+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of SPT-ORMDL3 (ORMDL3-N13A) complex | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | ceramide / TRANSFERASE-inhibitor complex | |||||||||
| Function / homology |  Function and homology information Function and homology informationsphinganine biosynthetic process / regulation of fat cell apoptotic process / negative regulation of ceramide biosynthetic process / intracellular sphingolipid homeostasis / serine palmitoyltransferase complex / sphingomyelin biosynthetic process / serine C-palmitoyltransferase / serine C-palmitoyltransferase activity / ceramide metabolic process / sphingosine biosynthetic process ...sphinganine biosynthetic process / regulation of fat cell apoptotic process / negative regulation of ceramide biosynthetic process / intracellular sphingolipid homeostasis / serine palmitoyltransferase complex / sphingomyelin biosynthetic process / serine C-palmitoyltransferase / serine C-palmitoyltransferase activity / ceramide metabolic process / sphingosine biosynthetic process / sphingolipid metabolic process / sphingolipid biosynthetic process / Sphingolipid de novo biosynthesis / regulation of smooth muscle contraction / ceramide biosynthetic process / positive regulation of lipophagy / negative regulation of B cell apoptotic process / motor behavior / adipose tissue development / specific granule membrane / myelination / positive regulation of autophagy / secretory granule membrane / positive regulation of protein localization to nucleus / intracellular protein localization / pyridoxal phosphate binding / Neutrophil degranulation / endoplasmic reticulum membrane / endoplasmic reticulum / plasma membrane Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 2.9 Å | |||||||||
 Authors Authors | Xie T / Liu P / Gong X | |||||||||
| Funding support | 1 items
| |||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2023 Journal: Nat Commun / Year: 2023Title: Ceramide sensing by human SPT-ORMDL complex for establishing sphingolipid homeostasis. Authors: Tian Xie / Peng Liu / Xinyue Wu / Feitong Dong / Zike Zhang / Jian Yue / Usha Mahawar / Faheem Farooq / Hisham Vohra / Qi Fang / Wenchen Liu / Binks W Wattenberg / Xin Gong /   Abstract: The ORM/ORMDL family proteins function as regulatory subunits of the serine palmitoyltransferase (SPT) complex, which is the initiating and rate-limiting enzyme in sphingolipid biosynthesis. This ...The ORM/ORMDL family proteins function as regulatory subunits of the serine palmitoyltransferase (SPT) complex, which is the initiating and rate-limiting enzyme in sphingolipid biosynthesis. This complex is tightly regulated by cellular sphingolipid levels, but the sphingolipid sensing mechanism is unknown. Here we show that purified human SPT-ORMDL complexes are inhibited by the central sphingolipid metabolite ceramide. We have solved the cryo-EM structure of the SPT-ORMDL3 complex in a ceramide-bound state. Structure-guided mutational analyses reveal the essential function of this ceramide binding site for the suppression of SPT activity. Structural studies indicate that ceramide can induce and lock the N-terminus of ORMDL3 into an inhibitory conformation. Furthermore, we demonstrate that childhood amyotrophic lateral sclerosis (ALS) variants in the SPTLC1 subunit cause impaired ceramide sensing in the SPT-ORMDL3 mutants. Our work elucidates the molecular basis of ceramide sensing by the SPT-ORMDL complex for establishing sphingolipid homeostasis and indicates an important role of impaired ceramide sensing in disease development. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_33869.map.gz emd_33869.map.gz | 59.8 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-33869-v30.xml emd-33869-v30.xml emd-33869.xml emd-33869.xml | 21.2 KB 21.2 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_33869.png emd_33869.png | 42 KB | ||
| Filedesc metadata |  emd-33869.cif.gz emd-33869.cif.gz | 6.7 KB | ||
| Others |  emd_33869_half_map_1.map.gz emd_33869_half_map_1.map.gz emd_33869_half_map_2.map.gz emd_33869_half_map_2.map.gz | 49.4 MB 49.4 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-33869 http://ftp.pdbj.org/pub/emdb/structures/EMD-33869 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-33869 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-33869 | HTTPS FTP |
-Related structure data
| Related structure data |  7yj2MC  7yiuC  7yiyC  7yj1C M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_33869.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_33869.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.08 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #1
| File | emd_33869_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
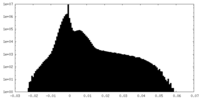
| Projections & Slices |
| ||||||||||||
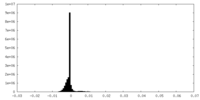
| Density Histograms |
-Half map: #2
| File | emd_33869_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
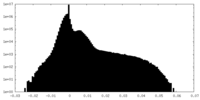
| Projections & Slices |
| ||||||||||||
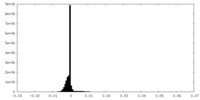
| Density Histograms |
- Sample components
Sample components
-Entire : SPT-ORMDL3 complex
| Entire | Name: SPT-ORMDL3 complex |
|---|---|
| Components |
|
-Supramolecule #1: SPT-ORMDL3 complex
| Supramolecule | Name: SPT-ORMDL3 complex / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#4 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: Serine palmitoyltransferase 2
| Macromolecule | Name: Serine palmitoyltransferase 2 / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO / EC number: serine C-palmitoyltransferase |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 63.00416 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: MRPEPGGCCC RRTVRANGCV ANGEVRNGYV RSSAAAAAAA AAGQIHHVTQ NGGLYKRPFN EAFEETPMLV AVLTYVGYGV LTLFGYLRD FLRYWRIEKC HHATEREEQK DFVSLYQDFE NFYTRNLYMR IRDNWNRPIC SVPGARVDIM ERQSHDYNWS F KYTGNIIK ...String: MRPEPGGCCC RRTVRANGCV ANGEVRNGYV RSSAAAAAAA AAGQIHHVTQ NGGLYKRPFN EAFEETPMLV AVLTYVGYGV LTLFGYLRD FLRYWRIEKC HHATEREEQK DFVSLYQDFE NFYTRNLYMR IRDNWNRPIC SVPGARVDIM ERQSHDYNWS F KYTGNIIK GVINMGSYNY LGFARNTGSC QEAAAKVLEE YGAGVCSTRQ EIGNLDKHEE LEELVARFLG VEAAMAYGMG FA TNSMNIP ALVGKGCLIL SDELNHASLV LGARLSGATI RIFKHNNMQS LEKLLKDAIV YGQPRTRRPW KKILILVEGI YSM EGSIVR LPEVIALKKK YKAYLYLDEA HSIGALGPTG RGVVEYFGLD PEDVDVMMGT FTKSFGASGG YIGGKKELID YLRT HSHSA VYATSLSPPV VEQIITSMKC IMGQDGTSLG KECVQQLAEN TRYFRRRLKE MGFIIYGNED SPVVPLMLYM PAKIG AFGR EMLKRNIGVV VVGFPATPII ESRARFCLSA AHTKEILDTA LKEIDEVGDL LQLKYSRHRL VPLLDRPFDE TTYEET ED UniProtKB: Serine palmitoyltransferase 2 |
-Macromolecule #2: Serine palmitoyltransferase 1
| Macromolecule | Name: Serine palmitoyltransferase 1 / type: protein_or_peptide / ID: 2 / Number of copies: 1 / Enantiomer: LEVO / EC number: serine C-palmitoyltransferase |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 5.898994 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: MATATEQWVL VEMVQALYEA PAYHLILEGI LILWIIRLLF SKTYKLQERS UniProtKB: Serine palmitoyltransferase 1 |
-Macromolecule #3: ORM1-like protein 3
| Macromolecule | Name: ORM1-like protein 3 / type: protein_or_peptide / ID: 3 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 17.469568 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: MNVGTAHSEV NPATRVMNSR GIWLSYVLAI GLLHIVLLSI PFVSVPVVWT LTNLIHNMGM YIFLHTVKGT PFETPDQGKA RLLTHWEQM DYGVQFTASR KFLTITPIVL YFLTSFYTKY DQIHFVLNTV SLMSVLIPKL PQLHGVRIFG INKY UniProtKB: ORM1-like protein 3 |
-Macromolecule #4: Serine palmitoyltransferase small subunit A
| Macromolecule | Name: Serine palmitoyltransferase small subunit A / type: protein_or_peptide / ID: 4 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 10.742409 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: MADYKDDDDK SGPDEVDASG RMAGMALARA WKQMSWFYYQ YLLVTALYML EPWERTVFNS MLVSIVGMAL YTGYVFMPQH IMAILHYFE IVQ UniProtKB: Serine palmitoyltransferase small subunit A |
-Macromolecule #5: Serine palmitoyltransferase 1
| Macromolecule | Name: Serine palmitoyltransferase 1 / type: protein_or_peptide / ID: 5 / Number of copies: 1 / Enantiomer: LEVO / EC number: serine C-palmitoyltransferase |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 46.925828 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: DLTVKEKEEL IEEWQPEPLV PPVPKDHPAL NYNIVSGPPS HKTVVNGKEC INFASFNFLG LLDNPRVKAA ALASLKKYGV GTCGPRGFY GTFDVHLDLE DRLAKFMKTE EAIIYSYGFA TIASAIPAYS KRGDIVFVDR AACFAIQKGL QASRSDIKLF K HNDMADLE ...String: DLTVKEKEEL IEEWQPEPLV PPVPKDHPAL NYNIVSGPPS HKTVVNGKEC INFASFNFLG LLDNPRVKAA ALASLKKYGV GTCGPRGFY GTFDVHLDLE DRLAKFMKTE EAIIYSYGFA TIASAIPAYS KRGDIVFVDR AACFAIQKGL QASRSDIKLF K HNDMADLE RLLKEQEIED QKNPRKARVT RRFIVVEGLY MNTGTICPLP ELVKLKYKYK ARIFLEESLS FGVLGEHGRG VT EHYGINI DDIDLISANM ENALASIGGF CCGRSFVIDH QRLSGQGYCF SASLPPLLAA AAIEALNIME ENPGIFAVLK EKC GQIHKA LQGISGLKVV GESLSPAFHL QLEESTGSRE QDVRLLQEIV DQCMNRSIAL TQARYLEKEE KCLPPPSIRV VVTV EQTEE ELERAASTIK EVAQAVLL UniProtKB: Serine palmitoyltransferase 1 |
-Macromolecule #6: PYRIDOXAL-5'-PHOSPHATE
| Macromolecule | Name: PYRIDOXAL-5'-PHOSPHATE / type: ligand / ID: 6 / Number of copies: 1 / Formula: PLP |
|---|---|
| Molecular weight | Theoretical: 247.142 Da |
| Chemical component information |  ChemComp-PLP: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Average electron dose: 50.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.0 µm / Nominal defocus min: 1.0 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller






 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)