (Rhodopsin-like receptors) / positive regulation of adiponectin secretion / negative regulation of lipid catabolic process / adenylate cyclase inhibitor activity / positive regulation of protein localization to cell cortex / T cell migration / positive regulation of relaxation of smooth muscle / Adenylate cyclase inhibitory pathway / response to prostaglandin E / D2 dopamine receptor binding / G protein-coupled serotonin receptor binding / adenylate cyclase-inhibiting serotonin receptor signaling pathway / cellular response to forskolin / chemokine-mediated signaling pathway / regulation of mitotic spindle organization / neuropeptide signaling pathway / Regulation of insulin secretion / positive regulation of cholesterol biosynthetic process / negative regulation of insulin secretion / G protein-coupled receptor binding / response to peptide hormone / adenylate cyclase-modulating G protein-coupled receptor signaling pathway / adenylate cyclase-inhibiting G protein-coupled receptor signaling pathway / G-protein beta/gamma-subunit complex binding / centriolar satellite / Olfactory Signaling Pathway / Activation of the phototransduction cascade / G beta:gamma signalling through PLC beta / Presynaptic function of Kainate receptors / Thromboxane signalling through TP receptor / G protein-coupled acetylcholine receptor signaling pathway / Activation of G protein gated Potassium channels / Inhibition of voltage gated Ca2+ channels via Gbeta/gamma subunits / G-protein activation / G beta:gamma signalling through CDC42 / Prostacyclin signalling through prostacyclin receptor / Glucagon signaling in metabolic regulation / G beta:gamma signalling through BTK / Synthesis, secretion, and inactivation of Glucagon-like Peptide-1 (GLP-1) / ADP signalling through P2Y purinoceptor 12 / photoreceptor disc membrane / GDP binding / Glucagon-type ligand receptors / Sensory perception of sweet, bitter, and umami (glutamate) taste / Adrenaline,noradrenaline inhibits insulin secretion / cell junction / Vasopressin regulates renal water homeostasis via Aquaporins / Glucagon-like Peptide-1 (GLP1) regulates insulin secretion / G alpha (z) signalling events / ADP signalling through P2Y purinoceptor 1 / ADORA2B mediated anti-inflammatory cytokines production / cellular response to catecholamine stimulus / G beta:gamma signalling through PI3Kgamma / adenylate cyclase-activating dopamine receptor signaling pathway / sperm principal piece / Cooperation of PDCL (PhLP1) and TRiC/CCT in G-protein beta folding / GPER1 signaling / cellular response to prostaglandin E stimulus / G-protein beta-subunit binding / heterotrimeric G-protein complex / G alpha (12/13) signalling events / Inactivation, recovery and regulation of the phototransduction cascade / extracellular vesicle / sensory perception of taste / adenylate cyclase-activating G protein-coupled receptor signaling pathway / Thrombin signalling through proteinase activated receptors (PARs) / signaling receptor complex adaptor activity / retina development in camera-type eye / G protein activity / GTPase binding / fibroblast proliferation / Ca2+ pathway / midbody / cell cortex / High laminar flow shear stress activates signaling by PIEZO1 and PECAM1:CDH5:KDR in endothelial cells / G alpha (i) signalling events / G alpha (s) signalling events / phospholipase C-activating G protein-coupled receptor signaling pathway / G alpha (q) signalling events / Hydrolases; Acting on acid anhydrides; Acting on GTP to facilitate cellular and subcellular movement / Ras protein signal transduction / Extra-nuclear estrogen signaling / cell population proliferation / ciliary basal body / G protein-coupled receptor signaling pathway / cell division / lysosomal membrane / GTPase activity / synapse / centrosome / GTP binding / protein-containing complex binding / nucleolus / magnesium ion binding / Golgi apparatus / signal transduction Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.23 Å | |||||||||
 Authors Authors | Pan X / Fang Y | |||||||||
| Funding support |  China, 1 items China, 1 items
| |||||||||
 Citation Citation |  Journal: Cell Discov / Year: 2023 Journal: Cell Discov / Year: 2023Title: Structural insights into ligand recognition and selectivity of the human hydroxycarboxylic acid receptor HCAR2. Authors: Xin Pan / Fang Ye / Peiruo Ning / Zhiyi Zhang / Xinyu Li / Binghao Zhang / Qian Wang / Geng Chen / Wei Gao / Chen Qiu / Zhangsong Wu / Jiancheng Li / Lizhe Zhu / Jiang Xia / Kaizheng Gong / Yang Du /  Abstract: Hydroxycarboxylic acid receptor 2 (HCAR2) belongs to the family of class A G protein-coupled receptors with key roles in regulating lipolysis and free fatty acid formation in humans. It is deeply ...Hydroxycarboxylic acid receptor 2 (HCAR2) belongs to the family of class A G protein-coupled receptors with key roles in regulating lipolysis and free fatty acid formation in humans. It is deeply involved in many pathophysiological processes and serves as an attractive target for the treatment of cardiovascular, neoplastic, autoimmune, neurodegenerative, inflammatory, and metabolic diseases. Here, we report four cryo-EM structures of human HCAR2-Gi1 complexes with or without agonists, including the drugs niacin (2.69 Å) and acipimox (3.23 Å), the highly subtype-specific agonist MK-6892 (3.25 Å), and apo form (3.28 Å). Combined with molecular dynamics simulation and functional analysis, we have revealed the recognition mechanism of HCAR2 for different agonists and summarized the general pharmacophore features of HCAR2 agonists, which are based on three key residues R111, S179, and Y284. Notably, the MK-6892-HCAR2 structure shows an extended binding pocket relative to other agonist-bound HCAR2 complexes. In addition, the key residues that determine the ligand selectivity between the HCAR2 and HCAR3 are also illuminated. Our findings provide structural insights into the ligand recognition, selectivity, activation, and G protein coupling mechanism of HCAR2, which shed light on the design of new HCAR2-targeting drugs for greater efficacy, higher selectivity, and fewer or no side effects. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_35484.map.gz emd_35484.map.gz | 64.3 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-35484-v30.xml emd-35484-v30.xml emd-35484.xml emd-35484.xml | 18.9 KB 18.9 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_35484.png emd_35484.png | 109.6 KB | ||
| Filedesc metadata |  emd-35484.cif.gz emd-35484.cif.gz | 6.4 KB | ||
| Others |  emd_35484_half_map_1.map.gz emd_35484_half_map_1.map.gz emd_35484_half_map_2.map.gz emd_35484_half_map_2.map.gz | 117.9 MB 118 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-35484 http://ftp.pdbj.org/pub/emdb/structures/EMD-35484 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-35484 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-35484 | HTTPS FTP |
-Related structure data
| Related structure data |  8ijbMC  8ij3C  8ijaC  8ijdC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_35484.map.gz / Format: CCP4 / Size: 125 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_35484.map.gz / Format: CCP4 / Size: 125 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.85 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #2
| File | emd_35484_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #1
| File | emd_35484_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Cryo-EM structure of human HCAR2-Gi complex with acipimox
| Entire | Name: Cryo-EM structure of human HCAR2-Gi complex with acipimox |
|---|---|
| Components |
|
-Supramolecule #1: Cryo-EM structure of human HCAR2-Gi complex with acipimox
| Supramolecule | Name: Cryo-EM structure of human HCAR2-Gi complex with acipimox type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#5 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: Hydroxycarboxylic acid receptor 2
| Macromolecule | Name: Hydroxycarboxylic acid receptor 2 / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 34.251973 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: DHFLEIDKKN CCVFRDDFIV KVLPPVLGLE FIFGLLGNGL ALWIFCFHLK SWKSSRIFLF NLAVADFLLI ICLPFLMDNY VRRWDWKFG DIPCRLMLFM LAMNRQGSII FLTVVAVDRY FRVVHPHHAL NKISNRTAAI ISCLLWGITI GLTVHLLKKK M PIQNGGAN ...String: DHFLEIDKKN CCVFRDDFIV KVLPPVLGLE FIFGLLGNGL ALWIFCFHLK SWKSSRIFLF NLAVADFLLI ICLPFLMDNY VRRWDWKFG DIPCRLMLFM LAMNRQGSII FLTVVAVDRY FRVVHPHHAL NKISNRTAAI ISCLLWGITI GLTVHLLKKK M PIQNGGAN LCSSFSICHT FQWHEAMFLL EFFLPLGIIL FCSARIIWSL RQRQMDRHAK IKRAITFIMV VAIVFVICFL PS VVVRIRI FWLLHTSGTQ NCEVYRSVDL AFFITLSFTY MNSMLDPVVY YFSSPSF UniProtKB: Hydroxycarboxylic acid receptor 2 |
-Macromolecule #2: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1
| Macromolecule | Name: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1 type: protein_or_peptide / ID: 2 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 37.069543 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: LDQLRQEAEQ LKNQIRDARK ACADATLSQI TNNIDPVGRI QMRTRRTLRG HLAKIYAMHW GTDSRLLVSA SQDGKLIIWD SYTTNKVHA IPLRSSWVMT CAYAPSGNYV ACGGLDNICS IYNLKTREGN VRVSRELAGH TGYLSCCRFL DDNQIVTSSG D TTCALWDI ...String: LDQLRQEAEQ LKNQIRDARK ACADATLSQI TNNIDPVGRI QMRTRRTLRG HLAKIYAMHW GTDSRLLVSA SQDGKLIIWD SYTTNKVHA IPLRSSWVMT CAYAPSGNYV ACGGLDNICS IYNLKTREGN VRVSRELAGH TGYLSCCRFL DDNQIVTSSG D TTCALWDI ETGQQTTTFT GHTGDVMSLS LAPDTRLFVS GACDASAKLW DVREGMCRQT FTGHESDINA ICFFPNGNAF AT GSDDATC RLFDLRADQE LMTYSHDNII CGITSVSFSK SGRLLLAGYD DFNCNVWDAL KADRAGVLAG HDNRVSCLGV TDD GMAVAT GSWDSFLKIW N UniProtKB: Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1 |
-Macromolecule #3: Guanine nucleotide-binding protein G(i) subunit alpha-1
| Macromolecule | Name: Guanine nucleotide-binding protein G(i) subunit alpha-1 type: protein_or_peptide / ID: 3 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 40.153672 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: TLSAEDKAAV ERSKMIDRNL REDGEKAARE VKLLLLGAGE SGKSTIVKQM KIIHEAGYSE EECKQYKAVV YSNTIQSIIA IIRAMGRLK IDFGDSARAD DARQLFVLAG AAEEGFMTAE LAGVIKRLWK DSGVQACFNR SREYQLNDSA AYYLNDLDRI A QPNYIPTQ ...String: TLSAEDKAAV ERSKMIDRNL REDGEKAARE VKLLLLGAGE SGKSTIVKQM KIIHEAGYSE EECKQYKAVV YSNTIQSIIA IIRAMGRLK IDFGDSARAD DARQLFVLAG AAEEGFMTAE LAGVIKRLWK DSGVQACFNR SREYQLNDSA AYYLNDLDRI A QPNYIPTQ QDVLRTRVKT TGIVETHFTF KDLHFKMFDV GAQRSERKKW IHCFEGVTAI IFCVALSDYD LVLAEDEEMN RM HESMKLF DSICNNKWFT DTSIILFLNK KDLFEEKIKK SPLTICYPEY AGSNTYEEAA AYIQCQFEDL NKRKDTKEIY THF TCSTDT KNVQFVFDAV TDVIIKNNLK DCGLF UniProtKB: Guanine nucleotide-binding protein G(i) subunit alpha-1 |
-Macromolecule #4: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2
| Macromolecule | Name: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2 type: protein_or_peptide / ID: 4 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 6.218162 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: SIAQARKLVE QLKMEANIDR IKVSKAAADL MAYCEAHAKE DPLLTPVPAS ENPFRE UniProtKB: Guanine nucleotide-binding protein G(I)/G(S)/G(O) subunit gamma-2 |
-Macromolecule #5: scFv16
| Macromolecule | Name: scFv16 / type: protein_or_peptide / ID: 5 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 26.424385 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: DVQLVESGGG LVQPGGSRKL SCSASGFAFS SFGMHWVRQA PEKGLEWVAY ISSGSGTIYY ADTVKGRFTI SRDDPKNTLF LQMTSLRSE DTAMYYCVRS IYYYGSSPFD FWGQGTTLTV SSGGGGSGGG GSGGGGSSDI VMTQATSSVP VTPGESVSIS C RSSKSLLH ...String: DVQLVESGGG LVQPGGSRKL SCSASGFAFS SFGMHWVRQA PEKGLEWVAY ISSGSGTIYY ADTVKGRFTI SRDDPKNTLF LQMTSLRSE DTAMYYCVRS IYYYGSSPFD FWGQGTTLTV SSGGGGSGGG GSGGGGSSDI VMTQATSSVP VTPGESVSIS C RSSKSLLH SNGNTYLYWF LQRPGQSPQL LIYRMSNLAS GVPDRFSGSG SGTAFTLTIS RLEAEDVGVY YCMQHLEYPL TF GAGTKLE L |
-Macromolecule #6: 5-methyl-4-oxidanyl-pyrazin-4-ium-2-carboxylic acid
| Macromolecule | Name: 5-methyl-4-oxidanyl-pyrazin-4-ium-2-carboxylic acid / type: ligand / ID: 6 / Number of copies: 1 / Formula: OJX |
|---|---|
| Molecular weight | Theoretical: 155.131 Da |
| Chemical component information | 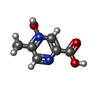 ChemComp-OJX: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.2 |
|---|---|
| Vitrification | Cryogen name: NITROGEN |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Average electron dose: 56.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: SPOT SCAN / Imaging mode: BRIGHT FIELD / Nominal defocus max: 1.4000000000000001 µm / Nominal defocus min: 1.0 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: NONE |
|---|---|
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 3.23 Å / Resolution method: FSC 0.143 CUT-OFF / Number images used: 221940 |
| Initial angle assignment | Type: NOT APPLICABLE |
| Final angle assignment | Type: NOT APPLICABLE |
 Movie
Movie Controller
Controller



































 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)




































