[English] 日本語
 Yorodumi
Yorodumi- EMDB-33468: cryo-EM structure of HK022 putRNA-less E.coli RNA polymerase elon... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
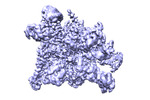
| Title | cryo-EM structure of HK022 putRNA-less E.coli RNA polymerase elongation complex | ||||||||||||||||||
 Map data Map data | local-filtered map after post-process | ||||||||||||||||||
 Sample Sample |
| ||||||||||||||||||
 Keywords Keywords | bacterial transcription / HK022 put / anti-termination / anti-pausing / cryo-EM / TRANSCRIPTION | ||||||||||||||||||
| Function / homology |  Function and homology information Function and homology informationRNA polymerase complex / submerged biofilm formation / cellular response to cell envelope stress / cytosolic DNA-directed RNA polymerase complex / regulation of DNA-templated transcription initiation / bacterial-type flagellum assembly / bacterial-type flagellum-dependent cell motility / nitrate assimilation / transcription elongation factor complex / regulation of DNA-templated transcription elongation ...RNA polymerase complex / submerged biofilm formation / cellular response to cell envelope stress / cytosolic DNA-directed RNA polymerase complex / regulation of DNA-templated transcription initiation / bacterial-type flagellum assembly / bacterial-type flagellum-dependent cell motility / nitrate assimilation / transcription elongation factor complex / regulation of DNA-templated transcription elongation / transcription antitermination / DNA-templated transcription initiation / cell motility / ribonucleoside binding / DNA-directed 5'-3' RNA polymerase activity / DNA-directed RNA polymerase / response to heat / protein-containing complex assembly / intracellular iron ion homeostasis / protein dimerization activity / response to antibiotic / magnesium ion binding / DNA binding / zinc ion binding / membrane / cytoplasm / cytosol Similarity search - Function | ||||||||||||||||||
| Biological species |   Shamshuipovirus HK022 Shamshuipovirus HK022 | ||||||||||||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.57 Å | ||||||||||||||||||
 Authors Authors | Hwang S / Kang JY | ||||||||||||||||||
| Funding support |  Korea, Republic Of, Korea, Republic Of,  United States, 5 items United States, 5 items
| ||||||||||||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2022 Journal: Nat Commun / Year: 2022Title: Structural basis of transcriptional regulation by a nascent RNA element, HK022 putRNA. Authors: Seungha Hwang / Paul Dominic B Olinares / Jimin Lee / Jinwoo Kim / Brian T Chait / Rodney A King / Jin Young Kang /   Abstract: Transcription, in which RNA polymerases (RNAPs) produce RNA from DNA, is the first step of gene expression. As such, it is highly regulated either by trans-elements like protein factors and/or by cis- ...Transcription, in which RNA polymerases (RNAPs) produce RNA from DNA, is the first step of gene expression. As such, it is highly regulated either by trans-elements like protein factors and/or by cis-elements like specific sequences on the DNA. Lambdoid phage HK022 contains a cis-element, put, which suppresses pausing and termination during transcription of the early phage genes. The putRNA transcript solely performs the anti-pausing/termination activities by interacting directly with the E.coli RNAP elongation complex (EC) by an unknown structural mechanism. In this study, we reconstituted putRNA-associated ECs and determined the structures using cryo-electron microscopy. The determined structures of putRNA-associated EC, putRNA-absent EC, and σ-bound EC suggest that the putRNA interaction with the EC counteracts swiveling, a conformational change previously identified to promote pausing and σ might modulate putRNA folding via σ-dependent pausing during elongation. | ||||||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_33468.map.gz emd_33468.map.gz | 2.7 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-33468-v30.xml emd-33468-v30.xml emd-33468.xml emd-33468.xml | 27.5 KB 27.5 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_33468.png emd_33468.png | 42.7 KB | ||
| Filedesc metadata |  emd-33468.cif.gz emd-33468.cif.gz | 9.3 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-33468 http://ftp.pdbj.org/pub/emdb/structures/EMD-33468 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-33468 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-33468 | HTTPS FTP |
-Validation report
| Summary document |  emd_33468_validation.pdf.gz emd_33468_validation.pdf.gz | 367.4 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_33468_full_validation.pdf.gz emd_33468_full_validation.pdf.gz | 366.9 KB | Display | |
| Data in XML |  emd_33468_validation.xml.gz emd_33468_validation.xml.gz | 6.3 KB | Display | |
| Data in CIF |  emd_33468_validation.cif.gz emd_33468_validation.cif.gz | 7.2 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-33468 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-33468 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-33468 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-33468 | HTTPS FTP |
-Related structure data
| Related structure data |  7xugMC  7xueC  7xuiC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_33468.map.gz / Format: CCP4 / Size: 98.9 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_33468.map.gz / Format: CCP4 / Size: 98.9 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | local-filtered map after post-process | ||||||||||||||||||||
| Voxel size | X=Y=Z: 1.06 Å | ||||||||||||||||||||
| Density |
| ||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
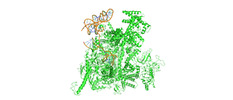
- Sample components
Sample components
+Entire : HK022 putRNA-less E.coli RNA polymerase elongation complex
+Supramolecule #1: HK022 putRNA-less E.coli RNA polymerase elongation complex
+Supramolecule #2: E.coli RNA polymerase
+Supramolecule #3: DNA/RNA
+Macromolecule #1: non-template DNA
+Macromolecule #2: template DNA
+Macromolecule #3: RNA (nun gene and immunity region)
+Macromolecule #4: DNA-directed RNA polymerase subunit alpha
+Macromolecule #5: DNA-directed RNA polymerase subunit beta
+Macromolecule #6: DNA-directed RNA polymerase subunit beta'
+Macromolecule #7: DNA-directed RNA polymerase subunit omega
+Macromolecule #8: ZINC ION
+Macromolecule #9: MAGNESIUM ION
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 4 mg/mL | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 8 Component:
Details: pH adjustment at 4'C | |||||||||||||||
| Grid | Model: Quantifoil R1.2/1.3 / Material: COPPER / Mesh: 400 / Support film - Material: CARBON / Support film - topology: HOLEY / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 25 sec. / Pretreatment - Atmosphere: AIR / Pretreatment - Pressure: 0.026000000000000002 kPa | |||||||||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 295 K / Instrument: FEI VITROBOT MARK IV / Details: using two layers of blot papers. |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Specialist optics | Energy filter - Name: GIF Bioquantum / Energy filter - Slit width: 20 eV |
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Number grids imaged: 1 / Number real images: 8175 / Average electron dose: 42.16 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 70.0 µm / Illumination mode: OTHER / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 2.6 µm / Nominal defocus min: 0.8 µm |
| Sample stage | Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller